We analyzed short, medium and long-term mortality in transplant recipients who received lungs from donors aged 55 years or more.
Patients and methodsAll patients who underwent lung transplantation from donors aged 55 years or more were included. The association between the different study variables and early death and death at 1 year and 5 years was studied. A logistic regression model was used to study the association between early death and variables with a trend towards significance (p<0.2) in the bivariate analysis. The risk factors for mortality at 1 year and 5 years were analyzed with a Cox regression model. The Kaplan–Meier method was used to analyze survival.
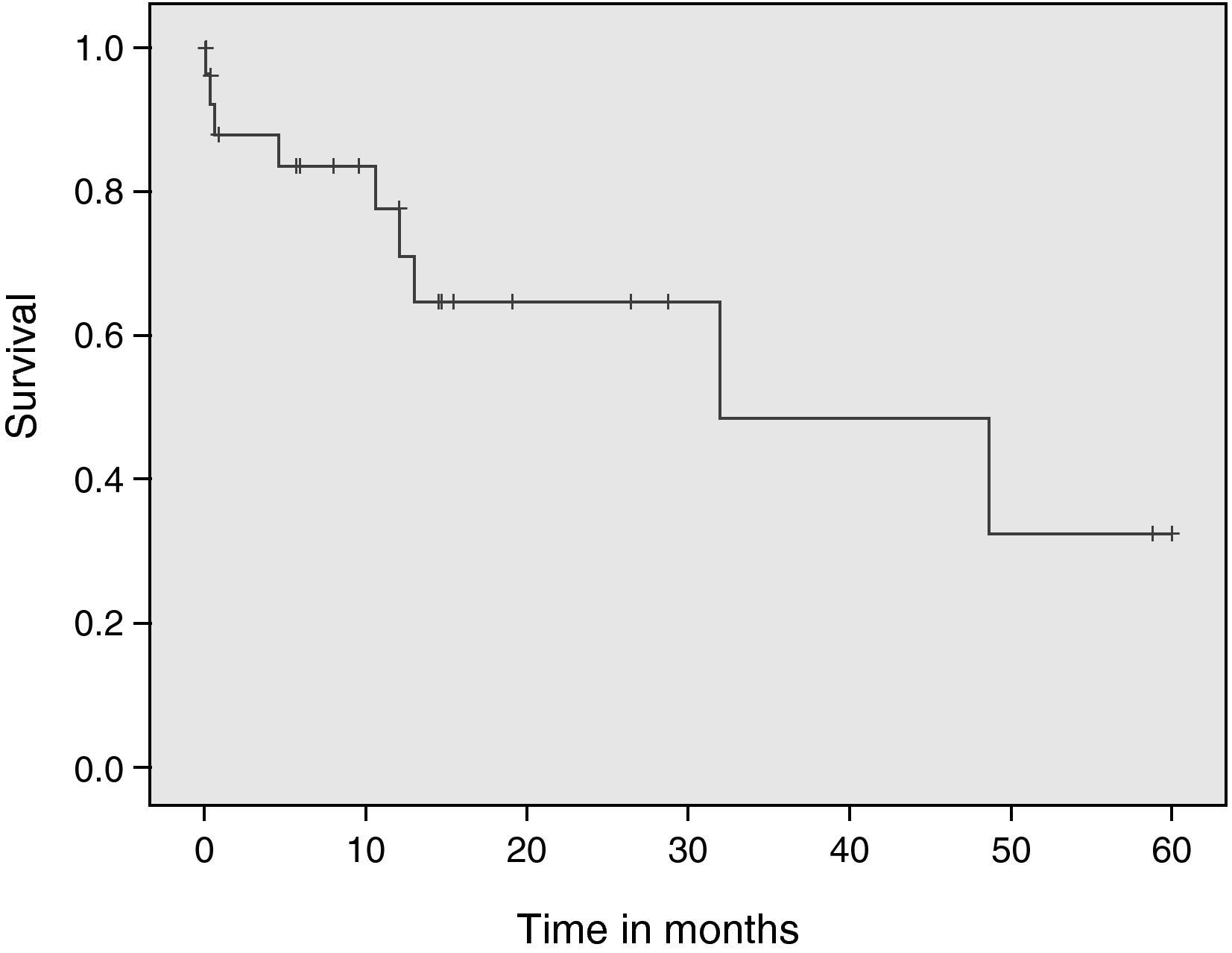
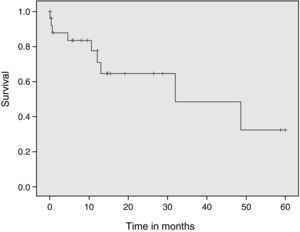
ResultsA total of 33 patients were included. The probability of survival was 90.9%, 78.5% and 44.8% at 1 month, 1 year, and 5 years after lung transplantation, respectively. The elevated age of the recipient (p=0.16) and single-lung transplantation (p=0.09) were the variables associated to or with a trend towards significant associations with mortality.
