Infections caused by Candida species are common in critically ill patients and contribute to significant morbidity and mortality. The EPICO Project (Epico 1 and Epico 2.0 studies) recently used a Delphi approach to elaborate guidelines for the diagnosis and treatment of this condition in critically ill adult patients. We aimed to evaluate the impact of a multifaceted educational intervention based on the Epico 1 and Epico 2.0 recommendations.
DesignSpecialists anonymously responded to two online surveys before and after a multifaceted educational intervention consisting of 60-min educational sessions, the distribution of slide kits and pocket guides with the recommendations, and an interactive virtual case presented at a teleconference and available for online consultation.
SettingA total of 74 Spanish hospitals.
ParticipantsSpecialists of the Intensive Care Units in the participating hospitals.
Variables of interestSpecialist knowledge and reported practices evaluated using a survey. The McNemar test was used to compare the responses in the pre- and post-intervention surveys.
ResultsA total of 255 and 248 specialists completed both surveys, in both periods, respectively. The pre-intervention surveys showed many specialists to be unaware of the best approach for managing invasive candidiasis. After both educational interventions, specialist knowledge and reported practices were found to be more in line with nearly all the recommendations of the Epico 1 and Epico 2.0 guidelines, except as regards de-escalation from echinocandins to fluconazole in Candida glabrata infections (p=0.055), and the duration of antifungal treatment in both candidemia and peritoneal candidiasis.
ConclusionsThis multifaceted educational intervention based on the Epico Project recommendations improved specialist knowledge of the management of invasive candidiasis in critically ill patients.
Las infecciones por Candida son frecuentes en los pacientes críticos y conllevan un incremento de la morbimortalidad. Nuestro objetivo es evaluar el impacto de una intervención educativa múltiple fundamentados en las recomendaciones de los proyectos basados en metodología Delphi Epico 1 y Epico 2.0.
DiseñoEspecialistas respondieron anónimamente 2 cuestionarios a través de Internet antes y después de una intervención educativa múltiple que consistió en: sesiones educativas de 60min, distribución de kits de diapositivas y guías de bolsillo, y un caso virtual interactivo presentado en una teleconferencia y disponible para su consulta a través de Internet.
ÁmbitoSetenta y cuatro hospitales españoles.
ParticipantesEspecialistas de las unidades de cuidados intensivos de los hospitales participantes.
Variables de interésConocimientos y práctica clínica valorada a través de un cuestionario. El test de McNemar se empleó para comparar las respuestas entre los periodos pre y postintervención.
ResultadosUn total de 255 y 248 especialistas completaron ambos cuestionarios en los 2 periodos, respectivamente. Los cuestionarios preintervención mostraron que muchos especialistas desconocían el mejor tratamiento de la candidiasis invasiva. Después de las 2 intervenciones educativas, el conocimiento de los especialistas y las prácticas reportadas estaban más próximos a las recomendaciones de las guías Epico 1 y 2.0, excepto para el desescalado de equinocandinas a fluconazol en las infecciones por C. glabrata (p=0,055) y en la duración del tratamiento antifúngico en la candidemia y en la peritonitis candidiásica.
ConclusionesEsta intervención educativa múltiple basada en las recomendaciones del proyecto EPICO mejoró el conocimiento de los profesionales acerca del manejo de la candidiasis invasiva en el paciente crítico.
The Extended Prevalence of Infection in Intensive Care (EPIC II) study highlighted the importance of infections by Candida species in patients in intensive care units (ICU). In this point prevalence study including critically ill patients in 1265 ICUs worldwide, Candida species were isolated in 17% of all positive cultures,1 and the prevalence of candidemia was 6.87 episodes per 1000 patients admitted to an ICU.2 The prevalence of invasive Candida infections varies with place and time, and methodological differences make it difficult to compare epidemiological reports. Thus, the real prevalence of Candida infections in non-neutropenic critically ill patients is unknown.
Invasive Candida infections are associated with high morbidity and mortality, especially in ICU patients. The crude mortality rate of patients who develop candidemia ranges between 40% and 60%.3–6 A French study found no significant differences in mortality (45.9%) between patients with invasive Candida infections without candidemia and those with candidemia.7 An Italian study found 40.2% overall mortality and pointed out that mortality varied widely with the species causing the infection, from 26.5% for C. parapsilosis to 77.8% for C. tropicalis.8 Nevertheless, underlying disease and comorbidities make it difficult to determine mortality directly attributable to Candida infection. Moreover, early detection and early appropriate treatment have an important impact on mortality.9–11
Although antifungal drugs approved in recent years may improve effectiveness against invasive candidiasis, scientific societies’ recommendations vary widely,12–14 and the best management strategy remains controversial. For this reason, five scientific societies in Spain collaborated in the EPICO Project, program of continued improvement in the quality of care of critically ill patients, in which the Delphi method was used to elaborate consensus guidelines for the diagnosis and treatment of invasive candidiasis in critically ill patients, developing twelve recommendations focused on epidemiology, scores, diagnostic tools, treatment and de-escalation strategies;15 and for the treatment of the invasive candidiasis in critically ill adult patients in special situations, developing 13 recommendations to improve the clinical management of this population in different specific scenarios.16
We aimed to determine whether a multifaceted educational intervention including serious games based on the EPICO Project recommendations could improve physicians’ management of neutropenic or non-neutropenic critically ill adult patients with invasive candidiasis in different scenarios. We hypothesized that the intervention would bring physicians’ attitude and behavior closer in line with the recommendations.
