To evaluate the efficacy of respiratory muscular training in the weaning of mechanical ventilation and respiratory muscle strength in patients on mechanical ventilation of 48h or more.
DesignRandomized controlled trial of parallel groups, double-blind. Ambit: Intensive Care Unit of a IV level clinic in the city of Cali.
Patients126 patients in mechanical ventilation for 48h or more.
InterventionsThe experimental group received daily a respiratory muscle training program with threshold, adjusted to 50% of maximal inspiratory pressure, additional to standard care, conventional received standard care of respiratory physiotherapy.
Main interest variablesWeaning of mechanical ventilation. Other variables evaluated: respiratory muscle strength, requirement of non-invasive mechanical ventilation and frequency of reintubation.
AnalysisIntention-to-treat analysis was performed with all variables evaluated and analysis stratified by sepsis condition.
ResultsThere were no statistically significant differences in the median weaning time of the MV between the groups or in the probability of extubation between groups (HR: 0.82; 95%CI: 0.55–1.20; p=0.29). The maximum inspiratory pressure was increased in the experimental group on average 9.43 (17.48)cm H2O and in the conventional 5.92 (11.90)cm H2O (p=0.48). The difference between the means of change in maximal inspiratory pressure was 0.46 (p=0.83; 95%CI −3.85 to −4.78).
ConclusionsRespiratory muscle training did not demonstrate efficacy in the reduction of the weaning period of mechanical ventilation nor in the increase of respiratory muscle strength in the study population. Registered study at ClinicalTrials.gov (NCT02469064).
Evaluar la eficacia del entrenamiento muscular respiratorio en el destete de la ventilación mecánica y en la fuerza muscular respiratoria en pacientes en ventilación mecánica.
DiseñoEnsayo clínico controlado aleatorizado de grupos paralelos, doble ciego. Ámbito: unidad de cuidados intensivos de una clínica de iv nivel de la ciudad de Cali.
PacientesCiento veintiséis pacientes en ventilación mecánica por 48horas o más.
IntervencionesEl grupo experimental recibió todos los días un programa de entrenamiento muscular respiratorio con treshold, ajustado al 50% de la presión inspiratoria máxima adicional al cuidado estándar; el convencional recibió el cuidado estándar de fisioterapia respiratoria.
Variables de interés principalDestete de la ventilación mecánica. Otras variables evaluadas: fuerza muscular respiratoria, requerimiento de ventilación mecánica no invasiva y la frecuencia de reintubación.
AnálisisAnálisis por intención a tratar con todas las variables evaluadas y estratificado por condición de sepsis.
ResultadosNo se presentaron diferencias estadísticamente significativas en la mediana de tiempo de destete de la VM entre los grupos ni en la probabilidad de extubación entre los grupos (HR: 0,82; IC 95%: 0,55-1,20; p=0,29). La presión inspiratoria máxima se incrementó en el grupo experimental en promedio 9,43 (17.48) cm H2O y en el convencional 5,92 (11,90) cm H2O (p=0,48). La diferencia entre los promedios del cambio en la presión inspiratoria máxima fue 0,46 (p=0,83 IC 95%: −3,85 a −4,78).
ConclusionesEl entrenamiento muscular respiratorio no demostró eficacia en la disminución del periodo del destete de la VM ni en el incremento de la fuerza muscular respiratoria en la población estudiada. Estudio registrado en ClinicalTrials.gov (NCT02469064).
Approximately 35% of all patients admitted to the Intensive Care Unit (ICU) require mechanical ventilation (MV). Of these, a minority (20–30%) present weaning difficulties and almost 50% of these individuals require MV for 7 or more days. Thus, approximately 15% of all patients admitted to the ICU require prolonged MV.1–3
Prolonged MV implies an increased risk of ventilator-associated pneumonia (VAP), tracheal ischemia, lung injury and diaphragmatic muscle dysfunction. Such complications in turn increase the mortality risk.1,4–12 Furthermore, patients with weaning difficulties consume approximately 40% of the resources in the ICU setting.2,7,13
Sepsis, which is observed in about 10.8% of all patients admitted to the ICU,14 has been regarded as a conditioning factor in weaning from MV and in respiratory muscle strength.15–18 Bolton et al. were the first to suggest this,18 and confirmation was provided by subsequent retrospective and prospective studies.15,17
The different management strategies used to improve respiratory muscle strength in patients subjected to MV and to facilitate weaning include respiratory muscle training (RMT). This technique seeks to improve respiratory muscle strength and resistance. However, few studies have evaluated RMT in patients subjected to MV as a strategy for facilitating weaning.19–25
At the time of the present study, four clinical trials had evaluated the efficacy of RMT in terms of weaning time in patients subjected to MV.22,26–28 Only two of them reported significant differences in weaning time, with the conclusion the RMT shortens this time,28,29 while the other two trials failed to confirm these observations. The mentioned trials differed in terms of the methodology used, the RMT technique applied, and the patient populations studied. Furthermore, the sample sizes were small, and no multivariate analyses were carried out.
Since no studies have been made on the effects of RMT in patients subjected to MV in Colombia, and the international evidence is contradictory and nonspecific, the present study was designed to assess the impact of RMT referred to weaning from MV and inspiratory muscle strength in patients subjected to MV for 48h or more.