ConclusionsThe final decision to accept a lung graft should be based on individual evaluation of each donor and recipient. However, given the lack of lung donors, donors aged 55 years or more should be considered for lung transplantation.
Analizar la supervivencia a corto (un mes), medio (un año) y largo plazo (cinco años) en todos los pacientes sometidos a un trasplante pulmonar (TP) cuyo donante tuviera al menos 55 años.
Pacientes y métodosSe incluyó a todos los pacientes sometidos a un TP cuyo donante tuviera 55 años o más. Se analizó la asociación de las diferentes variables estudiadas con la mortalidad precoz, anual y al quinto año. Se utilizó la regresión logística en el estudio de factores de riesgo para mortalidad precoz y se utilizó la regresión de riesgos proporcionales de Cox en el estudio de factores de riesgo para la mortalidad al año y al quinto año, introduciendo las variables con un valor de p < 0,2 en el análisis bivariante. Se efectuó un análisis de supervivencia mediante el método de Kaplan-Meier.
ResultadosSe analizó un total de 33 pacientes sometidos a un TP con donantes de 55 años o más. La probabilidad de supervivencia fue del 90,9, el 78,5 y el 44,8% al mes, año y cinco años respectivamente. La edad elevada del receptor (p=0,16) y la realización de un trasplante unipulmonar (p=0,09) fueron las variables que se asociaron o mostraron tendencia a la asociación con la mortalidad.
ConclusionesLa decisión final en la aceptación de un injerto pulmonar se debe basar en la evaluación individual de cada donante y receptor. Sin embargo, dada la escasez de injertos pulmonares, parece adecuado considerar de inicio para la donación pulmonar a los pacientes de 55 o más años.
Lung transplantation (LT) is indicated in patients with progressive lung disease in which medication or alternative treatments have failed, and in those cases characterized by rapid deterioration. Unfortunately, the scarcity of organ donors is the main limitation in transplant programs. This lack of organs is greater in the case of lung transplantation, since this is the solid organ that deteriorates most rapidly after brain death. According to Eurotrasplant data, in 2009 lung grafts for transplantation were obtained from 513 of 2074 registered donors (24.7%) (http://www.eurotransplant.org/?id=peryear_public).1
In Spain, according to information from the National Transplant Organization (Organización Nacional de Trasplantes, ONT), a total of 1605 donors (34.3 donors per million inhabitants) were registered in 2009. However, lung grafts were obtained in only 228 cases (14.2%) for LT (http://www.ont.es).2
A series of alternatives have been proposed for expanding the pool of lung grafts available for transplantation, such as lung donation in asystolia,3 with very good results, or live donation.4 However, these strategies, while adequate, are unable to solve the shortage of lungs for transplantation. Brain death donation is the main source of lungs for transplantation in Spain.
