Hospital antimicrobial stewardship programmes have achieved savings and a more rational use of antimicrobial treatments in general wards. The purpose of this report is to evaluate the experience of an antimicrobial stewardship programme in an intensive care unit (ICU).
DesignProspective interventional, before-and-after study.
Scope24-bed medical ICU in a tertiary hospital.
InterventionProspective audit and feedback antimicrobial stewardship programme.
EndpointsAntimicrobial consumption, antimicrobial related costs, multi-drug resistant microorganisms (MDRM) prevalence, nosocomial infections incidence, ICU length of stay, and ICU mortality rates were compared before and after one-year intervention.
ResultsA total of 218 antimicrobial episodes of 182 patients were evaluated in 61 team meetings. Antimicrobial stewardship suggestions were accepted in 91.5% of the cases. Total antimicrobial DDD/100 patient-days consumption was reduced from 380.6 to 295.2 (−22.4%; p=0.037). Antimicrobial stewardship programme was associated with a significant decrease in the prescription of penicillins plus b-lactamase inhibitors, linezolid, cephalosporins, and aminoglycosides. Overall antimicrobial spending was reduced by €119,636. MDRM isolation and nosocomial infections per 100 patient-days did not change after the intervention period. No changes in length of stay or mortality rate were observed.
ConclusionsAn ICU antimicrobial stewardship programme significantly reduced antimicrobial use without affecting inpatient mortality and length of stay. Our results further support the implementation of an antimicrobial stewardship programme in critical care units.
Los programas de optimización de antimicrobianos (PROA) han demostrado ser herramientas eficaces para reducir el uso de antimicrobianos. El propósito de este estudio es evaluar el efecto de la implantación de un PROA en una unidad de cuidados intensivos (UCI).
DiseñoEstudio prospectivo de intervención.
ÁmbitoUCI de 24 camas en un hospital terciario.
IntervenciónImplantación de un PROA basado en auditorías prospectivas.
Variables de interés principalesEl consumo de antimicrobianos y sus los costes, la prevalencia de colonización por bacterias multi-resistentes (BMR), la incidencia de infección nosocomial, la estancia en UCI y las tasas de mortalidad fueron comparadas antes y tras un año de implantación del programa.
ResultadosSe evaluaron un total de 218 episodios antimicrobianos de 182 pacientes en 61 reuniones. Él 91,5% de las sugerencias dadas por el equipo PROA fueron aceptadas por el prescriptor. El consumo total de antimicrobianos en DDD/100 estancias se redujo de 380,6 a 295,2 (-22,4%, p=0,037). La implantación del PROA se asoció con una disminución significativa en la prescripción de penicilinas/inhibidores de b-lactamasa, linezolid, cefalosporinas y aminoglucósidos. El gasto total en antimicrobianos se redujo en 119.636 €. La incidencia de colonización por BMR y de infecciones nosocomiales no cambiaron tras del período de intervención. No se observaron cambios en la duración de la estancia ni en la tasa de mortalidad.
ConclusionesLa implantación de un PROA en una UCI reduce significativamente el uso de antimicrobianos sin afectar a la evolución de los pacientes ingresados. Nuestros resultados apoyan la implementación de este tipo de programas en las unidades de pacientes críticos.
