Microcirculatory alterations play a pivotal role in sepsis-related morbidity and mortality. However, since the microcirculation has been a “black box”, current hemodynamic management of septic patients is still guided by macrocirculatory parameters. In the last decades, the development of several technologies has shed some light on microcirculatory evaluation and monitoring, and the possibility of incorporating microcirculatory variables to clinical practice no longer seems to be beyond reach. The present review provides a brief summary of the current technologies for microcirculatory evaluation, and attempts to explore the potential role and benefits of their integration to the resuscitation process in critically ill septic patients.
Las alteraciones microcirculatorias juegan un papel fundamental en la morbimortalidad asociada a la sepsis. Sin embargo, puesto que la microcirculación ha sido una «caja negra», el manejo hemodinámico actual del paciente séptico sigue basándose en la corrección de parámetros macrocirculatorios. Durante las últimas décadas, el desarrollo de diferentes tecnologías ha permitido arrojar algo de luz sobre la posibilidad de evaluar y monitorizar la microcirculación, y a día de hoy, la incorporación de variables microcirculatorias a la práctica clínica ya no parece una quimera. El presente trabajo de revisión presenta, de forma sucinta, las diferentes tecnologías que permiten evaluar la microcirculación, y pretende explorar el posible papel, así como los potenciales beneficios, de la integración de estas tecnologías en el proceso de reanimación del paciente crítico séptico.
Sepsis is associated with high morbidity and mortality rates, and its management represents a difficult challenge for the clinician. Septic shock is usually characterized by hemodynamic impairment, classically defined by a decreased vascular tone with some degree of hypovolemia, leading to circulatory failure. In current international sepsis guidelines, hemodynamic management pivots on supporting this failing circulation, aiming at correcting global markers of tissue oxygenation1,2 (specifically, lactate and central venous oxygen saturation –ScvO2–) via the optimization of oxygen delivery. Lactate has been proposed as a clinical marker of anaerobic metabolism, and ScvO2 as a marker of oxygen transport and oxygen consumption imbalance. However, these parameters allow an evaluation of tissue wellness or adequacy of perfusion from a global perspective, and may be of limited usefulness in diseases such as sepsis, where an important heterogeneity in tissue perfusion has been demonstrated. Furthermore, signs of tissue hypoperfusion may still persist even when these global flow parameters seem to be corrected. Indeed, available evidence strongly suggests that the principal motor of sepsis is microcirculatory dysfunction.3 These microcirculatory alterations may produce tissue hypoxia, as a result of oxygen supply-demand imbalance at the cellular level, leading to the development of cellular and organ dysfunction and potentially, death of the individual.4 Although until recent times microcirculation has been a “black box” because of technological limitations, development of microcirculatory evaluation techniques has allowed direct study of this phenomenon.
Microcirculation is considered to be the main responsible for ensuring tissue wellness. Its principal objective is to transport oxygen and nutrients to tissue cells and thus preserve organ function. It consists of a complex network of small blood vessels (<100μm diameter) such as arterioles, capillaries and venules. Arterioles are responsible for maintaining vascular tone, and respond to extrinsic and intrinsic stimuli to modulate local arteriolar tone to match local metabolic demands. Capillaries act as the primary exchange place for supplying oxygen and receiving metabolic cell waste products. These capillaries converge into the venules, where leukocyte interactions and vascular permeability changes take place. Moreover, the microcirculation is a complex system that also involves different cell types with different functions such as endothelial cells inside the microvessels, smooth muscle cells (mostly in arterioles), red blood cells, leukocytes, and plasma components in blood. All these elements interact between them and are regulated by different complex mechanisms controlling microcirculatory perfusion.3
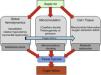
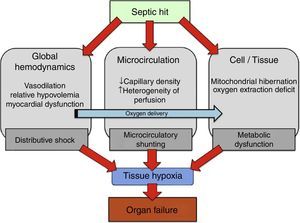
Recent experimental and clinical studies have reported microcirculatory alterations in sepsis. These studies observed a decrease in capillary density that determines an increase in the diffusion distance of oxygen to tissues,5 and furthermore an increase in heterogeneity of perfusion was also described.6 Overall, this microcirculatory derangements involve the consequent presence of under or not perfused capillaries in close proximity to well perfused capillaries. Therefore, these functionally vulnerable microcirculatory areas may become hypoxic, resulting in an oxygen extraction deficit. This phenomenon has been termed “microcirculatory shunting” and plays a major role in the pathophysiology of sepsis and multi-organ failure3,5 (Fig. 1). Of note, in clinical practice, systemic hemodynamic- and global oxygen-derived variables may fail to detect this phenomenon.