The criteria defining an optimum lung donor are: age under 55 years; partial pressure of oxygen in arterial blood (PaO2)>300mmHg after maintaining the potential donor with an inspired oxygen fraction (FiO2) of 1 and a positive end-expiratory pressure (PEEP) of 5cm H2O during 5min; duration of mechanical ventilation under 5 days; absence of purulent secretions; and absence of radiological alterations contraindicating donation.5 However, most transplantation teams and groups have moved beyond these strict criteria, and use donors with expanded criteria (i.e., donors not meeting all the suitability criteria)—with good results as refers to recipient survival.6–8 In the case of the donor age criterion, lung donors aged 65 years or even older have been accepted, with good results in terms of recipient survival.9,10 Nevertheless, advanced donor age continues to be viewed negatively for lung donation purposes.
The present study analyzes survival over the short (1 month), middle (1 year) and long term (5 years) in all lung transplant recipients in our center, in which the donor age was at least 55 years.
Patients and methodsThe lung transplantation program of our center was started in March 1997. The present study considered all LT patients in which the donor was aged 55 years or older. The data were analyzed on a retrospective basis from the prospectively compiled LT database of the center. The information relating to the donors included age and gender, smoking history, cause of death, time on mechanical ventilation, 100% FiO2 oxygenation and PEEP 5cm H2O, and ischemia time of the lung grafts. Likewise, we recorded data on the recipient (age and gender, reason for transplantation, nutritional condition, body mass index), the surgical procedure (surgery time, use of extracorporeal circulation (ECC)) and stay in the Intensive Care Unit (ICU), PaO2/FiO2 after 24h, intubation time, use of renal replacement techniques, and complications in the ICU.
Recipients were followed-up on until death or until 30 April 2010. Two patients who died during lung graft implantation were excluded from the study. In both of these cases the operation could not be completed, since both patients died in the operating room before being moved to the ICU.
A descriptive analysis was made of the data–quantitative variables being reported as the mean and standard deviation, while qualitative variables were expressed as absolute and relative frequencies (percentages). A bivariate analysis was made to evaluate the association between the different variables and early mortality (occurring in the first month after transplantation), annual mortality, and mortality after 5 years. The Student's t-test and Mann–Whitney U-test were used with the quantitative variables, depending on the distribution of the parameters. The chi-squared test or Fisher exact test in turn were applied in the case of the categorical variables.
Cox proportional hazards regression analysis was used in the study of risk factors for mortality after 1 year and after 5 years, based on the backward stepwise method—introducing the variables with a value of p<0.2 in the bivariate analysis referred to mortality. The risk factors for early mortality were subjected to logistic regression analysis, introducing the variables with a value of p<0.2. The results were reported as the relative risk (RR) and corresponding 95% CI for Cox regression, and as the OR and 95% CI for logistic regression.
A survival analysis was carried out, considering death as an event, and survival curves were plotted based on the Kaplan–Meier method.
ResultsUp until 30 April 2010, a total of 252 LTs had been performed in our center. Of these patients, 35 (13.9%) received the lung graft from a donor aged 55 years or older. As has been commented above, two patients were excluded from the study. Most of the LTs involving such elderly donors were carried out in the last 5 years. Specifically, 15 of the 33 LTs were performed between 2009 and April 2010. The characteristics of the patient sample are described in Table 1. The indication of LT was emphysema (10 cases; 30.3%), lung fibrosis (8 cases; 24.2%), alpha-1-antitrypsin deficit (4 cases; 12.1%), and other causes (11 cases; 33.3%). No LT was performed due to cystic fibrosis. Both lungs were transplanted in 27 patients, and a single lung in 6 patients. A total of 9 subjects (27.3%) developed primary graft failure (PGF), which proved mild in four patients, moderate in two and severe in three. The median donor time on mechanical ventilation was 24h (interquartile range (IQR), 50).
Principal characteristics of the study series.
| Donor age | 57.39±2.38 (range, 55–64 years) |
| Donor gender (female) | 17 (51.5%) |
| Donor hours on MV | 24 [50] |
| Recipient age | 54.1±9.1 |
| Recipient gender (female) | 16 (48.4%) |
| Recipient BMI | 24.5±3.5 |
| Pre-transplantation albumin | 3.7±0.7 |
| Maximum ischemia time (min) | 325±84 |
| Surgery time (min) | 340±83 |
| Primary graft failure | 9 (27.3%) |
| Use of ECC | 3 (9.1%) |
| PaO2/FiO2 after 24h | 321±105 |
| Stay in ICU (days) | 5.5 [9] |
| Use of hemofiltration in ICU | 3 (9.1%) |
| Suture dehiscence | 1 (3%) |
ECC, extracorporeal circulation; BMI, body mass index; ICU, intensive care unit; MV, mechanical ventilation.