Over the past two decades there has been an exponential increase in bacterial resistance worldwide.1,2 The inappropriate use of antimicrobial drugs favours the emergence and selection of resistant strains, and poor infection prevention and control practices contribute to further emergence and spread of resistance. Several studies have reported that high prevalence of antibiotics prescription is related directly to high antibiotic resistance rates, as well as to a significant increase in morbidity and mortality.3,4 These facts are particularly relevant in the hospital environment, since it has been reported that up to 50% of the prescribed antibiotic treatments are inadequate or incorrect.5 Social costs caused by antibiotic resistance have been estimated at 1.5 billion Euros, and furthermore antibiotic resistance has been linked to 25,000 deaths per year in the European Union.6
Nowadays, antimicrobial resistance has been identified as a serious problem in intensive care units (ICU).7 In the critically ill patient, a prompt institution of an effective antimicrobial therapy against the causative pathogens is crucial for the prognosis of those patients with severe infections and sepsis.8 However, this situation often leads to an indiscriminate use of broad-spectrum antibiotics during prolonged periods, resulting in a selective pressure, and therefore to the development of multidrug resistant bacteria, with the consequent effects on morbidity, mortality and costs.9,10
Antimicrobial consumption restraint has been identified as an effective measure to reduce bacterial resistance emergence.11 Along this line, antimicrobial stewardship (AMS) interventions have the greatest impact on patient outcomes.12 AMS programmes are a set of multidisciplinary interventions that aim to ensure the rational use of antimicrobials by preventing their unnecessary use, and to provide targeted and limited therapy in situations where they are warranted.13 Developing of this type of antimicrobial programmes has been stimulated by scientific societies around the world, and has been shown to represent a critical step in the control of bacterial resistance and spending on antimicrobials.14 Among different AMS interventions, prospective audit and feedback have demonstrated to be effective and tolerable. With the aim of optimising antimicrobials use in Spanish hospitals, in 2012 several scientific societies produced an AMS consensus document.15 Since then, more and more units are introducing such programmes in their centres. However, few experiences of AMS interventions based on prospective audit and feedback in critical care units have been reported. The aim of this study describes the implementation and the effects of new AMS practices in the ICU of our hospital.
Patients and methodsA before and after study was designed, in order to evaluate the influence of an AMS programme on antimicrobial consumption and in multi-drug resistant microorganisms (MDRM) prevalence. Patients admitted to our ICU from October 2013 to September 2014 formed the control group. The intervention period was from October 2014 to September 2015. Antimicrobial consumption, costs, MDRM prevalence, nosocomial infection incidence (including urinary tract infection, ventilation associated pneumonia and catheter-related infection), ICU mortality, and ICU length of stay were compared between periods.
The study was performed in a tertiary academic teaching hospital with 984 beds. The medical ICU of the hospital has 24 beds, with a mean of 1592 admissions annually during the last five years. Our ICU nosocomial infection control programme includes the performance of surveillance cultures (oral and rectal swabs and tracheobronchial aspirates in ventilated patients) on ICU admission, and weekly to detect MDRM colonisation, as well as a daily meeting with microbiologists, infectious diseases physicians and staff members of the infection control department (to assess the situation of nosocomial infection, prevalence of MDRM, and design appropriate interventions). In those patients with MDRM colonisation/infection, special surveillance and control measures were taken to prevent microbial dissemination.
In October 2014 prospective audit and feedback antimicrobial stewardship programme strategies were put into practice. The AMS team included an intensivist physician specialising in infectious diseases and a pharmacist. The pharmacist reviewed every patient with antimicrobial prescriptions daily, and those who met previously established alarm criteria were discussed with the intensivist in a 20min dedicated meeting. Alarm criteria were the following: (1) antimicrobial prescription of carbapenems, linezolid, tigecycline, daptomycin, colistin and antifungal agents (including azoles, equinocandines and amphotericin B); (2) Treatment durations exceeding the recommendations established in the local treatment guidelines for each type of infection (3) Dosage not considered appropriate to patient condition; (4) Treatment adjustment (including de-escalation) to microbiological results, and (5) Relevant interactions and adverse events associated with antimicrobial therapy.
If any antimicrobial treatment modification was recommended in the AMS meeting, a face-to-face interview was held between AMS team and the attending physician. In the end, the prescribing physician would decide whether or not to accept the recommendation. Patient data and actions taken by the AMS team were entered into a database. The degree of acceptance of the recommendation was recorded by reviewing the patient's prescription the day after the intervention. Monthly indicators of AMS team operations and antimicrobial consumption were obtained.
Demographic and clinical data, cause of ICU admission, APACHE-II score calculated during the first 24h of admission, relevant findings during ICU stay, antimicrobial and non-antimicrobial treatments, and outcome data were recorded. Total antimicrobial consumption and divided by therapeutic groups was calculated based on WHO standards,16 which is the cumulative antibacterial use expressed as DDD per 100 patient-days. Antimicrobials cost was obtained from the stock management system and dispensing software used in the hospital. Total cost of antimicrobials consumption was calculated based on the selling price (PVL) at the beginning of the pre-intervention period (September 2013).17
Time series analysis was used to evaluate the trends in ICU and total hospital antimicrobial consumption. Linear regression was carried out in order to assess the changes in trends (slopes) of ICU and hospital antimicrobial usage after AMS implementation. The linear trend by month is defined as the slope of the response over time, each expressed by a coefficient (β).