MethodsStudy sitesAll 74 hospitals involved in the validation phases of the EPICO Project – Epico 1 and Epico 2.0 – participated in this study. The ethics committees at each participating hospital approved the study and waived the informed consent requirement since the intervention was done on physician and no data were collected from patients. The Epico project has been endorsed by the Spanish Association of Mycology (AEM) as promoter, the Spanish Society of Infectious Diseases and Clinical Microbiology (SEIMC); the Spanish Society of Anesthesiology, Reanimation and Pain Therapeutics (SEDAR); The Spanish Society of Critical, Intensive and Coronary Medicine Units (SEMICYUC); and the Spanish Society of Chemotherapy (SEQ). No fees were provided for participation in the surveys.
Study designWe used a before-and-after design. Specialist physicians attending critically ill patients anonymously completed the same 13-question online survey of Epico 1 (See online material, Table 1) and the 12-question online survey of Epico 2.0 (See online material, Table 2), both designed to assess their knowledge and practices with respect to the recommendations in the Epico project at two times: preintervention (in the period from October 15, 2012 through October 24, 2012 prior to the educational meetings) and postintervention (in the period from November 25, 2012 through December 15, 2012 after the educational meetings) for Epico 1; and preintervention (in the period from October 7, 2013 through November 7, 2013 prior to the educational meetings) and postintervention (in the period from December 7, 2013 through February 7, 2014 after the educational meetings) for Epico 2.0.
We used a before-and-after design. Specialist physicians attending critically ill patients anonymously completed the same 13-question online survey of Epico 1 (See online material, Table 1) and the 12-question online survey of Epico 2.0 (See online material, Table 2), both designed to assess their knowledge and practices with respect to the recommendations in the Epico project at two times: preintervention (in the period from October 15, 2012 through October 24, 2012 prior to the educational meetings) and postintervention (in the period from November 25, 2012 through December 15, 2012 after the educational meetings) for Epico 1; and preintervention (in the period from October 7, 2013 through November 7, 2013 prior to the educational meetings) and postintervention (in the period from December 7, 2013 through February 7, 2014 after the educational meetings) for Epico 2.0.
Educational interventionBetween October 26, 2012 and November 24, 2012, and between November 8, 2013, and December 6, 2013, representatives from each of the hospitals participating in the validation phase of the Epico 1 and Epico 2.0 studies led 60-min educational sessions at their corresponding hospital. All specialists involved in treating critically ill patients with invasive candidiasis were encouraged to attend the sessions. Meetings were organized as follows: explanation of the objectives of the Epico Project (10min); explanation of the Delphi method and results of the surveys completed by the panel of 24 experts for each study (10min), discussion of the validation of the recommendations phase (10min), and specification of the, respectively, 12 and 13 final recommendations and the rationale behind each (30min). All attendees received a slide kit to help them explain the EPICO Project, the Delphi method, and the recommendations to their colleagues, as well as pocket guides with the recommendations. The recommendations of the EPICO Project are listed in the original manuscripts.15,16 Two interactive virtual clinical cases of invasive candidiasis (serious game) were presented at a teleconference including all participating centers and made available for consultation online (http://www.edusepsis.org/formacion/epico/index.html). The clinical cases included questions about invasive candidiasis diagnosis and treatment, with multiple choice answers, only one correct. Answers are followed by simulations of the clinical consequences and rationale for the correct answer.
Statistical analysisWe used the McNemar test or the T Student to compare the responses on the preintervention and postintervention surveys; we analyzed only the responses of the specialists who completed both surveys. We considered differences with p<0.05 significant. All analyses were conducted using SPSS version 15.0 (SPSS, Chicago, Illinois).
ResultsA total of 980 specialists attended the educational sessions of Epico 1; 617 of these had completed the preintervention survey and 276 went on to complete the postintervention survey. Both surveys were completed by 255 specialists, of whom 137 (53.7%) were intensivists, 116 (45.5%) were anesthesiologists, 1 (0.4%) was an internist, and 1 (0.4%) was a cardiologist.