MethodologyA randomized, controlled, double-blind parallel-group clinical trial was carried out. Inclusion criteria: patients aged 18 years or older, requiring MV for 48h or more, and admitted to a fourth level ICU pertaining to a healthcare institution in the city of Cali (Colombia). Written informed consent to participation in the study was required. The eligible patients presented with a first event requiring MV, were intubated in the mentioned institution or in peripheral centers, with reference to the institution in the 12h following intubation. The patients presented PaO2≥60mmHg, with FiO2≤0.5; PEEP≤8cm H2O; a RASS score of between −1 and 0, and a mean blood pressure of over 60mmHg in the absence of vasopressive drug support, or with only minimum vasopressor requirements (dobutamine or dopamine<5μg/kg/min or epinephrine≤1μg/kg/min). Exclusion criteria: patients with progressive neuromuscular disease, central nervous system damage, spinal cord injuries above level T5, thoracic or spinal skeletal disease, home ventilatory support administered prior to hospital admission, presence of excessive secretions, pregnant women and patients infected with multiresistant organisms.
The investigation was approved by the Ethics Committees of Universidad del Valle (Ref. 022-013) and of the healthcare institution where the study was carried out (Ref. 030-2014).
The control group received conventional respiratory management in the ICU, involving respiratory physiotherapy, physical therapy and MV management. As additional treatment, the experimental group was included in an RMT program with the Threshold IMT respiratory muscle trainer (Respironics Inc., Murrysville, PA, USA) every day, twice a day. Three series of 6–10 repetitions each were carried out, with two minutes of rest between series. The initial training load was adjusted considering 50% of the maximum inspiratory pressure (Pimax),29,30 and was applied by four previously trained physiotherapists with full dedication to the project. The physiotherapists followed the corresponding standard operating procedure developed by the investigators.
Respiratory muscle strength was measured from Pimax according to the protocols of the American Thoracic Society and the European Respiratory Society. A Carefusion digital manuvacuometer31was used to perform intra- and inter-operator reliability evaluations prior to the data collection process. The Pimax measurements were made in both groups.
Weaning from MV was defined as the time from the start of MV in pressure support mode (PS), with PS≤10cm H2O, to extubation. The respiratory muscle strength was evaluated from the change in Pimax. Weaning failure was defined as the need for reintubation or patient death within the first 48h postextubation.
Primary outcome: weaning from MV. Secondary outcomes: respiratory muscle strength, frequency of weaning failure, and need for noninvasive MV (NIMV) in each treatment group.
For the calculation of sample size, we defined weaning time as the response variable, with a standard deviation (SD) of 52.8h and an expected difference in weaning time of 24h in favor of the group experimental, according to the data of the controlled clinical trial carried out by Condessa et al.26 Assuming a dropout rate of 10%, a statistical power of 80% and an alpha error of 5%, the required sample size was found to be 63 patients in each group.
Stratified randomization was carried out according to condition of sepsis, defined as the presence of one or more of the following: fever (temperature>38.3°C), hypothermia (temperature<36°C), tachycardia (heart rate>90bpm), tachypnea (respiratory frequency>40rpm), leukocytosis (leukocyte count>12,000cells/μl), leukopenia (leukocyte count<4000cells/μl), and altered mental state in patients with signs suggestive of infection.32 Posteriorly, non-fixed block randomization was performed, with variable magnitudes of 4 and 6 individuals. Randomization was carried out with the Random allocation software for parallel group randomized trials, prior to entry of the first patient. Two physiotherapists performed treatment allocation. The combinations of the intervention were organized by a person external to the study in sealed envelopes of different colors according to the condition of sepsis. The envelopes were numbered on the outside and were kept in two sealed boxes that were opened once informed consent was obtained.
Masking of both the principal investigator and the ICU working team was carried out. To this effect, the data collection process and implementation of the intervention were conducted by the four physiotherapists that carried out part of the field work, excluding the principal investigator. Measurement of Pimax was made and implementation of the intervention was carried out in a previously defined time period. Accordingly, the intervention in the two groups had the same duration. Furthermore, the intervention was carried out with the curtains of the ICU boxes drawn, keeping the Threshold IMT all the time stored in a sealed opaque box.
Statistical analysisAn intention-to-treat (ITT) analysis was performed and final multiple models were generated for each of the study variables.
Selection of the variables of the final models was carried out by taking those with p<0.25 from each simple model.33 The stepwise variable elimination method was used for this purpose. Each time a variable was removed, the model was subjected to the Irtest. The presence of confusion and interaction was assessed with the variables that remained in the model.33
The univariate analysis of the hours of weaning from MV was based on the Mann–Whitney U-test. The univariate analysis of the probability of extubation was based on survival analysis (Kaplan–Meier) and the log-rank test. The multivariate analysis in turn was based on Cox regression.33 We evaluated the fit of the final model obtaining the Cox–Snell residuals with the corresponding plot. The difference in mean final Pimax between the groups was analyzed by means of the Student t-test, and the difference in mean Pimax within each group was analyzed using the paired t-test. To this effect, the change in Pimax within each group was regarded as the difference between mean final Pimax and initial Pimax. The multivariate analysis of the change in Pimax (initial Pimax−final Pimax) was based on linear regression analysis. We determined the odds ratio (OR) of weaning failure and of the need for NIMV. The multivariate analysis of these two variables was based on logistic regression analysis. The Mann–Whitney U-test was used to compare the time of NIMV requirement between the groups.
ResultsThe results referred to the global sample are detailed below.