The following pathogens were considered as MDRM, according to an international expert consensus18: Methicillin-resistant Staphylococcus aureus (MRSA), extended spectrum β-lactamase (ESBL) producing Enterobacteriaceae and carbapenem-resistant non-fermenting organisms such as Acinetobacter baumannii and Pseudomonas aeruginosa. Patients colonised with these MDRM were identified through the epidemiological surveillance cultures. MDRM prevalence was calculated based on total MDRM isolated from epidemiological surveillance samples, adjusted by patient admissions (number of MDRD isolated/100 admissions). Clostridium difficile and Candida spp infections (isolated in blood cultures) and nosocomial infections caused by MDRM were also compared between control and intervention periods as an ecological control.
Data were analysed using Stata 12.0. Categorical variables are presented as percentages and were compared using chi-squared test analysis. Monthly antimicrobial consumption and costs were compared using the non-parametric Wilcoxon signed-rank test. A significant result was defined as a p-value <0.05. The research was conducted in accordance with national and institutional standards. The study was approved by the local hospital bioethics committee (Reference number: 2017/0121). According to the local following the committee's guidelines, since data collection and analysis was performed retrospectively, informed consent was not necessary for the study.
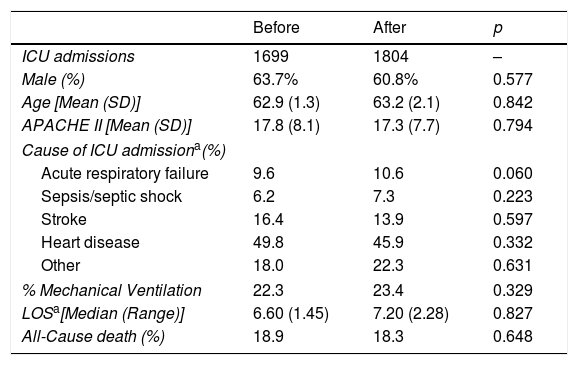
ResultsA total of 1699 and 1804 admissions, respectively, were included in the pre- and post-intervention periods. No significant differences were found in clinical or demographic variables, length of ICU stay or mortality between both periods (Table 1).
Demographic and clinical data of patients before and after antimicrobial stewardship intervention.
| Before | After | p | |
|---|---|---|---|
| ICU admissions | 1699 | 1804 | – |
| Male (%) | 63.7% | 60.8% | 0.577 |
| Age [Mean (SD)] | 62.9 (1.3) | 63.2 (2.1) | 0.842 |
| APACHE II [Mean (SD)] | 17.8 (8.1) | 17.3 (7.7) | 0.794 |
| Cause of ICU admissiona(%) | |||
| Acute respiratory failure | 9.6 | 10.6 | 0.060 |
| Sepsis/septic shock | 6.2 | 7.3 | 0.223 |
| Stroke | 16.4 | 13.9 | 0.597 |
| Heart disease | 49.8 | 45.9 | 0.332 |
| Other | 18.0 | 22.3 | 0.631 |
| % Mechanical Ventilation | 22.3 | 23.4 | 0.329 |
| LOSa[Median (Range)] | 6.60 (1.45) | 7.20 (2.28) | 0.827 |
| All-Cause death (%) | 18.9 | 18.3 | 0.648 |
In one year of AMS implementation, 282 antimicrobial prescriptions in 218 episodes from 182 patients presented one or more criteria that required the revision by the AMS team, being evaluated in 63 AMS meetings. Patients were already receiving antimicrobials before ICU admission in 23.4% of the cases. Antimicrobial prescription revisions were performed according to the following criteria: prescription of a restricted antimicrobial agent (140; 49.6%), inappropriate treatment according to microbiological results (78; 27.6%), inappropriate dose (37; 13.1%), inappropriate duration (35; 13.0%), possible antimicrobial de-escalation according to microbiological results (16; 5.7%), intravenous to oral switch-over therapy (10; 2.9%), and others (14; 4.9%). 18 prescriptions presented more than one review criteria. A total of 38 patients with relevant drug interactions were found and 30 adverse events related to antimicrobial therapy were recorded. Antimicrobials with a higher number of audits were linezolid (51; 18.1%), meropenem (46; 16.3%), daptomycin (24; 8.5%), and piperacillin/tazobactam (18; 6.4%).