Meanwhile, 551 specialists attended the educational sessions of Epico 2.0; 550 of these completed the preintervention survey and 249 went on to complete the postintervention survey. Both surveys were completed by 248 specialists, of whom 155 (62.5%) were intensivists, 35 (14.1%) were anesthesiologists, 5 (2.0%) were internists, 2 (0.8%) were gastroenterologists, 1 (0.4%) was a microbiologist, and 1 (0.4%) was a pulmonologist. 49 participants (19.8%) did not indicate their specialty.
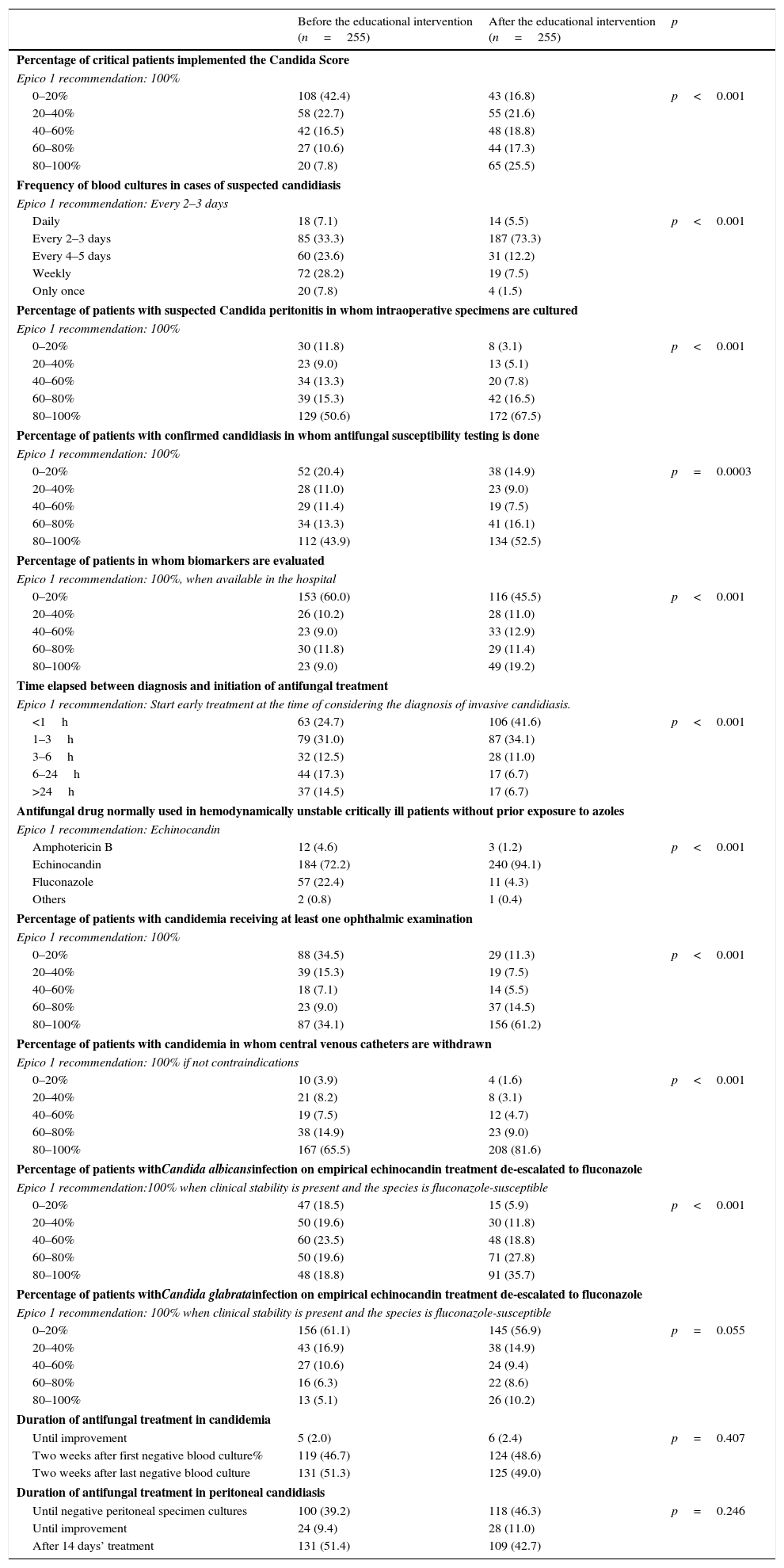
Tables 1 and 2 summarize the results of the McNemar tests comparing the preintervention and postintervention responses by the specialists who completed both surveys. The preintervention surveys showed that many specialists were unaware of the best approach to manage invasive candidiasis. Specialists’ knowledge and practices after the educational intervention were more in line with nearly all the recommendations specified in the EPICO Project consensus. After the educational intervention of the Epico 1 Study, physicians caring for critically ill patients were more prone to use the Candida Score and biomarkers for early identification of patients, and to obtain blood cultures or intraabdominal specimens in cases of suspected invasive candidiasis. The assessment of antifungal susceptibility in culture reports did also improve. Specialists reported also a shorter time to start of therapy, increased use of echinocandins in unstable patients, more frequent ophthalmic evaluations, and improved source control (central catheter removal). De-escalation from echinocandins to fluconazole also improved after the intervention for infections by C. albicans. The only issues for which the responses did not show a statistically significant shift toward the recommendations were de-escalation from echinocandins to fluconazole in Candida glabrata infections, where a trend (p=0.055) toward increased agreement with the recommendations was observed, and the duration of antifungal treatment in both candidemia and peritoneal candidiasis.