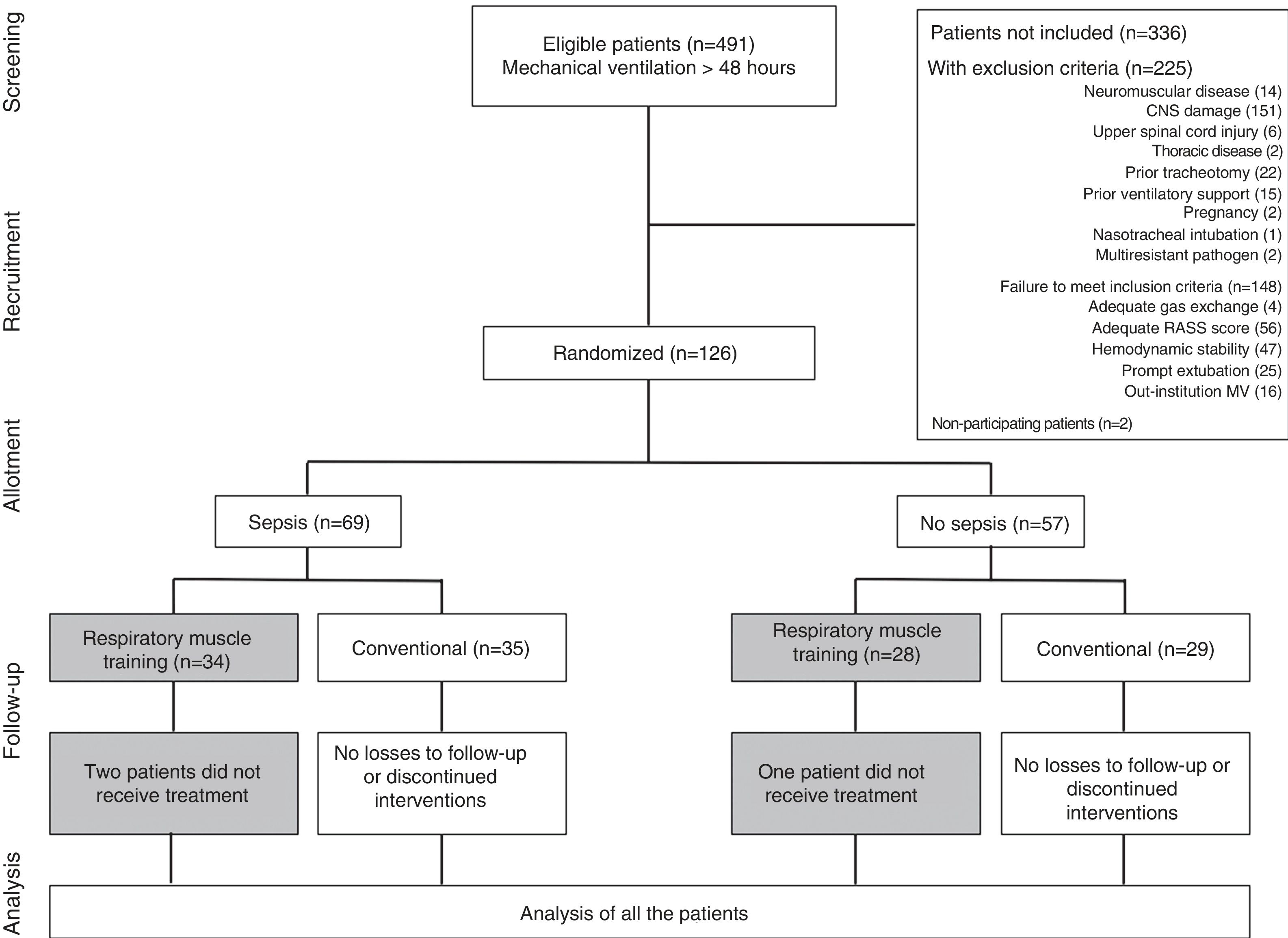
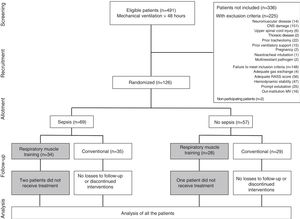
Characteristics of the patients included in the studyFig. 1 shows patient recruitment, randomization and flow according to group. Over 7 months a total of 491 patients required MV for a period of 48h or more; of these, 215 presented exclusion criteria, 148 failed to meet inclusion criteria, and two patients decided not to participate in the study.
A total of 126 patients were randomized. Of these, three did not comply with the protocol because they were assigned to the experimental treatment but received conventional care. None of the patients withdrew from the study, and there were no adverse effects.