Physiopathology of oxygen transport alterations in sepsis. Sepsis may induce complex alterations at different levels (including global hemodynamics, microcirculation, and cell metabolism) of the oxygen transport “cascade”, leading to tissue hypoxia and the consequent development of organ failure.
According to current evidence, bedside evaluation of microcirculation may complement our current approach in the management of septic patients.
Current methods for microcirculatory monitoring in septic statesEvaluation of the microcirculation in critically ill patients presents certain methodological and technical difficulties that have to be taken into account. It is fundamental to select tissue beds that are easily accessible to evaluate and, even more important, sufficiently representative of the central microcirculation,5 as any microcirculatory monitoring technique can just evaluate the tissue bed where it is applied. Conceptually, current techniques that permit monitoring the microcirculation can be classified in two main groups (Table 1):
- (1)
Methods that allow direct visualization of the microvascular network and microcirculatory blood flow.
- (2)
Methods that allow evaluation of local tissue oxygenation as a surrogate of microcirculatory blood flow.
Available techniques to evaluate the microcirculation at the bedside. Most relevant techniques currently available to monitor the microcirculation at the bedside are summarized.
| Bedside techniques to evaluate the microcirculation | Measured variable | Advantages | Limitations |
|---|---|---|---|
| Direct evaluation | |||
| Clinical examination (CRT, mottling score, central-to-toe T° gradient) | Regional peripheral perfusion | No technological device required Easy and rapid applicability | Qualitative evaluation Limited use for shock monitoring and titrating therapy |
| Videomicroscopy (second and third generation digital videomicroscopes) | Microcirculatory blood flow; Vascular density; Heterogeneity of perfusion | Gold Standard Semi-quantitative evaluation Potential use for bedside monitoring and titrating therapy | Technical issues for high-quality videos acquisition No immediate availability of microcirculatory video analysis |
| Indirect evaluation | |||
| Mucosal pCO2 derived measurements (gastric and sublingual capnometry) | Tissue CO2 | Quantitative evaluation Potential use for bedside assessment of flow adequacy | Technical issues interfering gastric tonometry measurements |
| pO2 electrodes (tissue and transcutaneous oxygenation) | Tissue pO2 Oxygen Challenge Test (dynamic test to assess adequacy of DO2) | Quantitative evaluation Potential use for bedside assessment of convective O2 transport adequacy | Measurement variability (value derived from a mixture of arterioles, capillaries and venules) |
| Near Infra-Red Spectroscopy (NIRS) (skeletal muscle oxygenation) | Tissue O2 saturation (StO2) Vascular occlusion test (dynamic test to assess O2 local consumption and endothelial function) | Quantitative evaluation | StO2 measurements altered by edema/fat tissue Technology diversity, exploring different tissue depths, causing lack of standardized values |
CRT, Capillary refill time; pCO2, carbon dioxide tension; pO2, oxygen tension; DO2, global oxygen delivery; StO2, tissue oxygen saturation.
Certain classic clinical signs may be used at the bedside as surrogates of the presence of an impaired circulation. This is supported by the physiological concept that the peripheral circulation is the first to reflect a disturbance of the hemodynamic status that may lead to shock. This noninvasive peripheral perfusion evaluation include clinical signs such as the capillary refill time (CRT), the mottling score,7 and body temperature related scores such as the central-to-toe temperature gradient.8,9 These signs are easy to evaluate at the bedside, and have shown to be related to the severity of organ dysfunction and outcome, independently of systemic hemodynamics.8 However, these methods present some important limitations that have to be taken into account: First, they are difficult to quantify, limiting its application for management of shock and for titrating therapy. Moreover, these clinical methods focus particularly on the evaluation of skin (a circulatory bed with independent mechanisms of regulation), probably not an optimal tissue bed for evaluating central microcirculation.10 These limitations have moved clinical signs evaluation away from daily clinical practice, and they are considered a merely qualitative assessment tool, but not a real monitoring approach.