Evaluation of knowledge and practices of clinicians before and after the educational intervention of Epico 1 Study.
| Before the educational intervention (n=255) | After the educational intervention (n=255) | p | |
|---|---|---|---|
| Percentage of critical patients implemented the Candida Score | |||
| Epico 1 recommendation: 100% | |||
| 0–20% | 108 (42.4) | 43 (16.8) | p<0.001 |
| 20–40% | 58 (22.7) | 55 (21.6) | |
| 40–60% | 42 (16.5) | 48 (18.8) | |
| 60–80% | 27 (10.6) | 44 (17.3) | |
| 80–100% | 20 (7.8) | 65 (25.5) | |
| Frequency of blood cultures in cases of suspected candidiasis | |||
| Epico 1 recommendation: Every 2–3 days | |||
| Daily | 18 (7.1) | 14 (5.5) | p<0.001 |
| Every 2–3 days | 85 (33.3) | 187 (73.3) | |
| Every 4–5 days | 60 (23.6) | 31 (12.2) | |
| Weekly | 72 (28.2) | 19 (7.5) | |
| Only once | 20 (7.8) | 4 (1.5) | |
| Percentage of patients with suspected Candida peritonitis in whom intraoperative specimens are cultured | |||
| Epico 1 recommendation: 100% | |||
| 0–20% | 30 (11.8) | 8 (3.1) | p<0.001 |
| 20–40% | 23 (9.0) | 13 (5.1) | |
| 40–60% | 34 (13.3) | 20 (7.8) | |
| 60–80% | 39 (15.3) | 42 (16.5) | |
| 80–100% | 129 (50.6) | 172 (67.5) | |
| Percentage of patients with confirmed candidiasis in whom antifungal susceptibility testing is done | |||
| Epico 1 recommendation: 100% | |||
| 0–20% | 52 (20.4) | 38 (14.9) | p=0.0003 |
| 20–40% | 28 (11.0) | 23 (9.0) | |
| 40–60% | 29 (11.4) | 19 (7.5) | |
| 60–80% | 34 (13.3) | 41 (16.1) | |
| 80–100% | 112 (43.9) | 134 (52.5) | |
| Percentage of patients in whom biomarkers are evaluated | |||
| Epico 1 recommendation: 100%, when available in the hospital | |||
| 0–20% | 153 (60.0) | 116 (45.5) | p<0.001 |
| 20–40% | 26 (10.2) | 28 (11.0) | |
| 40–60% | 23 (9.0) | 33 (12.9) | |
| 60–80% | 30 (11.8) | 29 (11.4) | |
| 80–100% | 23 (9.0) | 49 (19.2) | |
| Time elapsed between diagnosis and initiation of antifungal treatment | |||
| Epico 1 recommendation: Start early treatment at the time of considering the diagnosis of invasive candidiasis. | |||
| <1h | 63 (24.7) | 106 (41.6) | p<0.001 |
| 1–3h | 79 (31.0) | 87 (34.1) | |
| 3–6h | 32 (12.5) | 28 (11.0) | |
| 6–24h | 44 (17.3) | 17 (6.7) | |
| >24h | 37 (14.5) | 17 (6.7) | |
| Antifungal drug normally used in hemodynamically unstable critically ill patients without prior exposure to azoles | |||
| Epico 1 recommendation: Echinocandin | |||
| Amphotericin B | 12 (4.6) | 3 (1.2) | p<0.001 |
| Echinocandin | 184 (72.2) | 240 (94.1) | |
| Fluconazole | 57 (22.4) | 11 (4.3) | |
| Others | 2 (0.8) | 1 (0.4) | |
| Percentage of patients with candidemia receiving at least one ophthalmic examination | |||
| Epico 1 recommendation: 100% | |||
| 0–20% | 88 (34.5) | 29 (11.3) | p<0.001 |
| 20–40% | 39 (15.3) | 19 (7.5) | |