The data relating to the patients are expressed as the mean±standard deviation, n (%) or median [interquartile range].
In all cases triple immunosuppressive therapy was prescribed with a calcineurin inhibitor (tacrolimus in 26 cases and cyclosporine in 7), mycophenolate and corticosteroids. The fundamental cause of donor death was cerebral hemorrhage (26 cases; 78.8%). In all cases donor PaO2/FiO2 was >300mmHg (464±72mmHg; range: 357–600mmHg).
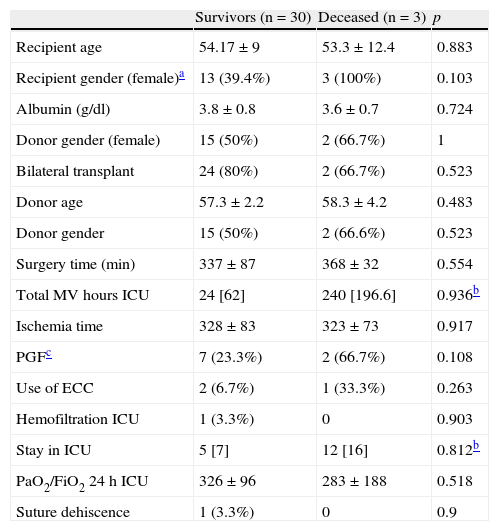
Early mortality (in the first month) was 9.09% (95% CI, 3.14–23.57 (3 patients)). In the bivariate analysis, recipient female gender and the development of PGF tended to be associated with increased early mortality (Table 2). In the logistic regression analysis, only PGF tended to be associated with increased mortality (Table 2). The probability of early survival (in the first month) was 90.9% in our series (95% CI, 0.8–1).
Mortality after 1 month.
| Survivors (n=30) | Deceased (n=3) | p | |
| Recipient age | 54.17±9 | 53.3±12.4 | 0.883 |
| Recipient gender (female)a | 13 (39.4%) | 3 (100%) | 0.103 |
| Albumin (g/dl) | 3.8±0.8 | 3.6±0.7 | 0.724 |
| Donor gender (female) | 15 (50%) | 2 (66.7%) | 1 |
| Bilateral transplant | 24 (80%) | 2 (66.7%) | 0.523 |
| Donor age | 57.3±2.2 | 58.3±4.2 | 0.483 |
| Donor gender | 15 (50%) | 2 (66.6%) | 0.523 |
| Surgery time (min) | 337±87 | 368±32 | 0.554 |
| Total MV hours ICU | 24 [62] | 240 [196.6] | 0.936b |
| Ischemia time | 328±83 | 323±73 | 0.917 |
| PGFc | 7 (23.3%) | 2 (66.7%) | 0.108 |
| Use of ECC | 2 (6.7%) | 1 (33.3%) | 0.263 |
| Hemofiltration ICU | 1 (3.3%) | 0 | 0.903 |
| Stay in ICU | 5 [7] | 12 [16] | 0.812b |
| PaO2/FiO2 24h ICU | 326±96 | 283±188 | 0.518 |
| Suture dehiscence | 1 (3.3%) | 0 | 0.9 |
ECC, extracorporeal circulation; FiO2, inspired oxygen fraction; PGF, primary graft failure; OR, odds ratio; PaO2, partial oxygen pressure; ICU, intensive care unit; MV, mechanical ventilation.
The data relating to the patients are expressed as the mean±standard deviation, n (%) or median [interquartile range].
Logistic regression: OR=23.5 (95% CI, 0.1–6.08); p=0.69.