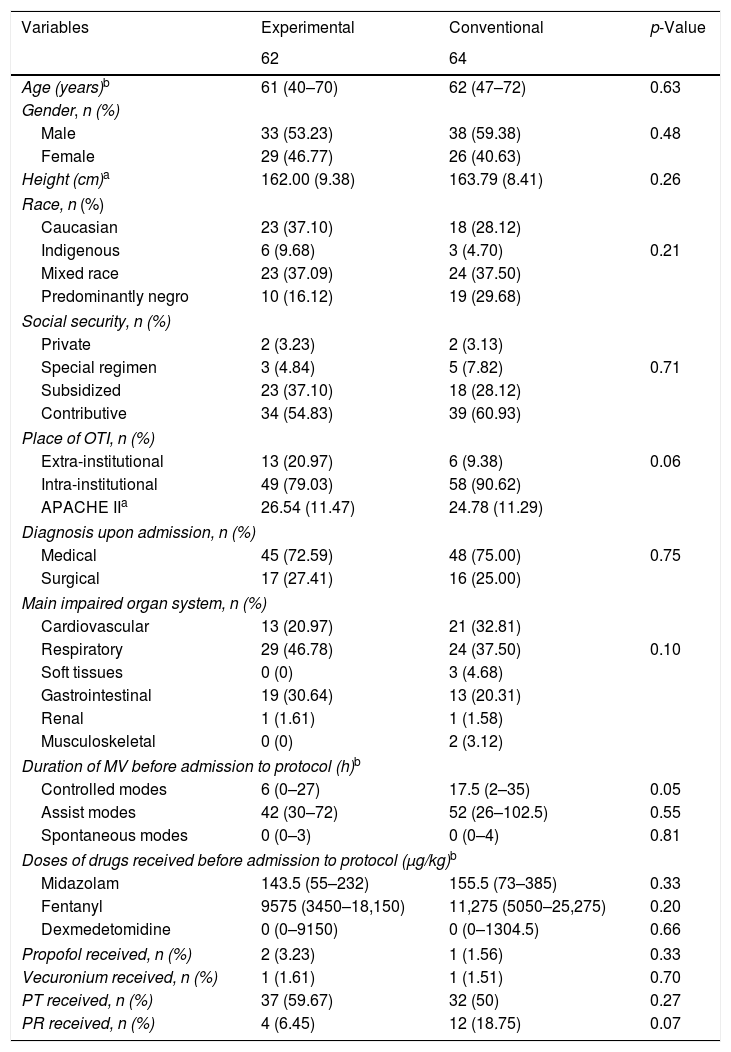
The characteristics of the patients at baseline according to the assigned treatment are reported in Table 1.
Patient characteristics upon admission according to the assigned treatment.
| Variables | Experimental | Conventional | p-Value |
|---|---|---|---|
| 62 | 64 | ||
| Age (years)b | 61 (40–70) | 62 (47–72) | 0.63 |
| Gender, n (%) | |||
| Male | 33 (53.23) | 38 (59.38) | 0.48 |
| Female | 29 (46.77) | 26 (40.63) | |
| Height (cm)a | 162.00 (9.38) | 163.79 (8.41) | 0.26 |
| Race, n (%) | |||
| Caucasian | 23 (37.10) | 18 (28.12) | |
| Indigenous | 6 (9.68) | 3 (4.70) | 0.21 |
| Mixed race | 23 (37.09) | 24 (37.50) | |
| Predominantly negro | 10 (16.12) | 19 (29.68) | |
| Social security, n (%) | |||
| Private | 2 (3.23) | 2 (3.13) | |
| Special regimen | 3 (4.84) | 5 (7.82) | 0.71 |
| Subsidized | 23 (37.10) | 18 (28.12) | |
| Contributive | 34 (54.83) | 39 (60.93) | |
| Place of OTI, n (%) | |||
| Extra-institutional | 13 (20.97) | 6 (9.38) | 0.06 |
| Intra-institutional | 49 (79.03) | 58 (90.62) | |
| APACHE IIa | 26.54 (11.47) | 24.78 (11.29) | |
| Diagnosis upon admission, n (%) | |||
| Medical | 45 (72.59) | 48 (75.00) | 0.75 |
| Surgical | 17 (27.41) | 16 (25.00) | |
| Main impaired organ system, n (%) | |||
| Cardiovascular | 13 (20.97) | 21 (32.81) | |
| Respiratory | 29 (46.78) | 24 (37.50) | 0.10 |
| Soft tissues | 0 (0) | 3 (4.68) | |
| Gastrointestinal | 19 (30.64) | 13 (20.31) | |
| Renal | 1 (1.61) | 1 (1.58) | |
| Musculoskeletal | 0 (0) | 2 (3.12) | |
| Duration of MV before admission to protocol (h)b | |||
| Controlled modes | 6 (0–27) | 17.5 (2–35) | 0.05 |
| Assist modes | 42 (30–72) | 52 (26–102.5) | 0.55 |
| Spontaneous modes | 0 (0–3) | 0 (0–4) | 0.81 |
| Doses of drugs received before admission to protocol (μg/kg)b | |||
| Midazolam | 143.5 (55–232) | 155.5 (73–385) | 0.33 |
| Fentanyl | 9575 (3450–18,150) | 11,275 (5050–25,275) | 0.20 |
| Dexmedetomidine | 0 (0–9150) | 0 (0–1304.5) | 0.66 |
| Propofol received, n (%) | 2 (3.23) | 1 (1.56) | 0.33 |
| Vecuronium received, n (%) | 1 (1.61) | 1 (1.51) | 0.70 |
| PT received, n (%) | 37 (59.67) | 32 (50) | 0.27 |
| PR received, n (%) | 4 (6.45) | 12 (18.75) | 0.07 |
OTI: orotracheal intubation; PR: pulmonary rehabilitation; PT: physical therapy; MV: mechanical ventilation.
The mean age of the patients was 57.49 years (median 62, range: 18–88), and the mean height was 162.90cm (median 163, range: 145–185). There were 71 males and 55 females. A total of 107 patients were intubated within the healthcare institution and 19 external to the institution. Of the patients admitted to the ICU, 93 were medical patients while 33 corresponded to surgical cases. The mean MV time prior to admission to the protocol was 86.53h (median 66.5, range: 49–314); the mean midazolam dose administered prior to admission to the protocol was 277.73μg/kg (median 153, range: 0–5200); the mean fentanyl dose received was 16,113μg/kg (median 10,075, range: 0–153,800); three and two patients respectively received propofol and vecuronium prior to admission to the protocol; 69 patients underwent physical therapy; and 16 received pulmonary rehabilitation during follow-up.
There were no differences in the clinical and sociodemographic characteristics between the two patients that decided not to participate, and the patients admitted to the study.