| 40–60% | 18 (7.1) | 14 (5.5) | |
| 60–80% | 23 (9.0) | 37 (14.5) | |
| 80–100% | 87 (34.1) | 156 (61.2) | |
| Percentage of patients with candidemia in whom central venous catheters are withdrawn | |||
| Epico 1 recommendation: 100% if not contraindications | |||
| 0–20% | 10 (3.9) | 4 (1.6) | p<0.001 |
| 20–40% | 21 (8.2) | 8 (3.1) | |
| 40–60% | 19 (7.5) | 12 (4.7) | |
| 60–80% | 38 (14.9) | 23 (9.0) | |
| 80–100% | 167 (65.5) | 208 (81.6) | |
| Percentage of patients withCandida albicansinfection on empirical echinocandin treatment de-escalated to fluconazole | |||
| Epico 1 recommendation:100% when clinical stability is present and the species is fluconazole-susceptible | |||
| 0–20% | 47 (18.5) | 15 (5.9) | p<0.001 |
| 20–40% | 50 (19.6) | 30 (11.8) | |
| 40–60% | 60 (23.5) | 48 (18.8) | |
| 60–80% | 50 (19.6) | 71 (27.8) | |
| 80–100% | 48 (18.8) | 91 (35.7) | |
| Percentage of patients withCandida glabratainfection on empirical echinocandin treatment de-escalated to fluconazole | |||
| Epico 1 recommendation: 100% when clinical stability is present and the species is fluconazole-susceptible | |||
| 0–20% | 156 (61.1) | 145 (56.9) | p=0.055 |
| 20–40% | 43 (16.9) | 38 (14.9) | |
| 40–60% | 27 (10.6) | 24 (9.4) | |
| 60–80% | 16 (6.3) | 22 (8.6) | |
| 80–100% | 13 (5.1) | 26 (10.2) | |
| Duration of antifungal treatment in candidemia | |||
| Until improvement | 5 (2.0) | 6 (2.4) | p=0.407 |
| Two weeks after first negative blood culture% | 119 (46.7) | 124 (48.6) | |
| Two weeks after last negative blood culture | 131 (51.3) | 125 (49.0) | |
| Duration of antifungal treatment in peritoneal candidiasis | |||
| Until negative peritoneal specimen cultures | 100 (39.2) | 118 (46.3) | p=0.246 |
| Until improvement | 24 (9.4) | 28 (11.0) | |
| After 14 days’ treatment | 131 (51.4) | 109 (42.7) | |
Data are showed in No. (%).
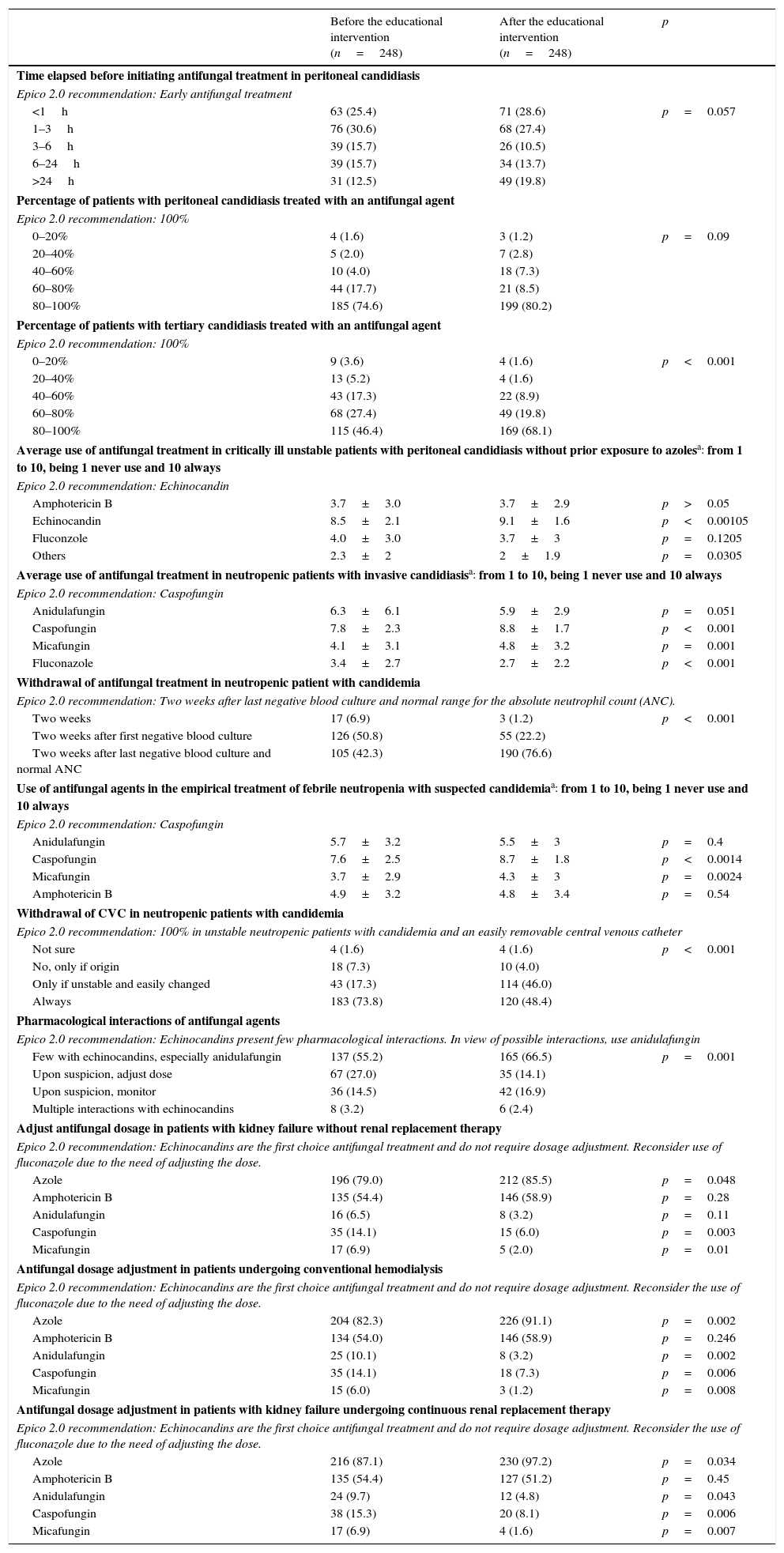
Evaluation of knowledge and practices of clinicians before and after the educational intervention of Epico 2.0.
| Before the educational intervention (n=248) | After the educational intervention (n=248) | p | |
|---|---|---|---|
| Time elapsed before initiating antifungal treatment in peritoneal candidiasis | |||
| Epico 2.0 recommendation: Early antifungal treatment | |||
| <1h | 63 (25.4) | 71 (28.6) | p=0.057 |
| 1–3h | 76 (30.6) | 68 (27.4) | |
| 3–6h | 39 (15.7) | 26 (10.5) | |
| 6–24h | 39 (15.7) | 34 (13.7) | |
| >24h | 31 (12.5) | 49 (19.8) | |
| Percentage of patients with peritoneal candidiasis treated with an antifungal agent | |||
| Epico 2.0 recommendation: 100% | |||
| 0–20% | 4 (1.6) | 3 (1.2) | p=0.09 |
| 20–40% | 5 (2.0) | 7 (2.8) | |
| 40–60% | 10 (4.0) | 18 (7.3) | |
| 60–80% | 44 (17.7) | 21 (8.5) | |
| 80–100% | 185 (74.6) | 199 (80.2) | |
