In mechanical ventilation (MV), the configuration of ventilation parameters is key for pulmonary protection purposes. The term mechanical power is used to define the amount of energy transmitted from the ventilator to pulmonary parenchyma in each respiratory cycle.1 The most recent medical literature available confirms that in patients on MV due to SARS-CoV-2-induced respiratory failure, a MP threshold of 17J/min could be associated with a higher risk of death.2
In this context, our objective was to study the association of MP with short-term survival (28 days) of patients admitted to an intensive care unit (ICU) due to SARS-CoV-2. Therefore, using data from our registry of patients with COVID-19 developed after gaining approval from the local research ethics committee and obtaining consent from the patients or their legal representatives (written or over the phone) we conducted a retrospective analysis of all the cases admitted to our ICU from March 2020 through July 2021 who had been mechanically ventilated due to SARS-CoV-2-induced ARDS. Sample was consecutive and divided into 2 different cohorts based on the value of MP within the first 24h after endotracheal intubation: MP≤17J/min and MP>17J/min. MP was measured using the simplified formula proposed by Gattinoni et al.3
A descriptive analysis of the sample was initially conducted. Afterwards, a 28-day survival analysis was conducted with the Kaplan-Meier method for the variable of time of death (Log-rank test). To avoid confounding factors, a multivariable analysis of survival was conducted adjusting a Cox model (method: forward; introduction of variables to the model if P<.2, and exclusion of variables if P>.5; status: death at the ICU at 28 days; covariables used: age, the PaO2/FiO2 ratio prior to intubation, and the value of pulmonary compliance). Statistical significance was established at P<.05 for all analyses.
Out of the 565 patients admitted to the ICU with COVID-19 during the study period, only those with confirmed SARS-CoV-2 infections who received controlled MV were eventually analyzed. Also, the variables necessary to estimate MP in the supine position were estimated, after sedation and muscular paralysis, and within the first 24h after starting MV.
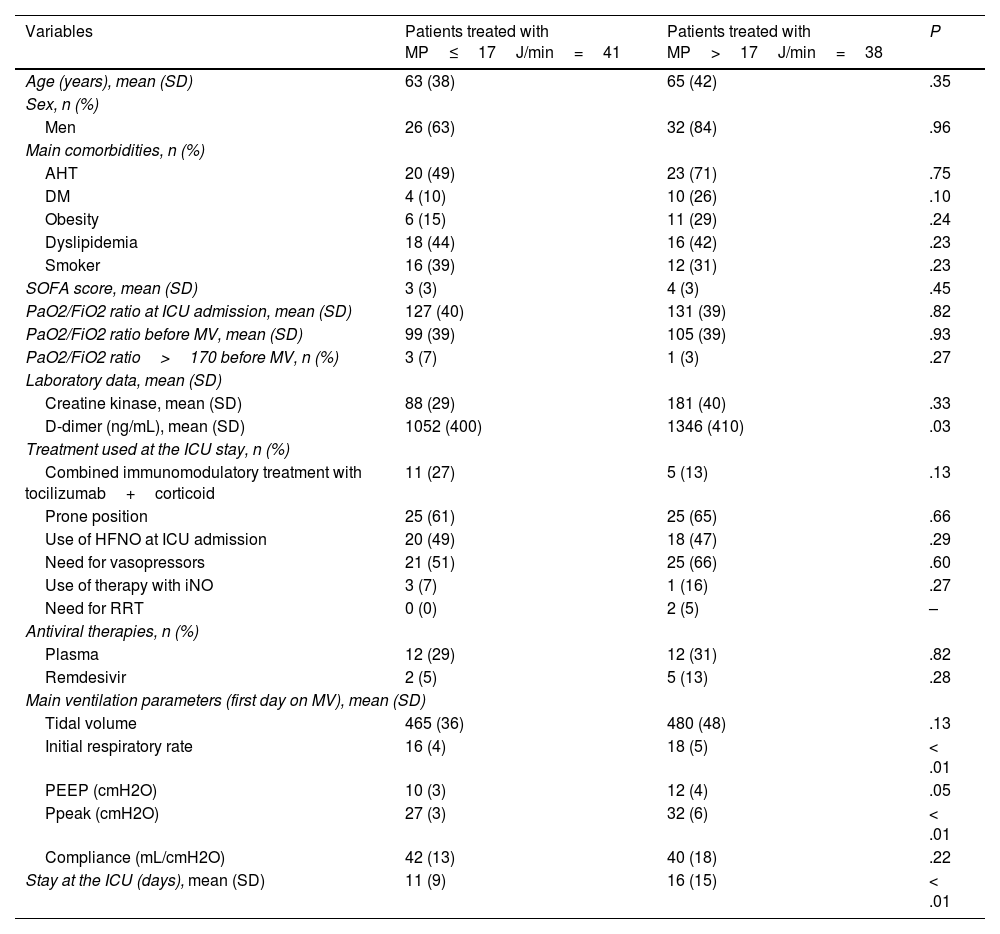
Table 1 shows the main differences among the 79 patients analyzed categorized based on their MP.