Mann–Whitney U-test. Rest of quantitative variables analyzed according to Student's t-test.
Logistic regression: OR=11.2 (95% CI, 0.646–187); p=0.097.
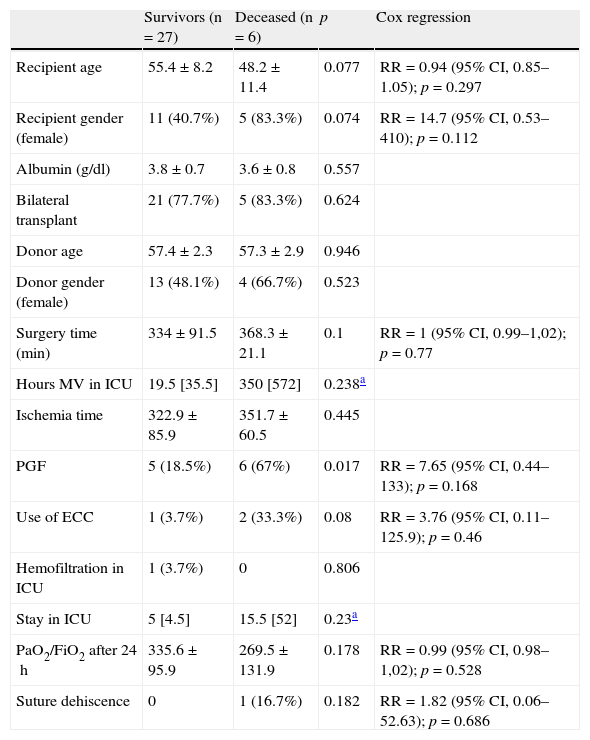
The mortality rate after 1 year was 18.18% (95% CI, 8.61–34.38) (6 patients, including those who died in the first month). The probability of middle term survival (1 year) was 78.5% (95% CI, 0.62–0.94). In the bivariate analysis, different demographic parameters (age and female gender in the recipient), surgical variables (prolonged surgery time and use of ECC), and different complications (PGF, low PaO2 after 24h and suture dehiscence) were correlated to or tended to be associated to increased mortality. However, in the Cox regression analysis, none of the variables was associated to increased mortality 1 year after transplantation (Table 3).
Mortality after 1 year.
| Survivors (n=27) | Deceased (n=6) | p | Cox regression | |
| Recipient age | 55.4±8.2 | 48.2±11.4 | 0.077 | RR=0.94 (95% CI, 0.85–1.05); p=0.297 |
| Recipient gender (female) | 11 (40.7%) | 5 (83.3%) | 0.074 | RR=14.7 (95% CI, 0.53–410); p=0.112 |
| Albumin (g/dl) | 3.8±0.7 | 3.6±0.8 | 0.557 | |
| Bilateral transplant | 21 (77.7%) | 5 (83.3%) | 0.624 | |
| Donor age | 57.4±2.3 | 57.3±2.9 | 0.946 | |
| Donor gender (female) | 13 (48.1%) | 4 (66.7%) | 0.523 | |
| Surgery time (min) | 334±91.5 | 368.3±21.1 | 0.1 | RR=1 (95% CI, 0.99–1,02); p=0.77 |
| Hours MV in ICU | 19.5 [35.5] | 350 [572] | 0.238a | |
| Ischemia time | 322.9±85.9 | 351.7±60.5 | 0.445 | |
| PGF | 5 (18.5%) | 6 (67%) | 0.017 | RR=7.65 (95% CI, 0.44–133); p=0.168 |
| Use of ECC | 1 (3.7%) | 2 (33.3%) | 0.08 | RR=3.76 (95% CI, 0.11–125.9); p=0.46 |
| Hemofiltration in ICU | 1 (3.7%) | 0 | 0.806 | |
| Stay in ICU | 5 [4.5] | 15.5 [52] | 0.23a | |
| PaO2/FiO2 after 24h | 335.6±95.9 | 269.5±131.9 | 0.178 | RR=0.99 (95% CI, 0.98–1,02); p=0.528 |
| Suture dehiscence | 0 | 1 (16.7%) | 0.182 | RR=1.82 (95% CI, 0.06–52.63); p=0.686 |
ECC, extracorporeal circulation; FiO2, inspired oxygen fraction; PGF, primary graft failure; CI, confidence interval; PaO2, partial oxygen pressure; RR, relative risk; ICU, intensive care unit; MV, mechanical ventilation.