| Percentage of patients with tertiary candidiasis treated with an antifungal agent | |||
| Epico 2.0 recommendation: 100% | |||
| 0–20% | 9 (3.6) | 4 (1.6) | p<0.001 |
| 20–40% | 13 (5.2) | 4 (1.6) | |
| 40–60% | 43 (17.3) | 22 (8.9) | |
| 60–80% | 68 (27.4) | 49 (19.8) | |
| 80–100% | 115 (46.4) | 169 (68.1) | |
| Average use of antifungal treatment in critically ill unstable patients with peritoneal candidiasis without prior exposure to azolesa: from 1 to 10, being 1 never use and 10 always | |||
| Epico 2.0 recommendation: Echinocandin | |||
| Amphotericin B | 3.7±3.0 | 3.7±2.9 | p>0.05 |
| Echinocandin | 8.5±2.1 | 9.1±1.6 | p<0.00105 |
| Fluconzole | 4.0±3.0 | 3.7±3 | p=0.1205 |
| Others | 2.3±2 | 2±1.9 | p=0.0305 |
| Average use of antifungal treatment in neutropenic patients with invasive candidiasisa: from 1 to 10, being 1 never use and 10 always | |||
| Epico 2.0 recommendation: Caspofungin | |||
| Anidulafungin | 6.3±6.1 | 5.9±2.9 | p=0.051 |
| Caspofungin | 7.8±2.3 | 8.8±1.7 | p<0.001 |
| Micafungin | 4.1±3.1 | 4.8±3.2 | p=0.001 |
| Fluconazole | 3.4±2.7 | 2.7±2.2 | p<0.001 |
| Withdrawal of antifungal treatment in neutropenic patient with candidemia | |||
| Epico 2.0 recommendation: Two weeks after last negative blood culture and normal range for the absolute neutrophil count (ANC). | |||
| Two weeks | 17 (6.9) | 3 (1.2) | p<0.001 |
| Two weeks after first negative blood culture | 126 (50.8) | 55 (22.2) | |
| Two weeks after last negative blood culture and normal ANC | 105 (42.3) | 190 (76.6) | |
| Use of antifungal agents in the empirical treatment of febrile neutropenia with suspected candidemiaa: from 1 to 10, being 1 never use and 10 always | |||
| Epico 2.0 recommendation: Caspofungin | |||
| Anidulafungin | 5.7±3.2 | 5.5±3 | p=0.4 |
| Caspofungin | 7.6±2.5 | 8.7±1.8 | p<0.0014 |
| Micafungin | 3.7±2.9 | 4.3±3 | p=0.0024 |
| Amphotericin B | 4.9±3.2 | 4.8±3.4 | p=0.54 |
| Withdrawal of CVC in neutropenic patients with candidemia | |||
| Epico 2.0 recommendation: 100% in unstable neutropenic patients with candidemia and an easily removable central venous catheter | |||
| Not sure | 4 (1.6) | 4 (1.6) | p<0.001 |
| No, only if origin | 18 (7.3) | 10 (4.0) | |
| Only if unstable and easily changed | 43 (17.3) | 114 (46.0) | |
| Always | 183 (73.8) | 120 (48.4) | |
| Pharmacological interactions of antifungal agents | |||
| Epico 2.0 recommendation: Echinocandins present few pharmacological interactions. In view of possible interactions, use anidulafungin | |||
| Few with echinocandins, especially anidulafungin | 137 (55.2) | 165 (66.5) | p=0.001 |
| Upon suspicion, adjust dose | 67 (27.0) | 35 (14.1) | |
| Upon suspicion, monitor | 36 (14.5) | 42 (16.9) | |
| Multiple interactions with echinocandins | 8 (3.2) | 6 (2.4) | |