Weaning from mechanical ventilationOne patient in the experimental group evolved with septic shock and could not be weaned from MV. During follow-up, three patients required tracheostomy without weaning from MV having been started (two belonging to the experimental group and one to the conventional group).
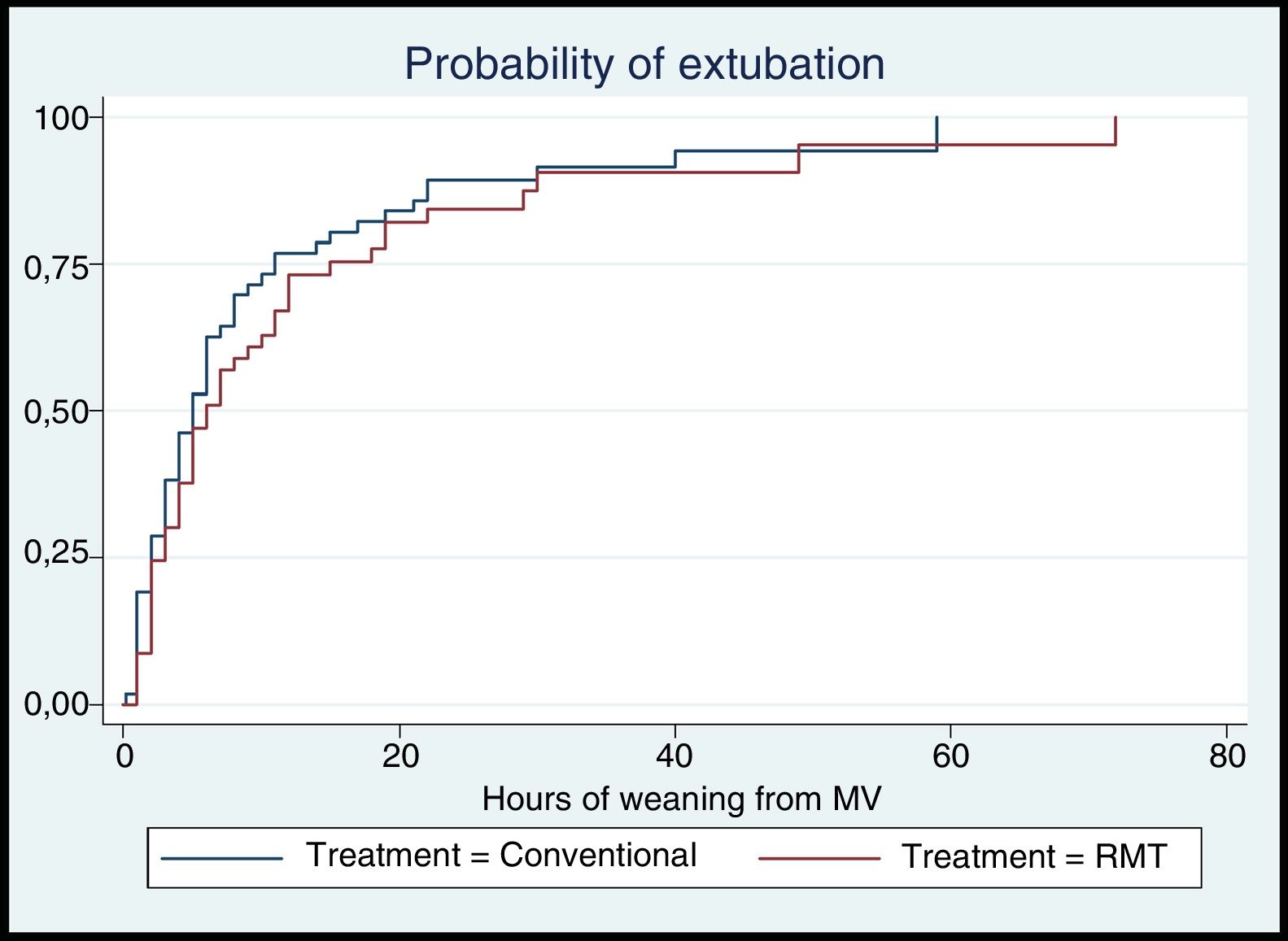
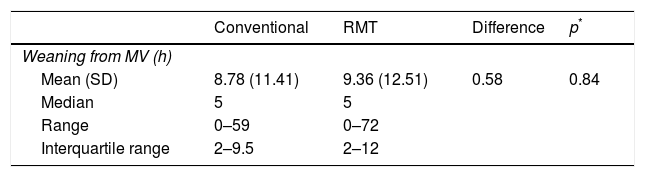
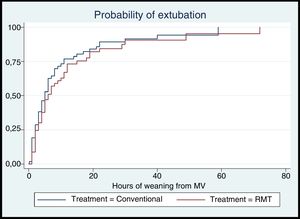
The mean time to weaning from MV in the conventional group was 8.78 (11.41)h, versus 9.36 (12.51)h in the conventional group. In both groups the median time to weaning from MV was 5h; in the conventional group the interquartile range (IQR) was 2–9.5h, versus 2–12h in the experimental group. There were no statistically significant differences in the median weaning time between the groups (Table 2). Likewise, there were no statistically significant differences in the probability of extubation over follow-up according to the treatment provided (hazard ratio [HR]: 0.82; log-rank test=1.12, p=0.29) (Fig. 2).
Mechanical ventilation weaning time according to the treatment administered.
| Conventional | RMT | Difference | p* | |
|---|---|---|---|---|
| Weaning from MV (h) | ||||
| Mean (SD) | 8.78 (11.41) | 9.36 (12.51) | 0.58 | 0.84 |
| Median | 5 | 5 | ||
| Range | 0–59 | 0–72 | ||
| Interquartile range | 2–9.5 | 2–12 | ||
RMT: respiratory muscle training; MV: mechanical ventilation.