The data relating to the patients are expressed as the mean±standard deviation, n (%) or median [interquartile range].
Mann–Whitney U-test. Rest of quantitative variables analyzed according to Student's t-test.
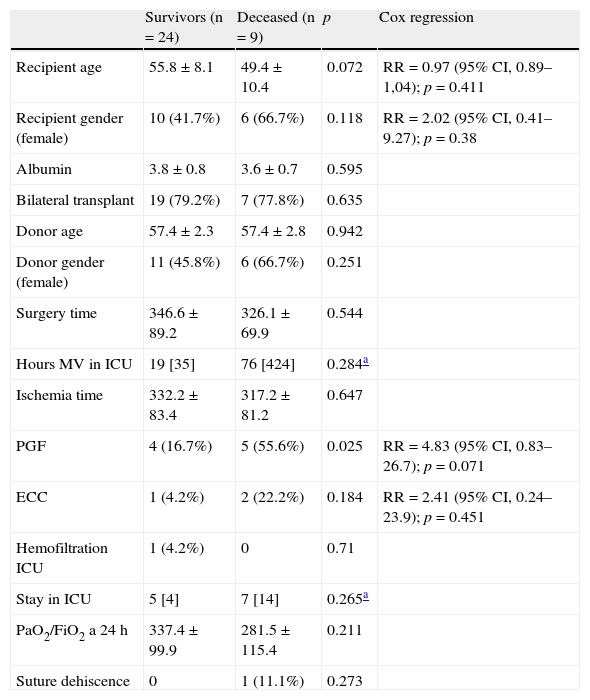
The mortality rate after 5 years was 27.27% (95% CI, 15.06–44.22) (9 patients, including those that died in the previously analyzed periods). In the bivariate analysis, PGF, age and the female gender in the recipient, as well as the use of ECC, were correlated to or tended to be associated to increased mortality. In the Cox regression analysis, only the development of PGF tended to be associated to increased patient mortality after 5 years (Table 4). The probability of long term survival (5 years) was 44.8% in our series (95% CI, 0.14–0.74).
Mortality after 5 years.
| Survivors (n=24) | Deceased (n=9) | p | Cox regression | |
| Recipient age | 55.8±8.1 | 49.4±10.4 | 0.072 | RR=0.97 (95% CI, 0.89–1,04); p=0.411 |
| Recipient gender (female) | 10 (41.7%) | 6 (66.7%) | 0.118 | RR=2.02 (95% CI, 0.41–9.27); p=0.38 |
| Albumin | 3.8±0.8 | 3.6±0.7 | 0.595 | |
| Bilateral transplant | 19 (79.2%) | 7 (77.8%) | 0.635 | |
| Donor age | 57.4±2.3 | 57.4±2.8 | 0.942 | |
| Donor gender (female) | 11 (45.8%) | 6 (66.7%) | 0.251 | |
| Surgery time | 346.6±89.2 | 326.1±69.9 | 0.544 | |
| Hours MV in ICU | 19 [35] | 76 [424] | 0.284a | |
| Ischemia time | 332.2±83.4 | 317.2±81.2 | 0.647 | |
| PGF | 4 (16.7%) | 5 (55.6%) | 0.025 | RR=4.83 (95% CI, 0.83–26.7); p=0.071 |
| ECC | 1 (4.2%) | 2 (22.2%) | 0.184 | RR=2.41 (95% CI, 0.24–23.9); p=0.451 |
| Hemofiltration ICU | 1 (4.2%) | 0 | 0.71 | |
| Stay in ICU | 5 [4] | 7 [14] | 0.265a | |
| PaO2/FiO2 a 24h | 337.4±99.9 | 281.5±115.4 | 0.211 | |
| Suture dehiscence | 0 | 1 (11.1%) | 0.273 |
ECC, extracorporeal circulation; FiO2, inspired oxygen fraction; PGF, primary graft failure; CI, confidence interval; PaO2, partial oxygen pressure; RR, relative risk; ICU, intensive care unit; MV, mechanical ventilation.