VideomicroscopyBased on intra-vital microscopy techniques, incident dark-field illumination techniques incorporated into handheld microscopes allowed the birth of the first handheld videomicroscopes. First, orthogonal polarization spectral (OPS) imaging and later, sidestream dark field (SDF) imaging, were introduced to directly evaluate the microcirculation at the bedside. After applying a light source on a surface, the light is reflected by the deeper layers of the tissue providing trans-illumination of the superficial layers of the tissue (thus limiting its application on organs or tissue surfaces covered by thin epithelial layers). As the selected wavelength (530nm) is absorbed by the hemoglobin, red blood cells appear as black/gray bodies flowing inside capillaries (absorbed light) over a white tissue background (reflected light). Thus, only functional capillaries (with red blood cells flow) will be observed in contrast to nonfunctional capillaries (without red blood cells flow). In contrast with animal studies or patients undergoing surgery where several internal organs have been explored with videomicroscopy, in critically ill patients this technique has been applied in more accessible surfaces such as skin or mucosa, and especially the sublingual area.5
Microcirculatory image analysis has been a difficult challenge mainly as a result of hardware limitations, but also because different scoring systems have been proposed. In addition, interpretation of microcirculatory parameters presents certain considerations, and deserves to be properly addressed. Following the expert consensus conference conclusions,11 the ideal microcirculation analysis report should evaluate microvascular blood flow, vascular density, and heterogeneity of perfusion. Microcirculatory perfusion is evaluated assessing microvascular flow index (MFI) and the proportion of perfused vessels (PPV). Vascular density is evaluated assessing total vessel density (TVD) and perfused vessel density (PVD). Importantly, tissue perfusion is dependent on functional capillary density (reflected by PVD) and blood velocity (reflected by MFI). Given that cells are able to regulate oxygen extraction depending on variable flow, vascular density is believed to be a more limiting factor than blood velocity in assuring tissue oxygenation. Accordingly, homogeneous low flow should be better tolerated than heterogeneous flow by the cells, even when total blood flow is low.12 On the other hand, the presence of very high blood flow may theoretically reduce the time of hemoglobin to unload oxygen to the cells, and also may induce capillary endothelial damage due to shear-stress lesions.10 Finally, heterogeneity of perfusion is reflected by PPV in the investigated area and the heterogeneity index (Het Index) in the investigated organ. Assessing heterogeneity of perfusion is an essential key factor for evaluating the shunted fraction in septic shock.13 Most of these variables are quantitative, except for the flow related parameters, which are semi-quantitative, but nevertheless have demonstrated to be sensitive enough to evaluate microcirculatory performance in the literature.
Although routine microcirculatory evaluation using handheld videomicroscopes has not been implemented in clinical practice because of its technical limitations (use of analog video-cameras, lack of direct computer control, inability to automatically analyze images at the bedside, etc.), recent introduction of a third generation handheld videomicroscope referred as Cytocam – Incident Dark Field (IDF) imaging has allowed to overcome most of this discussed issues.14 This technology has a computer controlled high-resolution high-pixel density digital camera and, furthermore, advanced computer-assisted software that should allow automatic image analysis and thus, almost immediate access to microcirculatory evaluation. This important advance may facilitate the application of this technique at the bedside for clinical decision-making and guiding therapy. Nevertheless, while automatic real-time image analysis software is not available, recent studies have demonstrated that real-time qualitative assessment of microcirculatory perfusion with new generation videomicroscopes might be highly sensitive and specific for detecting impaired microcirculatory flow and capillary density.15 Therefore, bedside screening of patients at risk using qualitative real-time assessment of the microcirculation appears as a reality.