From the 218 antimicrobial episodes reviewed, 142 (65.1%) resulted in a proposed amendment that was discussed with the prescribing physician. Recommended amendments by the AMS team were: stop antimicrobial treatment (84; 59.2%), antimicrobial de-escalation (29; 20.4%), change in dose (13; 9.2%), monitoring drug concentrations (7; 4.9%), and start a new antimicrobial (9; 6.3%). Most of these recommendations (91.5%) were accepted by the prescribing physician.
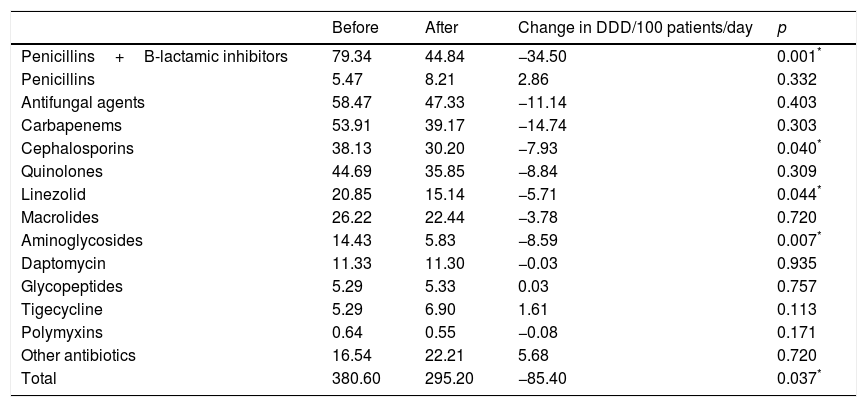
Total antimicrobial DDD/100 patient-days consumption was reduced from 380.6 to 295.2 (−22.4%; p=0.037). Antimicrobials in which the AMS programme had a significant impact were penicillins plus b-lactamase inhibitors, linezolid, cephalosporins, and aminoglycosides (Table 2). A noticeable reduction was also observed in carbapenems, quinolones, and antifungal agents, although no significant differences were obtained. On the other hand, a non-significant increase of reduced-spectrum penicillins was observed. Antimicrobial global spending after one year of AMS programme was reduced by €119.636 compared with the previous control year (€520.186 vs. €400.550; p=0.012).
Changes of antimicrobial consumption (DDD/100 patients/days) before and after antimicrobial stewardship programme.
| Before | After | Change in DDD/100 patients/day | p | |
|---|---|---|---|---|
| Penicillins+B-lactamic inhibitors | 79.34 | 44.84 | −34.50 | 0.001* |
| Penicillins | 5.47 | 8.21 | 2.86 | 0.332 |
| Antifungal agents | 58.47 | 47.33 | −11.14 | 0.403 |
| Carbapenems | 53.91 | 39.17 | −14.74 | 0.303 |
| Cephalosporins | 38.13 | 30.20 | −7.93 | 0.040* |
| Quinolones | 44.69 | 35.85 | −8.84 | 0.309 |
| Linezolid | 20.85 | 15.14 | −5.71 | 0.044* |
| Macrolides | 26.22 | 22.44 | −3.78 | 0.720 |
| Aminoglycosides | 14.43 | 5.83 | −8.59 | 0.007* |
| Daptomycin | 11.33 | 11.30 | −0.03 | 0.935 |
| Glycopeptides | 5.29 | 5.33 | 0.03 | 0.757 |
| Tigecycline | 5.29 | 6.90 | 1.61 | 0.113 |
| Polymyxins | 0.64 | 0.55 | −0.08 | 0.171 |
| Other antibiotics | 16.54 | 22.21 | 5.68 | 0.720 |
| Total | 380.60 | 295.20 | −85.40 | 0.037* |
The time-series analysis showed that antimicrobial stewardship programme period was associated with a reduction in antimicrobial consumption (β=−1.48, p=0.043). On the other hand, no changes in total hospital antimicrobial consumption was observed during the same period (β=−0.22; p=0.822). No significant changes were observed in type of patients admitted to the hospital (Surgical patients: 67.9% vs. 67.4%; p=0.184; Unscheduled admissions: 62.5% vs. 61.9%; p=0.071), length of hospital stay (6.92 vs. 6.88; p=0.062) or hospital mortality (3.42% vs. 3.56%; p=0.245) between both periods. No differences were found in the length of stay or mortality in those patients with sepsis, ventilator-associated pneumonia and catheter-related bacteremia.