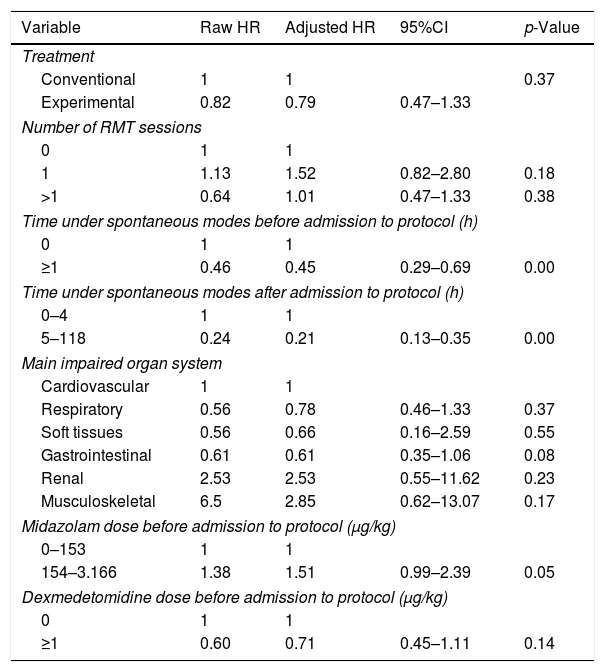
Table 3 shows the raw and adjusted hazard ratios referred to extubation.
Raw and adjusted hazard ratios referred to extubation.
| Variable | Raw HR | Adjusted HR | 95%CI | p-Value |
|---|---|---|---|---|
| Treatment | ||||
| Conventional | 1 | 1 | 0.37 | |
| Experimental | 0.82 | 0.79 | 0.47–1.33 | |
| Number of RMT sessions | ||||
| 0 | 1 | 1 | ||
| 1 | 1.13 | 1.52 | 0.82–2.80 | 0.18 |
| >1 | 0.64 | 1.01 | 0.47–1.33 | 0.38 |
| Time under spontaneous modes before admission to protocol (h) | ||||
| 0 | 1 | 1 | ||
| ≥1 | 0.46 | 0.45 | 0.29–0.69 | 0.00 |
| Time under spontaneous modes after admission to protocol (h) | ||||
| 0–4 | 1 | 1 | ||
| 5–118 | 0.24 | 0.21 | 0.13–0.35 | 0.00 |
| Main impaired organ system | ||||
| Cardiovascular | 1 | 1 | ||
| Respiratory | 0.56 | 0.78 | 0.46–1.33 | 0.37 |
| Soft tissues | 0.56 | 0.66 | 0.16–2.59 | 0.55 |
| Gastrointestinal | 0.61 | 0.61 | 0.35–1.06 | 0.08 |
| Renal | 2.53 | 2.53 | 0.55–11.62 | 0.23 |
| Musculoskeletal | 6.5 | 2.85 | 0.62–13.07 | 0.17 |
| Midazolam dose before admission to protocol (μg/kg) | ||||
| 0–153 | 1 | 1 | ||
| 154–3.166 | 1.38 | 1.51 | 0.99–2.39 | 0.05 |
| Dexmedetomidine dose before admission to protocol (μg/kg) | ||||
| 0 | 1 | 1 | ||
| ≥1 | 0.60 | 0.71 | 0.45–1.11 | 0.14 |
RMT: respiratory muscle training.
The univariate and multivariate analyses showed a prolongation of weaning time to be associated to the duration of MV in spontaneous modes before (≥1h) and after (5–118h) admission to the protocol.
Change in maximum inspiratory pressureTwenty-four patients failed to yield a measurement of final Pimax (14 belonging to the experimental group and 10 to the conventional group). The final Pimax was thus analyzed in 102 patients.
Table 4 describes the change in Pimax within each group and the difference in the change in Pimax between the groups. There were statistically significant differences between final Pimax and initial Pimax within each group (p<0.05): in the experimental group, Pimax increased 9.43cm H2O, while in the conventional group it increased 5.92cm H2O. There were no statistically significant differences in the mean change in Pimax between the groups (p=0.48).
Change in maximum inspiratory pressure according to the assigned treatment.
| Parameter | Change in Pimax within each group | Difference between groups | ||||
|---|---|---|---|---|---|---|
| Experimental N=48 | p | Conventional N=54 | p | N=48 | p | |
| Mean (SD) | 9.43 (17.48) | 0.00a | 5.92 (11.90) | 0.001a | 3.51 (14.78) | 0.48b |
Pimax: maximum inspiratory pressure.
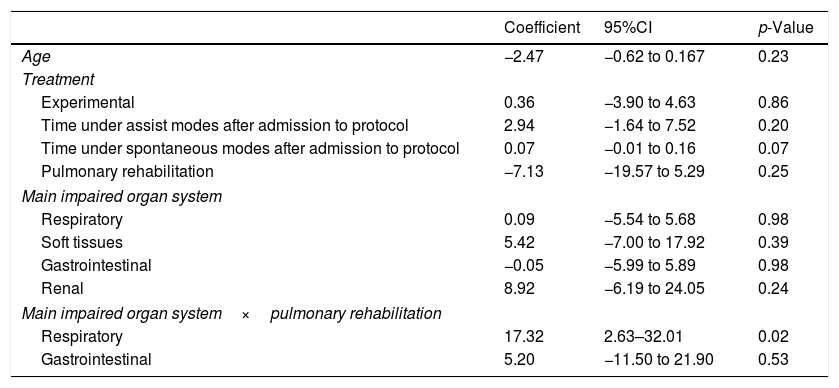
We found no association between the mean change in Pimax and the assigned treatment. The final model showed a significant increase in Pimax to be associated to the interaction between the main compromised system and pulmonary rehabilitation (p<0.05) (Table 5).