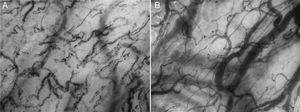
In the early phase of severe sepsis and septic shock, a significant decrease in vessel density and in proportion of perfused capillaries has been reported as compared to control subjects in sublingual videomicroscopy studies16,17 (Fig. 2. See supplementary material at the web). Furthermore, an increase in heterogeneity of vascular density and blood velocity between coexisting areas was also observed in these studies. In addition, these alterations were more severe in non-survivors and the rapid resolution of these microcirculatory changes following interventional therapy was also correlated with improved outcome, including mortality.16,18,19 Conversely, the persistence of these microcirculatory alterations after the first 24h was strongly and independently correlated with mortality secondary to circulatory failure in the early phase, and to multi-organ failure in the late phase.20
Cytocam-Incident Dark Field (IDF) images of sublingual microcirculation. Image A: early phase septic shock patient. A decrease of vessel density and of perfused vessels, and an increase of non-perfused/under-perfused vessels can be observed. Although not appreciable in static images, heterogeneity of microvascular blood flow velocity between coexisting areas is also present (normal or high microvascular blood flow vessels in close vicinity to non-perfused or low microvascular blood flow vessels). Image B: healthy volunteer. A normal vessel density and proportion of perfused vessels can be observed. Although not appreciable in static images, microvascular blood flow velocity is normal and homogeneous overall the studied areas. (Images courtesy of Braedius Medical and Prof. D. Payen, H. Lariboisiere).
Microcirculatory image-acquisition using handheld videomicroscopes presents some practical issues that may become important limitations of the technique if not addressed correctly. Optimal sublingual videomicroscopy is only feasible in cooperative or sedated patients. Furthermore, given the intrinsic variability of the microcirculation, it is recommended to record at least three sites, and up to possible five sites whenever possible, at each time-point evaluation. As the investigated surface is usually the sublingual area, secretions or blood clots may act as content artifacts and impair quality image, and need to be removed before image recording. In addition, movement artifacts can also alter the quality of the recorded video and difficult the image analysis task, so recording of steady videos is mandatory. Moreover, it is crucial to avoid excessive pressure while applying the videomicroscope on the tissue surface because this may impair flow in the microvessels and lead to underscoring microcirculatory perfusion.7
Overall, videomicroscopy is considered to be the gold standard technique for assessing microcirculation at the bedside. In a nearby future, this technique may allow to monitor the last frontier of tissue perfusion in daily clinical practice.
Indirect methods: evaluation of tissue oxygenationMucosal PCO2 derived measurementsTissue CO2 monitoring has been, for decades, the cornerstone of regional and microcirculatory evaluation in the critically ill patient. This technology is based on the principle that tissue CO2 depends on three factors: tissue CO2 production, blood flow to the tissue and arterial CO2 content. Presuming that arterial CO2 is constant (stable respiratory conditions), tissue CO2 reflects the balance between local CO2 production (dependent on basal metabolism) and blood flow to the tissue (CO2 washout phenomenon). Provided that arterial CO2 influences tissue CO2, the gradient between the two is usually calculated, and so expressed as the PCO2 gap. This parameter is considered to reflect the adequacy of blood flow to the tissue and has been proposed as a hypoperfusion marker and an additional endpoint for resuscitation.21
Given that the anatomical characteristics of intestinal mucosa make it particularly vulnerable to hypoxia, initially gastric tonometry raised a lot of interest. Gastric mucosal PCO2 gap values above 20mmHg have shown significant correlation with outcome in critically ill patients.22 Furthermore, in septic patients, an increased pCO2 gap showed a strong prognostic correlation with outcome, even when global hemodynamic parameters were already corrected.23,24 Unfortunately, gastric tonometry has been progressively abandoned primary due to its technical difficulties, complex measurements, and the interference of other factors such as enteral nutrition.
Other areas have been explored for the study of mucosal CO2, and sublingual optic capnometry has shown an excellent correlation with gastric CO2 measurements, and with similar prognostic value than gastric tonometry in critically ill patients.25 Interestingly, Creteur et al. demonstrated an inverse correlation between sublingual PCO2 and the density of perfused capillaries assessed by videomicroscopy in a population of septic patients, further strengthening the physiological value of CO2 measurement as a surrogate of tissue perfusion.26 Surprisingly, this attractive and non-invasive technique has not been properly developed for its clinical use, and is now restricted to experimental studies.