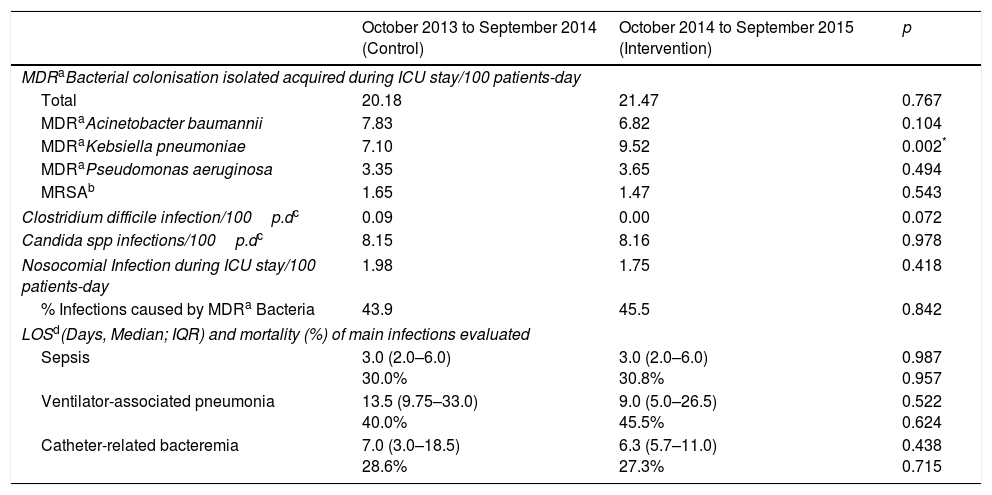
After AMS programme implementation, no changes in C. difficile infection or Candida spp. bloodstream infection rates were observed (Table 3). We observed a reduction in MDR A. baumannii, and MRSA isolated per 100 patient-days, although no statistically significant differences were found, whereas extended spectrum beta-lactamase (ESBL) Klebsiella pneumoniae increased significantly. During the intervention period, there was a two-month ESBL K. pneumoniae outbreak in our ICU, affecting total results. Global MDRM and total nosocomial infections during ICU stay did not change significantly.
Comparison in clinical and microbiologist results before and after antimicrobial stewardship implementation.
| October 2013 to September 2014 (Control) | October 2014 to September 2015 (Intervention) | p | |
|---|---|---|---|
| MDRaBacterial colonisation isolated acquired during ICU stay/100 patients-day | |||
| Total | 20.18 | 21.47 | 0.767 |
| MDRaAcinetobacter baumannii | 7.83 | 6.82 | 0.104 |
| MDRaKebsiella pneumoniae | 7.10 | 9.52 | 0.002* |
| MDRaPseudomonas aeruginosa | 3.35 | 3.65 | 0.494 |
| MRSAb | 1.65 | 1.47 | 0.543 |
| Clostridium difficile infection/100p.dc | 0.09 | 0.00 | 0.072 |
| Candida spp infections/100p.dc | 8.15 | 8.16 | 0.978 |
| Nosocomial Infection during ICU stay/100 patients-day | 1.98 | 1.75 | 0.418 |
| % Infections caused by MDRa Bacteria | 43.9 | 45.5 | 0.842 |
| LOSd(Days, Median; IQR) and mortality (%) of main infections evaluated | |||
| Sepsis | 3.0 (2.0–6.0) 30.0% | 3.0 (2.0–6.0) 30.8% | 0.987 0.957 |
| Ventilator-associated pneumonia | 13.5 (9.75–33.0) 40.0% | 9.0 (5.0–26.5) 45.5% | 0.522 0.624 |
| Catheter-related bacteremia | 7.0 (3.0–18.5) 28.6% | 6.3 (5.7–11.0) 27.3% | 0.438 0.715 |
In this study, we have been able to demonstrate the effectiveness of a prospective audit and feedback AMS programme in the critical care setting, in order to safely reduce antibiotic consumption.