Final multiple model of the change in maximum inspiratory pressure.
| Coefficient | 95%CI | p-Value | |
|---|---|---|---|
| Age | −2.47 | −0.62 to 0.167 | 0.23 |
| Treatment | |||
| Experimental | 0.36 | −3.90 to 4.63 | 0.86 |
| Time under assist modes after admission to protocol | 2.94 | −1.64 to 7.52 | 0.20 |
| Time under spontaneous modes after admission to protocol | 0.07 | −0.01 to 0.16 | 0.07 |
| Pulmonary rehabilitation | −7.13 | −19.57 to 5.29 | 0.25 |
| Main impaired organ system | |||
| Respiratory | 0.09 | −5.54 to 5.68 | 0.98 |
| Soft tissues | 5.42 | −7.00 to 17.92 | 0.39 |
| Gastrointestinal | −0.05 | −5.99 to 5.89 | 0.98 |
| Renal | 8.92 | −6.19 to 24.05 | 0.24 |
| Main impaired organ system×pulmonary rehabilitation | |||
| Respiratory | 17.32 | 2.63–32.01 | 0.02 |
| Gastrointestinal | 5.20 | −11.50 to 21.90 | 0.53 |
In the experimental group, 15 patients suffered weaning failure (24.19%), versus 16 in the conventional group (25%) – the difference being nonsignificant (p=0.54).
The univariate and multivariate analyses showed weaning failure to be associated to the APACHE score (0–24) and to the duration of MV in spontaneous modes (≥1) after admission to the protocol (p<0.05). No association was observed between weaning failure risk (OR) and the assigned treatment.
Need for noninvasive mechanical ventilationIn the experimental group, 33 patients underwent NIMV (64.71%), with a median duration of 5 (4–13)h, while in the conventional group, 38 patients underwent NIMV (64.41%), with a median duration of 7.5 (4–11)h – the differences being nonsignificant (p>0.05).
The univariate and multivariate analyses revealed no association between the need (OR) for NIMV and the assigned treatment.
DiscussionThe present study is the first controlled clinical trial at national level in Colombia to investigate the impact of RMT in patients subjected to MV for 48h or more upon weaning from MV and respiratory muscle strength, with masking of the investigators and clinical staff, and with an estimation of sample size based on weaning time – as this is considered the most useful indicator for the patients and the clinical staff. The results revealed no significant differences in weaning time between the groups. The survival analysis showed the probability of extubation to independent of the treatment administered.
The patients in the experimental group (n=62) underwent 1–14 RMT sessions, and 50% underwent 3–14 sessions, due to the short MV period of the patients once compliance with the inclusion criteria was confirmed. Some studies have reported that at least 10 days of muscle training are needed to increase muscle strength of the extremities to the point of guaranteeing a relevant clinical effect.34 The behavior of the respiratory muscles is similar: it has been described that about 14 days of RMT are needed to obtain evidence of significant changes in muscle strength, such as proliferation of the type I and II fibers of the intercostal muscles, and increased inspiratory muscle perfusion.30,35 In this regard, the patients probably did not receive the required number of RMT sessions to produce clinically significant changes.
An association was found between prolonged weaning from MV and the duration of MV in spontaneous modes before and after admission to the protocol. Mechanical ventilation in spontaneous modes has been suggested as a therapeutic strategy for preventing respiratory muscle dysfunction and thus contributing to shorten MV time.36–38 However, only partial modes (assist/spontaneous) with low assist levels prevent respiratory muscle dysfunction and thus help accelerate weaning from MV. In contrast, high assist levels in partial modes (assist/spontaneous) are deleterious in controlled MV and cause diaphragmatic muscle dysfunction, increased ventilatory asynchronicity, and a prolongation of weaning time.38–40 In this study we did not quantify the duration of MV in pressure support with specific pressure levels. However, given the mean duration of MV in pressure support (11.1±18h in the conventional group and 17.26±34.92h in the experimental group) and the mean MV weaning time (8.78±11.41h in the conventional group and 9.36±12.51h in the experimental group), it is assumed that the patients received MV with pressure support levels above 10cm H2O, thereby explaining the results obtained.
Four clinical trials have evaluated the efficacy of RMT in terms of time to weaning from MV.22,26–28 To date, only two of them have reported statistically significant differences in weaning time in favor of the experimental group.27,28 The conclusions of these studies are limited by the small sample sizes involved, and by the absence of masking of the group in charge of patient intervention, of a precise definition of weaning time, and of multivariate analyses.
With regard to the change in Pimax, the results revealed no significant differences in the median change in Pimax between the groups. In the final linear regression model of change in Pimax, the interaction variable impaired respiratory system and pulmonary rehabilitation showed statistical significance (interaction p=0.01; 95% confidence interval [95%CI]: 4.25–34.4). This may be related to specific interventions of pulmonary rehabilitation capable of influencing changes in respiratory muscle strength, and which are preferentially found in patients with respiratory disease, where the indication of pulmonary rehabilitation is more apparent.36
Four controlled clinical trials4 have reported results referred to Pimax20,22,26–28. Three of them have described significant differences in the change in Pimax between groups, in favor of the experimental group.20,26,28 The differences in results between studies may be due to differences in the RMT protocols used and to differences in the characteristics of the study population.