PO2 electrodesIn vivo measurements of tissue oxygen tension in humans date back to the 1950s, and have evolved from then until dynamic luminescence-based oxygen-sensing optodes technology, the current method for tissue oxygen tension monitoring in most animal and clinical studies. Tissue oxygen tension (tissue PO2, tPO2) is a measure of the partial pressure of oxygen in the interstitial space of a sampling volume of tissue (approximately 0.5mm3). Consequently, it could provide an indicator of the balance between local oxygen supply (dependent on microcirculation) and demand (dependent on mitochondrial respiration). It has been applied in deep vital organs in experimental conditions, and in peripheral tissues such as skin or skeletal muscle in human studies. As a general rule, baseline tissue oxygen tension values are lower in deeper and metabolically active organs in contrast with peripheral resting tissues. Although it appears to be a sensitive indicator of organ perfusion, static tPO2 values present a variable response between organs and different shock states, as well as differences depending on the timing of sepsis evolution, and the use of concomitant supportive therapies (i.e. fluid resuscitation).27 This high variability has prompted the use of a dynamic challenge to unmask tissue hypoxia, the so-called oxygen challenge test. The test consists in assessing the ability of the circulation to transport high partial pressures of oxygen to the tissues, reflected as a downstream increase in tissue PO2 following a rise in PaO2.28 A poor rise in tPO2 would correspond to an insufficient blood flow. Therefore, adequacy of tissue perfusion may be explored evaluating the consistency of the response observed in all organs to a dynamic physiological challenge. Accordingly, the tPO2 response to an oxygen challenge test has been proposed as a reliable marker of adequacy of tissue perfusion. In high-output fluid-resuscitated septic patients, the presence of an altered tPO2 response to an oxygen challenge test may suggest local microcirculatory dysfunction. Indeed, the increase in transcutaneous tissue PO2 in response to an oxygen challenge test was associated with mortality and organ failure in a population of septic shock patients,29 and its inclusion in a resuscitation algorithm was associated with improved outcomes.30
Although tPO2 monitoring may be of potential interest, it still needs significant validation before it becomes a bedside tool for clinical management of septic patients.
Near-infrared spectroscopy (NIRS)The NIRS system consists of a light source, optical bundles (optodes) for light emission and reception, a processor, and a display system. NIRS technology is based on measuring the attenuation of light in the near-infrared spectrum (700–1000nm wavelengths) to measure chromophores, mainly hemoglobin, present in the sampled tissue. Minimizing the impact of other tissue chromophores that can influence NIRS signal is achieved by choosing specific lengths. Thus, the final signal is derived primary from oxy- and deoxyhemoglobin contained in the microvascular tree (vessels <100μm) present in the sampled area. Finally, tissue hemoglobin saturation or tissue oxygen saturation (StO2) is calculated from oxy- and deoxyhemoglobin measures.31,32
Skeletal muscle StO2 due to its non-vital peripheral condition has emerged as a potential early detector of occult hypoperfusion. The thenar eminence has been proposed as a reliable site for StO2 measurements, since it is the area less altered by local factors such as edema and fat tissue thickness.32 In healthy basal conditions, approximately 75% of the blood present in the skeletal muscle is located in the venous compartment, and therefore the NIRS signal reflects predominantly the venous oxygenation. StO2 reflects the balance between local oxygen supply and consumption, and thus, changes in StO2 might be interpreted in both directions: change in local microcirculatory flow and/or change in local consumption.33
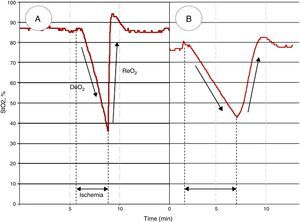
In addition to monitoring the absolute value, the StO2 response to a brief ischemic challenge has been explored in order to obtain tissue dynamic information.34 The so-called vascular occlusion test (VOT) consists in executing an arterial occlusion proximal to the StO2 probe, until a pre-defined ischemic threshold is reached, and after that the occlusion is then released. Thereby, dynamic StO2 parameters are generated: First, following ischemia an initial de-oxygenation slope (DeO2) is observed. This has been proposed as a marker of local oxygen extraction (and from which correcting the DeO2 for the estimated amount of hemoglobin derives a parameter of local oxygen consumption, expressed as nirVO2). Second, once the vascular occlusion is released, a re-oxygenation slope (ReO2) is observed. This has been proposed as a marker of endothelial function, as it depends on blood inflow and capillary recruitment after the hypoxic stimulus.34 Of note, a correlation between ReO2 with perfusion pressure was observed in several studies, suggesting that the resulting ReO2 seems to be derived from the interaction of perfusion pressure and endothelial integrity.35