The use of antimicrobial agents is essential in critically ill patients with serious infections. In fact, it is known that antimicrobials are used widely in the ICUs.19 However, it is well-known that antimicrobial overuse can promote the emergence of multidrug-resistant bacteria in these units.20 Recent reports suggest that drug development will not adequately address the problem of antibiotic resistance among common bacterial pathogens.21 Several reasons for the current situation have been proposed, including difficulties in identifying new bacterial targets, or even the possibility that the majority of targets amenable to antibacterial activity have already been identified.22 The lack of promotion of public and private research in discovery and development of new antimicrobial agents also contributes to this situation. These circumstances leave clinicians with limited effective antibiotic options for their patients.21,22
Therefore, different strategies have been evaluated to control antimicrobial use, including formulary restriction, pre-authorisation, antimicrobial cycling, or a combination of these. However, only interventions focused on increasing effective prescription through AMS programmes have been associated with an improvement in clinical outcomes in the hospital general wards.12,23,24 AMS programmes have shown to be an effective tool to control antimicrobial use, improve patient outcomes, and reduce healthcare cost in hospital general wards and in critical care units.25–29
Among different AMS programmes, prospective audit and feedback interventions have several advantages, including a better acceptance by prescribers, and the opportunity to conduct an educational action through the feedback.30,31 However, few experiences of a prospective audit and feedback AMS programme has been reported from the critical care setting. Elliggsen et al. achieved a clear decrease in the consumption of antibiotics, and also a decrease in resistances to the monitored antibiotics, as well as a decrease in C. difficile infections. However, although this is an extremely careful study, they only intervened on specific antibiotics (third-generation cephalosporins, β-lactam and β-lactamase inhibitor combinations, fluoroquinolones, and vancomycin) and there was no intervention on the use of antifungal drugs.32 Currently, at least in our area, AMS programmes should also include the most controversial antimicrobials such as tigecycline, linezolid, daptomycin, or antifungals drugs. Taggart et al. described a one-year experience in a medical and a surgical unit.33 However, in this study antimicrobial reduction was limited to anti-psuedomonal antibiotics. No differences in resistance patterns, length of stay and mortality rate was observed. As the authors concluded, we agree that a longer follow up period is required to appreciate changes in ICU ecology. Other experiences described in critical care units have found reduction in antimicrobial use after AMS implementation.34–36 However, the description of the clinical impact of these interventions, as well as its effect on the infection and colonisation by multiresistant strains is highly variable, making difficult to draw firm conclusions.
In our study, we found that ICU antimicrobial consumption significantly decreased with AMS programme implementation. Antimicrobial consumption dropped from 380.6 to 295.2 DDD/100 patient-days, which was a decrease of 22.4%. Of great significance is the high percentage of acceptance among members of the critical care team, which was even higher than that achieved in the study previously mentioned (82% of acceptance).31 This could be justified by the direct participation of a member of the team and the continuity of the pharmacist involved in the AMS programme. The importance of an intensivist involved in this type of programmes depends not only on the selection and duration of the antibiotic treatment in a critical situation, but also on the knowledge of all those situations that require a dose adjustment in the antimicrobial dosage, as well as those interventions beyond antibiotic management that can prevent infections and improve patients’ prognosis and shorten the duration of their treatment,37 including a proper management of the airway, maintenance and early catheter withdrawal, and so on.