| Adjust antifungal dosage in patients with kidney failure without renal replacement therapy | |||
| Epico 2.0 recommendation: Echinocandins are the first choice antifungal treatment and do not require dosage adjustment. Reconsider use of fluconazole due to the need of adjusting the dose. | |||
| Azole | 196 (79.0) | 212 (85.5) | p=0.048 |
| Amphotericin B | 135 (54.4) | 146 (58.9) | p=0.28 |
| Anidulafungin | 16 (6.5) | 8 (3.2) | p=0.11 |
| Caspofungin | 35 (14.1) | 15 (6.0) | p=0.003 |
| Micafungin | 17 (6.9) | 5 (2.0) | p=0.01 |
| Antifungal dosage adjustment in patients undergoing conventional hemodialysis | |||
| Epico 2.0 recommendation: Echinocandins are the first choice antifungal treatment and do not require dosage adjustment. Reconsider the use of fluconazole due to the need of adjusting the dose. | |||
| Azole | 204 (82.3) | 226 (91.1) | p=0.002 |
| Amphotericin B | 134 (54.0) | 146 (58.9) | p=0.246 |
| Anidulafungin | 25 (10.1) | 8 (3.2) | p=0.002 |
| Caspofungin | 35 (14.1) | 18 (7.3) | p=0.006 |
| Micafungin | 15 (6.0) | 3 (1.2) | p=0.008 |
| Antifungal dosage adjustment in patients with kidney failure undergoing continuous renal replacement therapy | |||
| Epico 2.0 recommendation: Echinocandins are the first choice antifungal treatment and do not require dosage adjustment. Reconsider the use of fluconazole due to the need of adjusting the dose. | |||
| Azole | 216 (87.1) | 230 (97.2) | p=0.034 |
| Amphotericin B | 135 (54.4) | 127 (51.2) | p=0.45 |
| Anidulafungin | 24 (9.7) | 12 (4.8) | p=0.043 |
| Caspofungin | 38 (15.3) | 20 (8.1) | p=0.006 |
| Micafungin | 17 (6.9) | 4 (1.6) | p=0.007 |
Data are shown in No. (%).
Meanwhile, after the educational intervention of the Epico 2.0 Study, specialists caring for critically ill adult patients in special situations were more prone to initiate antifungal treatment in patients with tertiary peritonitis; administer echinocandins in patients with peritoneal candidiasis with previous exposure to azoles and in neutropenic patients with invasive candidiasis; discontinue the antifungal treatment 14 days following the first negative blood culture; and adjust the dose of azoles in patients with kidney failure without renal replacement therapy. In cases of patients undergoing hemodialysis, a significant shift toward dosage adjustment of all antifungals has been observed. Also, the educational intervention lead to a greater administration of echinocandins–caspofungin, micafungin–in patients with febrile neutropenia and suspected candidemia, and a greater withdrawal of central venous catheter in unstable patients. Finally, a greater tendency of initiating antifungal treatment in peritoneal candidiasis was observed, although it did not reach statistical significance (p=0.09).