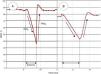
Septic patients have shown lower StO2 values than healthy subjects in the literature, but a huge overlap exists between these two populations.33 These observations may be explained by the heterogeneity of microcirculatory derangements in sepsis: the coexistence of ischemic and highly oxygenated areas, a phenomenon previously referred as microcirculatory shunting. This circumstance also explains why overall “normal” oxygen content may be observed in a given sensed area. Still, some authors have demonstrated that low absolute StO2 values after initial macrocirculatory resuscitation are associated with worse outcome, in terms of survival.36–38 However, VOT-derived variables have yielded much more promising performance when assessing prognostic (Fig. 3). Some authors have shown that local impairment in oxygen extraction, characterized as slower DeO2 slopes, is associated with worse evolution in organ dysfunction and increased mortality.39,40 Other authors have demonstrated that alterations in vascular reactivity, expressed as decreased ReO2 slopes, are also associated with lower survival rates from septic shock.40–43
Different StO2 patterns in response to a vascular occlusion test (VOT) in healthy subjects and septic shock. Figure A shows a normal StO2 response to a VOT, whereas figure B shows an altered response in a septic shock patient. Note that, independently of the baseline absolute StO2 value, the response to ischemia is different. The DeO2 slope is slower, and a longer period in order to reach a predetermined ischemic threshold (i.e. 40%)34 is needed. The vascular reactivity is also impaired, leading to a slower StO2 recovery rate (ReO2), and a blunted hyperemic response before returning to baseline StO2.
As we have already exposed, over the past 30 years, several studies have shown that microcirculatory alterations have prognostic implications in sepsis. Importantly, the association between microcirculatory alterations with organ failure development and mortality has been consistent regardless of the monitoring technology used. From the initial clinical studies using gastric tonometry, in the early 90s, to the latest direct microvasculatory visualization using in vivo videomicroscopy, the degree of alteration either in local oxygenation, local CO2 production, or capillary perfusion characteristics has been reliably associated to the evolution of septic patients.18–20,24,29,36–43 Of note, microcirculatory alterations have been associated to outcome even after restoring normal values of macrocirculatory variables, when the resuscitation process was considered to be accomplished, according to current international guidelines.23,44 On the whole, these observations have derived into a widespread idea regarding the need for including microcirculatory endpoints in the resuscitation process of septic patients. Still, the inclusion of microcirculatory variables as resuscitation endpoints appears quite complex, and seems to have a long way to go.
How can we manipulate the microcirculation?Despite our current ability to detect microcirculatory alterations in sepsis, most therapeutic interventions are still used for their impact on systemic hemodynamics, inferring that increasing global oxygen delivery will improve microvascular perfusion. Combining fluid administration, vasopressors and inotropic drugs is the cornerstone of the hemodynamic resuscitation process in sepsis. These therapies are delivered in order to increase global blood flow, finally aiming at achieving the normalization of global markers of tissue hypoxia, such as serum lactate and central venous oxygen saturation (ScvO2).2,44 However, given the heterogeneous nature of microcirculatory alterations in sepsis, increasing global organ blood flow might not be equal to recruiting the microcirculation. Recently, Ospina-Tascon and colleagues showed that the evolution over time of metabolic resuscitation endpoints was not related to changes in global hemodynamic variables, but to changes in microcirculatory perfusion parameters obtained using videomicroscopy.45 These observations would reinforce the idea that integrating microcirculatory variables will be required in order to redefine our resuscitation strategies, especially if we consider that the microcirculatory response might not be equal to the macrocirculatory.45–51
In addition to the effect of manipulating pressure and global flow, some other interventions might have a positive impact on microcirculatory perfusion, such as hydrocortisone, activated protein C, red blood cell transfusions, and perfusion of vasodilator agents, such as nitroglycerine.52–56 Almost each one of these therapies has been accompanied by controversial results, generating profound debates, and one might conclude that the need of randomized trials specifically selecting patients with microcirculatory alterations to better analyze the impact of any of these interventions is mandatory.