After AMS implementation, the effect on carbapenem, tigecycline and colistin consumption did not reach a statistically significant reduction. These results could be due to the coincidence during the study of an ESBL K. pneumoniae outbreak in our ICU, only susceptible to carbapenems, amikacin, colistin and tigecycline. Therefore, our results could have been affected by this specific epidemiological context. It should be notice that de-escalation has been one of the main interventions recommended by our AMS team. Antimicrobial de-escalation in critically ill patients has been associated not only with a reduction in antibiotic resistance, but also with an improvement in clinical evolution of patients.38 Even some broad-spectrum antimicrobials could not be avoided in critically ill patients and/or ICUs with high prevalence of MDRM, treatment duration and de-escalation could be guided by AMS programmes in order to reduce resistance selection.
After one year of intervention, we did not achieve a global significant decrease of total MDRM colonisation rate. Although AMS interventions have been associated with a reduction in the emergence of MDRM, a longer follow up period may be required to appreciate changes in ICU ecology.39 On the other hand, it should be considered that during the intervention period, there was an ESBL K. pneumonia outbreak in the unit that has had a great impact on the number of patients colonised. Our results show that AMS interventions must be carried out jointly with an increase in measures to prevent environmental bacterial transmission in order to reduce the spread of multiresistant pathogens.
Total antimicrobial consumption in the hospital did not change during the study period. This phenomenon is of great relevance, given that an important part of patients admitted to the ICU come from other hospital units, and antibiotic resistance generated in other wards may end up in the ICU. This idea reinforces the need for a global AMS all over the hospital to ensure the rational use of antibiotics and to prevent antibiotic resistance. Nevertheless, our AMS programme efficacy (in terms of antimicrobial consumption, antimicrobial costs, and decrease in other MDRM), even in the presence of an ESBL K. pneumoniae outbreak, reinforces the utility of this kind of intervention in the critical care setting.
Our study has several limitations. We described an intervention study in a single-centre. Intervention safety was assured by the lack of effect on ICU nosocomial infection rates, length of stay, and mortality. However, long-term effects of AMS programmes should be evaluated in order to assess the real impact of this multidisciplinary intervention on patient mortality and in bacterial resistance. Moreover, the existence of an ESBL K. pneumoniae outbreak during the study period could have affected our results, but in any case this would give more strength to the positive ones. On the other hand, given the lack of information available during the prior to the AMS implementation, we have not been able to evaluate two of the indicators recommended to assess the appropriateness of antimicrobial treatment15: Days of treatment (DOT) and percentage of appropriate empirical treatment. However, it is important to highlight the difficulties in evaluating this last indicator in the critically ill patient, given the high number of patients with negative microbiological cultures, as well as the possible confusion generated by colonisation samples. Moreover, despite the fact that DDDs have the limitation of being dependent on the dose of antimicrobial prescribed and therefore its value could be modified in the most severe patients, since the characteristics of the patients have not varied significantly between both periods, we consider that the comparison of DDDs value between both periods reflects the real decrease in antimicrobial consumption after AMS implementation.
Antibiotic resistance has become a major clinical and public health problem. Our results further support the implementation of an AMS programme in critical care units, and reinforces the efficacy of an audit and feedback design, lead by an intensivist expert in the management of infections in the critically ill patients. These interventions require continuous tracking, monitoring and assessment. Antimicrobial consumption and outcome indicators should be used to provide feedback to prescribers, and even to the hospital management team.
In conclusion, the implementation of antimicrobial stewardship strategies in an audit and feedback design significantly reduced antimicrobial use in a relatively short period. Long-term effects of these programmes over the local microbial resistance and patient outcomes should be evaluated in future studies.
FundingNo funding has been received for this article.
Authors’ contributionsJR participated in the design of the study, data collection and performed the statistical analysis. PR was responsible for the coordination of the study, participated in its design and helped to draft the manuscript. MG participated in data collection. EV participated in the design of the study and statistical analysis. JF participated in the collection and analysis of microbiological data. JLP, MSL, and AC participated in its design, coordination and review of the manuscript. All authors read and approved the final manuscript.
Conflict of interestsNo conflicts of interest have been declared.
We appreciate the cooperation and commitment of all staff of the ICU to the project.