DiscussionAs far as we know, this is the first time that multifaceted educational interventions based on previous Delphi recommendations about managing invasive candidiasis in critically ill patients and critically ill adult patients in special situations have been reported. More than a quarter of all ICUs in Spain participated in this study, underlining specialists’ recognition of the importance of invasive candidiasis in critical patients because of its high prevalence and associated morbidity and mortality. Before the EPICO Project, there was no strong consensus on the best approach to managing invasive candidiasis, so it is not surprising that many specialists’ responses on the preintervention survey were not in line with the recommendations specified in the Epico 1 and Epico 2.0 consensus.
After the educational interventions, specialists’ knowledge and practices were more in line with all the recommendations specified by the EPICO Project. However, the intervention was not successful in changing specialist's attitude in de-escalation of echinocandins in C. glabrata infections and treatment duration. Probably, the main reason explaining this lack of change is that the rationale supporting echinocandins de-escalation in this setting could be considered weak by the clinicians and that there is not a specific Epico recommendation for the duration of treatment.
Likewise, no change in the attitude regarding the time elapsed before initiation of treatment in patients with peritoneal candidiasis was observed.
One strength of our intervention was the use of serious games. Despite the evidence about the use of games as an educational strategy to improve health professionals’ performance (e.g. adherence to standards of care) through improving their knowledge, skills and attitudes is controversial,17 some studies have shown promising results to improve the treatment of severe infections.18
Educational programs have been successfully used to improve quality in many areas. Numerous programs in the ICU have achieved gains in both process of care variables and outcomes. Ferrer et al.19 showed that an educational program based on the Surviving Sepsis Campaign guidelines improved compliance with recommendations and reduced mortality in patients with severe sepsis or septic shock in a large group of Spanish ICUs. Another large-scale project carried out in Spanish ICUs, the Bacteremia Zero project, successfully reduced the number of catheter-related bloodstream infections, showing that it is possible to improve the safety culture.20
In a recent systematic review of knowledge translation interventions, Sinuff et al.21 concluded that the most important factors for improving process of care were establishing protocols rather than education per se. To establish useful protocols, the current evidence about best practices needs to be available. In the EPICO Project, a large number of experts in invasive candidiasis used the Delphi method to review the available evidence and reach a consensus about key issues to establish recommendations for the management of invasive candidiasis. These recommendations are a starting point for further studies aimed at improving care for this widespread infection.
LimitationsOur study has several limitations. First, there is a risk of participation bias; the ICUs participating in the study may not be representative of all ICUs in Spain, let alone of ICUs in other countries. However, the sample included both university and community hospitals in all regions of the country and represented 26.3% of all ICUs in Spain. Likewise, the physicians that participated in the study and especially those that responded to any of both surveys may be more motivated to improve than those who did not. Furthermore, the design of our study in which behavior was self-reported allows us to appreciate changes in participants’ knowledge and attitude but cannot be considered a reliable measure of actual behavior. The time between the educational interventions and assessments was short, and we cannot know the long-term impact of the intervention. However, the short interval between the interventions and assessments makes it unlikely that the improvements observed are due to a secular trend. The duration of the interventions were also short; a longer educational period might have enabled greater improvements in compliance with the recommendations. Future studies should evaluate the long-term impact of a more sustained intervention on physicians’ actual behavior and on patients’ outcomes.
To overcome these limitations, the EPICO Project implemented a sustained intervention with new consensus and educational programs on invasive candidiasis management in critically ill patients every year: Epico 1 including recommendations of the use of non-culture based methods of microbiological diagnostic techniques and of the de-escalation of the antifungal treatment; Epico 2.0 including recommendations of treatment in patients with organ failures,21 Epico 3.0 including recommendations of treatment for postoperative patients, medical patients and hematological patients (not reported), and Epico 4 including recommendations for antifungal stewardship (on-going). The EPICO recommendations are concordant with the recently published guidelines.22
ConclusionsBefore the EPICO Project, there was no consensus about the best approach to diagnosing and treating invasive candidiasis. The preintervention surveys revealed that specialists involved in treating critical patients with invasive candidiasis often lacked knowledge about important specifics in managing these patients. These multifaceted educational interventions improved participants’ knowledge and attitudes about the management of invasive candidiasis.
EPICO Project Group: List of project coordinators, panel members and attending physicians are listed in the Online material.
EPICO Project Group: List of project coordinators, panel members and attending physicians are listed in the Online material.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestsThese interventions have been sponsored by MSD Laboratories, Spain.
Carmen Romero and Ainhoa Torres (Entheos editorial group) for their excellent work and dedication to this project.