Description of the main differences of the variables between patients treated with MP≤17J/min and those treated with MP>17J/min.
| Variables | Patients treated with MP≤17J/min=41 | Patients treated with MP>17J/min=38 | P |
|---|---|---|---|
| Age (years), mean (SD) | 63 (38) | 65 (42) | .35 |
| Sex, n (%) | |||
| Men | 26 (63) | 32 (84) | .96 |
| Main comorbidities, n (%) | |||
| AHT | 20 (49) | 23 (71) | .75 |
| DM | 4 (10) | 10 (26) | .10 |
| Obesity | 6 (15) | 11 (29) | .24 |
| Dyslipidemia | 18 (44) | 16 (42) | .23 |
| Smoker | 16 (39) | 12 (31) | .23 |
| SOFA score, mean (SD) | 3 (3) | 4 (3) | .45 |
| PaO2/FiO2 ratio at ICU admission, mean (SD) | 127 (40) | 131 (39) | .82 |
| PaO2/FiO2 ratio before MV, mean (SD) | 99 (39) | 105 (39) | .93 |
| PaO2/FiO2 ratio>170 before MV, n (%) | 3 (7) | 1 (3) | .27 |
| Laboratory data, mean (SD) | |||
| Creatine kinase, mean (SD) | 88 (29) | 181 (40) | .33 |
| D-dimer (ng/mL), mean (SD) | 1052 (400) | 1346 (410) | .03 |
| Treatment used at the ICU stay, n (%) | |||
| Combined immunomodulatory treatment with tocilizumab+corticoid | 11 (27) | 5 (13) | .13 |
| Prone position | 25 (61) | 25 (65) | .66 |
| Use of HFNO at ICU admission | 20 (49) | 18 (47) | .29 |
| Need for vasopressors | 21 (51) | 25 (66) | .60 |
| Use of therapy with iNO | 3 (7) | 1 (16) | .27 |
| Need for RRT | 0 (0) | 2 (5) | – |
| Antiviral therapies, n (%) | |||
| Plasma | 12 (29) | 12 (31) | .82 |
| Remdesivir | 2 (5) | 5 (13) | .28 |
| Main ventilation parameters (first day on MV), mean (SD) | |||
| Tidal volume | 465 (36) | 480 (48) | .13 |
| Initial respiratory rate | 16 (4) | 18 (5) | < .01 |
| PEEP (cmH2O) | 10 (3) | 12 (4) | .05 |
| Ppeak (cmH2O) | 27 (3) | 32 (6) | < .01 |
| Compliance (mL/cmH2O) | 42 (13) | 40 (18) | .22 |
| Stay at the ICU (days), mean (SD) | 11 (9) | 16 (15) | < .01 |
AHT, arterial hypertension; DM, diabetes mellitus; HFNO, high-flow nasal oxygen; ICU, intensive care unit; iNO, inhaled nitric oxide; MP, mechanical power; MV, mechanical ventilation; PEEP, positive end-expiratory pressure; RRT, renal replacement therapy; SD, standard deviation; SOFA, Sequential Organ Failure Assessment score.