The RMT protocol used by Caruso et al. was based on ventilator sensitivity adjustments.22 The other studies used the Threshold IMT device, but with different loading levels.20,26–28 The present investigation used a loading level equivalent to 50% of the Pimax, which in the same way as in the study of Condessa et al. was evaluated by means of daily Pimax measurements. This loading level is the highest reported to date in controlled trials.19 However, no significant differences were found in the change in Pimax between the groups – a fact that may be related to the frequency of the RMT sessions received by the patients in the experimental group, which was not enough to generate significant clinical changes.
The failed weaning odds ratio was not associated to the treatment provided (OR: 0.96; p=0.95; 95%CI: 0.36–2.56). The failure of weaning from MV is a multifactorial event involving conditions such as respiratory and cardiovascular integrity, neuromuscular competence, and the patient psychological conditions and nutritional status, among other factors.41 None of these conditions were evaluated in the present investigation, due to the measurement difficulties found in the ICU. In the experimental group, 15 patients suffered weaning failure (24.19%), versus 16 in the conventional group (25%) (p=0.54). The reported incidence of weaning failure ranges from 26 to 42%,41–46 which is consistent with our own findings.
The need for NIMV was not associated to the treatment administered. There were no statistically significant differences in the median requirement of NIMV between the groups. Esteban et al. reported that 10% of the patients that are extubated require management in the form of post-extubation NIMV.4 In this respect, the proportion of patients requiring NIMV in the present study was greater than reported in the literature – a fact that may be related to management protocols at institutional level.
The methodological strengths of the study are related to the strategies used to reduce the risk of selection and information bias, and to control confounding factors, such as double masking, quality control of the measurement instruments, and the data collection processes and information analysis. The external validity is high, since all the patients that met the inclusion criteria were randomized and included in the study, and the evaluated intervention moreover was conducted in the in-hospital setting. Adherence to treatment was guaranteed in most of the patients; accordingly, 123 patients completed follow-up to the defined primary endpoint. The characteristics of the patients that decided not to participate in the study did not differ from those who were included in the trial, thereby evidencing the absence of selection bias.
The present study comprises the largest sample size reported to date (n=126), and is the only trial to have conducted survival analyses based on Cox regression – thereby ensuring inclusion in the analysis of the censored patients and identification of the variables influencing the primary outcome.
The main limitation of the study was the short RMT period. This was related to the characteristics of the eligible population and to the institutional ventilatory management protocols, since once the patients were seen to comply with the inclusion criteria, the MV weaning process was started and the patients were consequently extubated early – thereby suggesting the absence of respiratory muscle dysfunction associated to MV and non-dependency upon MV. Thus, it was assumed that the patients probably deriving benefit were those in which respiratory muscle dysfunction proved evident.
This investigation constitutes a reference at both national and local level. Since the results do not evidence efficacy of RMT in terms of weaning from MV or respiratory muscle strength, it is not possible to recommend the intervention in this population.
The results reflect the negative relationship between the duration of MV in spontaneous modes with high ventilatory assist levels and weaning from MV – thus suggesting the incursion of spontaneous ventilatory modalities with pressure support levels of under 10cm H2O.
Despite the lack of statistically significant findings, from the clinical perspective it is plausible to assume that RMT might offer benefit as a strategy for preventing respiratory muscle dysfunction associated to MV in patients with disease of respiratory origin who simultaneously undergo pulmonary rehabilitation. In this regard, the patients in our study were seen to increase their respiratory muscle strength.
It is advisable to conduct further studies including patients with difficult weaning and prolonged MV times, in which respiratory muscle dysfunction proves evident, and to evaluate the outcomes of RMT over longer periods of follow-up. Since this population represents a minority of the patients requiring MV in the ICU of the institution involved in the present study, multicenter studies would be advisable to assess the mentioned outcomes.
ConclusionsEvaluation of the impact of RMT revealed no statistically significant differences in MV weaning time or changes in respiratory muscle strength between the experimental group and control group.
A longer duration of MV in spontaneous modes with high ventilatory assist levels prolongs weaning from MV in patients requiring MV for more than 48h. In this sense it is necessary to consider opportune interventions in patients in which the respiratory system is the main impaired organ system, seeking to increase respiratory muscle strength.
There were no statistically significant differences in weaning failure risk or in the need for NIMV between the experimental group and control group.
Financial supportThis study was financed by the Vice-Rectorate for Research of Universidad del Valle.
AuthorshipStudy concept/idea/design: L.M. Sandoval, I.C. Casas, E.C. Wilches, A.F. Garcia; Drafting of the article: L.M. Sandoval, I.C. Casas, E.C. Wilches, A. F. Garcia; Coordination of data compilation and analysis: L.M. Sandoval, I.C. Casas; Project management: L.M. Sandoval, E.C. Wilches, A.F. Garcia; Procurement of funding: L.M. Sandoval, E.C. Wilches, A.F. Garcia; Manuscript review before submission: M. Sandoval, I.C. Casas, E.C. Wilches, A.F. Garcia.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Thanks are due to the Fundación Valle del Lili for granting conduction of the study in its facilities.
Please cite this article as: Sandoval Moreno LM, Casas Quiroga IC, Wilches Luna EC, García AF. Eficacia del entrenamiento muscular respiratorio en el destete de la ventilación mecánica en pacientes con ventilación mecánica por 48 o más horas: un ensayo clínico controlado. Med Intensiva. 2019;43:79–89.