The data relating to the patients are expressed as the mean±standard deviation, n (%) or median [interquartile range].
Mann–Whitney U-test. Rest of quantitative variables analyzed according to Student's t-test.
The causes of death among the patients were sepsis and/or multiorgan failure (MOF) (n=3), bronchiolitis obliterans (n=3), primary graft failure (n=2), and cancer (n=1).
Cox regression analysis of the risk factors in the entire series showed advanced recipient age (RR=1.04 per unit time; 95% CI, 0.98–1.1; p=0.16) and the performance of single-lung transplantation (RR=2.68; 95% CI, 0.92–7.8; p=0.09) to be related (significantly or exhibiting a tendency) to mortality in the study cohort.
DiscussionIn recent years there has been a progressive liberalization or expansion of the LT donor selection criteria. In effect, LTs have been performed involving smoking donors, prolonged intubation periods, or donors with minor anomalies as evidenced by the chest X-rays.6,7 However, up until 5 years ago, very few operations had been performed involving donors over 55 years of age, and the published series all comprised very few cases.10,11 A review of large lung transplantation series found that the groups with the most extensive patient series often used donors with expanded criteria. The most frequently expanded criteria were smoking (over 20 packs/year) and the presence of radiological anomalies. In contrast, the use of donors over 55 years of age constituted an infrequent practice.12
The publications on LTs involving donors over 55 years of age are limited to small patient series. In this context, Pizanis et al., in a series of 19 patients, recently found that an elderly donor (≥55 years) does not affect long-term survival, even when compared with donors under age 55 years.10 Dezza et al. obtained similar results in a series of 10 transplant recipients from donors aged ≥55 years.11 The most surprising series was that published by the Toronto group.9 These authors reported good survival over the short (1 month) and middle term (1 year) with donors over 60 years of age (and up to 77 years of age). However, it was seen that very long term survival (10 years) could be affected by the use of elderly donors, due to an increased appearance of bronchiolitis obliterans. Nevertheless, it must be noted that this study was extreme in that 16.6% of the lung donors regarded as elderly were in fact 70 years old or older. There is very little information on survival beyond 5 years in lung graft recipients involving elderly donors (≥55 years). At the start of the decade it was considered that prolonged ischemia times in elderly lung grafts implied a notorious increase in recipient mortality.13 However, maintenance of the grafts from harvesting to reimplantation has clearly improved with the new preservation fluids, and ischemia time is no longer such a relevant factor.14
Despite the increase in the number of LTs performed, in Spain there were 164 patients on the waiting list on 31 December 2009 (www.ont.es). Therefore, despite the limited information available, and considering the scarcity of lung grafts and the growing waiting list for LT, it seems reasonable not to exclude elderly donors (≥55 years). The final decision regarding acceptance of a lung graft is clearly influenced by other variables, such as inspection of the graft at harvesting, and the condition and type of recipient for that particular organ graft.
The analysis of the survival data in our series (Fig. 1) shows excellent survival probability over the short (90.9% [95% CI, 0.80–1]), middle (78.5% [95% CI, 0.62–0.94]) and long term (44.8% [95% CI, 0.14–0.74]), with values not very different from those corresponding to the latest update of the International Society for Heart and Lung Transplantation (ISHLT), in which the survival rates of all registered patients, regardless of donor age, were 89% after three months, 79% after 1 year, and 52% after 5 years.