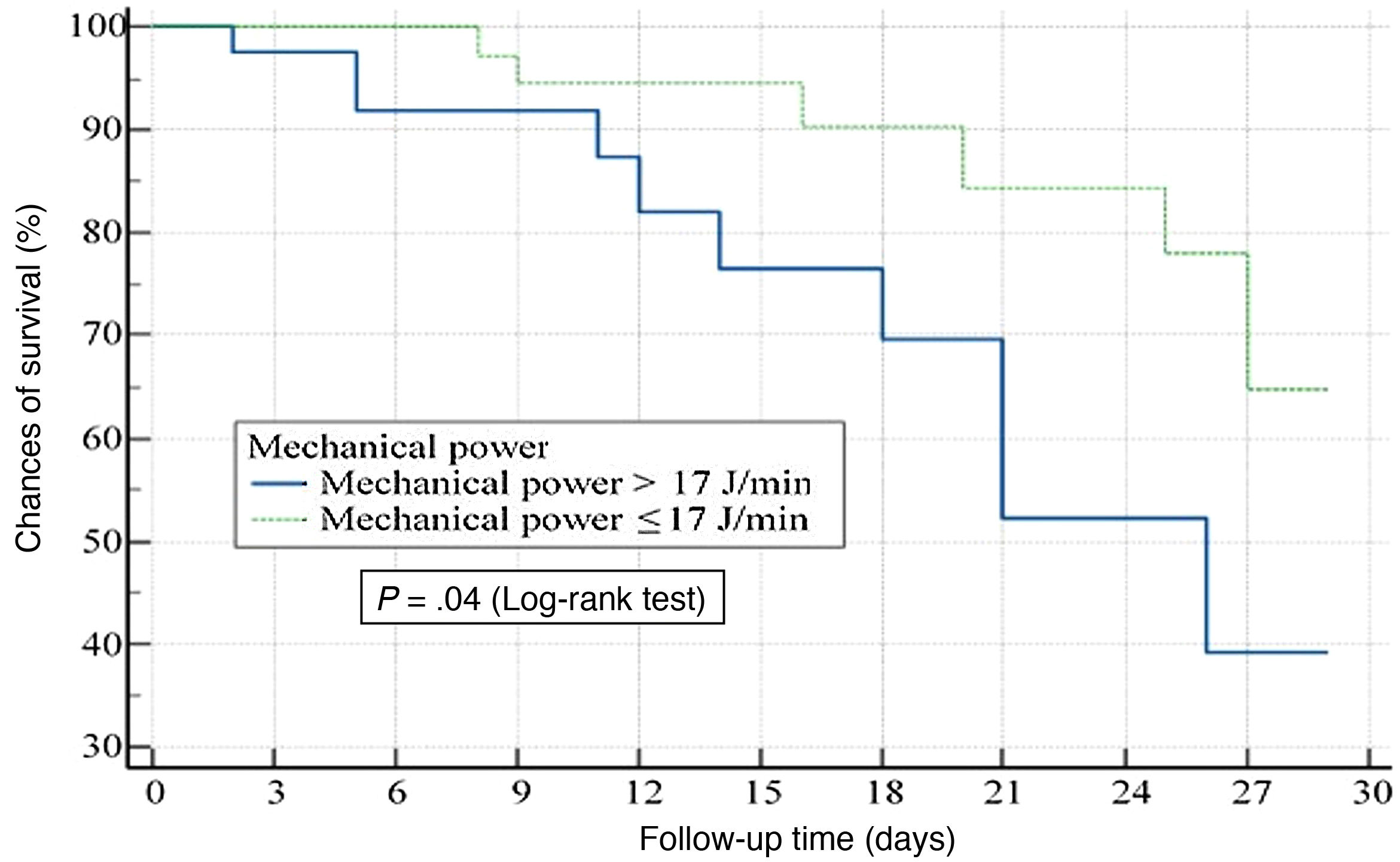
The median survival times of both cohorts were 16 days (p25–75: 3–27) for the cohort of patients with MP≤17J/min vs 11 days (p25–75: 2–18) for the cohort of patients with MP>17J/min (P=.02). The group of patients with MP values>17J/min was associated significantly with greater chances of death at 28 days (OR, 2.91; 95%CI, 1.04–8.09; P=.04) (Fig. 1).
In the Cox regression analysis, values>17J/min of MP within the first 24h after starting MV were independently associated with mortality (HR, 2.70; 95%CI, 1.31–6.47; P=.02).
Our results are consistent with recent studies that confirmed the adverse events of exposure to higher MP values in critically ill patients treated with MV due to SARS-CoV-2-induced respiratory failure or for a completely different reason.2,4
These findings can be considered expected outcomes if we understand MP as a variable that includes all components traditionally associated with the production of VILI: pressures, volume, flow, respiratory rate. Also, we should mention that rheology theory foresees that energy densities exceeding the resilience of a material would be responsible for the production of VILI. This energy density (energy per unit of surface) invites us, in our routine clinical practice, to assess at all time the association between MP and the alveolar area exposed to the energy supplied.5,6
On the other hand, the appearance of lung alterations (stress raisers)—that can eventually trigger VILI due to the application of MP—seems like a threshold effect phenomenon. In animals used in experiments (Young module or specific lung elastance,7 5.4 cmH2O±2.2 cmH2O) a threshold of 12J/min has been established as an energy power threshold.1 In humans8 (Young module, 13.4 cmH2O±4.1 cmH2O), recent clinical studies confirm that MP levels >18−20J/min are associated with a higher risk of death in patients on MV.9,10
Recognizing MP as a combination of parameters that can predispose to VILI is an important step towards the optimization of MV in critically ill patients.