The benefits of targeting the microcirculation in sepsisFinally, although current efforts have been directed to increase our knowledge on how can we evaluate and manipulate the microcirculation, whether the ability to improve microcirculatory perfusion will have impact on the outcome of septic patients remains to be explored. To date, there is a lack of prospective trials targeting microcirculatory endpoints in the resuscitation process. In 1992, Gutierrez et al.24 reported significant benefits on survival when targeting gastric mucosal pH, assessed by gastric tonometry. The benefit of the intervention was limited to those patients presenting with a normal gastric pH, but was lacking when patients presented already with established gastric mucosa hypoperfusion. These results appear to reinforce the thesis of futility of resuscitation interventions once tissue or organ damage is already present, also postulated in large prospective trials using macrocirculatory endpoints.57,58 Later on, in 2007, after some observational studies,29 Yu and coworkers conducted a prospective interventional trial, comparing global resuscitation endpoints versus transcutaneous oxygen tension (PtO2) goals. Seventy patients were enrolled, and the PtO2-guided group showed a significant mortality reduction.30 Regrettably, the results of these trials have not been reproduced afterwards. More recently available technologies, such as videomicroscopy or NIRS, despite their exponential appearance in clinical studies, have not been included in prospective trials as resuscitation guiding tools in septic shock patients.
Even with its physiological sense, and the great amount of evidence supporting its prognostic relevance, the inclusion of microcirculatory variables in the resuscitation process of septic shock appears complex. Some authors have proposed to integrate microcirculatory endpoints at the end of the macrocirculatory resuscitation process, once current global endpoints are achieved.59 On the other hand, some other authors propose to “leave behind” global macrocirculatory endpoints, and guide resuscitation only using microcirculatory endpoints.60 To date, objective data supporting any of these two approaches is lacking, and the arguments are merely conjectural. Whether one strategy or the other offers better results will be part of future clinical research.
In the end, microcirculation-monitoring tools will face the same debate that has accompanied hemodynamic monitoring devices for the past decades: No monitoring device, per se, can improve outcome, unless coupled to a treatment that does. Current evidence is encouraging, but there is still a long way to go for microcirculation integration in daily hemodynamic resuscitation.
ConclusionsSeveral technologies allow the evaluation and monitoring of the microcirculation. Ranging from the oldest mucosal carbon dioxide or tissue oxygen tension, to the newest videomicroscopic systems, each one of these technologies has demonstrated its prognostic value in septic patients, independently of the macrocirculatory status. However, the incorporation of microcirculatory parameters in the clinical practice is still challenging. To date, sublingual videomicroscopy has emerged as the gold standard for microcirculatory evaluation, but its technical limitations need to be overcome for routine bedside application. Learning how to manipulate the microcirculation, and whether this manipulation results in better outcomes will require further research efforts.
Conflict of interestsDr. Gruartmoner and Dr. Mesquida have no conflict of interests to declare. Dr. Ince has received honoraria and independent research grants from Fresenius-Kabi, Bad Homburg, Germany; Baxter Health Care, Deerfield, Illinois and AM-Pharma, Bunnik, The Netherlands. Dr Ince has developed SDF imaging and is listed as inventor on related patents commercialized by MicroVision Medical (MVM) under a license from the Academic Medical Center (AMC). He has been a consultant for MVM in the past, but has not been involved with this company for more than five years now, except that he still holds shares. Braedius Medical, a company owned by a relative of dr Ince, has developed and designed a hand held microscope called CytoCam-IDF imaging. Dr Ince has no financial relation with Braedius Medical of any sort, i.e., never owned shares, or received consultancy or speaker fees from Braedius Medical.