Nevertheless, comparisons with global series including all types of patients are not adequate, since they can be affected by biases due to the effects of subjective factors that influence LT survival. In this context, a lung graft from an elderly donor is usually not indicated in a young patient. These age differences among LT patients who receive an organ from a young or an elderly donor have already been commented in other series.9 An older age of the recipient has been associated to increased mortality in LT patients.15 Moreover, according to the ISHLT registry,16 a recipient age of over 50 years increases mortality over the short (1 month) and long term (5 years); in our series, 24 of the 33 recipients (72.7%) were over 50 years old. Although good survival rates have been obtained among recipients over 60 years of age, advanced LT recipient age does appear to be associated to increased mortality, even after adjusting for life expectancy according to age.17 In Spain, the mean age of LT recipients has gradually increased, reaching 46 years in 2009 (www.ont.es). In our study the mean recipient age exceeded 54 years, and as can be seen from the results, age was a risk factor with a tendency to be related to increased patient mortality. Another possible bias is the indication of LT when the donor is of marginal age (≥55 years). In effect, in our series there was a clear predominance of emphysema and idiopathic pulmonary fibrosis as indications of LT, and no patient with cystic fibrosis was included in the series–the latter being the indication offering the best survival rates over the short (1 month), middle (1 year) and long term (5 years).16
Unilateral LT has already been associated with increased mortality.16 In contrast to other studies, we found no association between certain variables such as the use of ECC in the operation, the use of renal replacement techniques in the ICU, prolonged stay in Intensive Care, etc., often associated to increased mortality. We believe that the explanation for this is the limited number of subjects included in our series.
Spain is the country with the highest organ donation rate. Despite this high rate, however, there is great disparity in the obtainment of lung grafts among the different regions or Autonomous Communities. The regional differences in lung donation rate are much greater than the differences in actual organ donor rates (www.ont.es). It is clear that the use of lung donors over 55 years of age allows for expansion of the number of available grafts. In this context, the contribution of the intensivist is crucial in order to consider this age group as a potential source of lung grafts from the outset, and to start the specific care required for lung donors,18 while ensuring the maintenance of optimum conditions with a view to avoiding the addition of further negative factors that lead to lung donation contraindication. Undoubtedly, improvement of the results will depend on the exquisite care of lung donors (particularly when dealing with expanded criteria donors). In this context, a consensus document has been developed by the ONT (www.ont.es) and different scientific societies, together with a recently published excellent review of the management of lung donors.18
A series of endocrine and hormonal alterations affect the lung tissue after brain death. Strict and careful management of lung donors allows for a marked increase in the number of grafts suitable for transplantation.18 Furthermore, strict hemodynamic and ventilatory control makes it possible to recover grafts initially not suited for LT.19,20 In our center, the use of lung donors aged 55 years or older has become generalized in the last 3 years. In effect, between January 2009 and April 2010 we performed 15 of the 33 LTs involving donors aged 55 years or older considered in the present study. Likewise, the number of lung grafts obtained for transplantation in our center has increased from 4.8 lung donors/year (mean between 2003 and 2008; www.ont.es) to 20 lung donors in the year 2009 – with a lung donation rate of over 50%.
The development and application of scales or scores can facilitate the work of deciding whether a kidney graft is suitable for transplantation or not.21,22 In the same way, some authors have proposed a score for helping to decide whether a marginal lung graft is apt for transplantation or not, and for establishing comparisons among different centers.23 However, its use is very limited and subject to debate; as a result, it cannot be recommended at the present time.
Our study has a number of limitations, fundamentally the limited size of the series, which prevents us from identifying variables that probably would be significant if the study sample were larger. Likewise, the confidence intervals obtained show great instability, with extreme or outlier values. Undoubtedly, future expansion of the number of cases, or shared data analysis in the context of a national LT registry, will allow us to draw firmer conclusions. Thus, the results of the present study must be viewed with caution. On the other hand, no assessment was made of the potential donors over 55 years of age that finally were excluded from donation. Nevertheless, in view of the good recipient survival results obtained, we feel it to be adequate to initially consider lung donation in donors aged 55 years or older.
Please cite this article as: Miñambres E, et al. Trasplante pulmonar con donantes de edad marginal (≥ 55 años). Med Intensiva. 2011;35:403–9.