To define clinical features associated with Intensive Care Unit (ICU) infections caused by multi-drug resistant organisms (MDRO) and their impact on patient outcome.
DesignA single-center, retrospective case–control study was carried out between January 2010 and May 2010.
SettingA medical ICU (MICU) in the United States.
PatientsThe study included a total of 127 MDRO-positive patients and 186 MDRO-negative patients.
InterventionsNo interventions were carried out.
ResultsOut of a total of 313 patients, MDROs were present in 127 (41.7%). Based on the multivariate analysis, only infection as a cause of admission [OR 3.3 (1.9–5.8)]), total days of ventilation [OR 1.07 (1.01–1.12)], total days in hospital [OR 1.04 (1.01–1.07)], immunosuppression [OR 2.04 (1.2–3.5)], a history of hyperlipidemia [OR 2.2 (1.2–3.8)], surgical history [OR 1.82 (1.05–3.14)] and age [OR 1.02 (1.00–1.04)] were identified as clinical factors independently associated to MDROs, while the Caucasian race was negatively associated to MDROs. The distribution of days on ventilation, days in hospital and days of antibiotic treatment prior to infection differed between the MDRO-positive and MDRO-negative groups. The MDRO-positive patients showed a greater median number of days in hospital and days of antibiotic treatment before infection, with a greater median number of days in hospital, days of antibiotic treatment and days of ventilation after infection, compared to the MDRO-negative patients. The mortality rate was not significantly different between the two groups. Appropriate empirical antibiotic therapy was prescribed in 82% of the MDRO-positive cases – such treatment being started within 24h after onset of the infection in 68.5% of the cases.
ConclusionDefining clinical factors associated with MDRO infections and administering timely and appropriate empirical antibiotic therapy may help reduce the mortality associated with these infections. In our hospital we did not withhold broad spectrum drugs as empirical therapy in patients with clinical features associated to MDRO infection. Our rate of appropriate empirical therapy was therefore high, which could explain the absence of excessive mortality in patients infected with MDROs.
Definir las características clínicas asociadas a las infecciones en la unidad de cuidados intensivos causadas por organismos multirresistentes (OMR) y el impacto asociado en los resultados del paciente.
DiseñoSe llevó a cabo un estudio de casos y controles, retrospectivo y unicéntrico entre enero de 2010 y mayo de 2010.
ÁmbitoUnidad de cuidados intensivos médica en Estados Unidos.
PacientesSe incluyó en el estudio a un total de 127 pacientes con infección positiva para OMR y a 186 pacientes con infección negativa para OMR.
IntervencionesNo se ha llevado a cabo ninguna intervención.
ResultadosDe un total de 313 pacientes se observaron OMR en 127 (41,7%). En un análisis multivariable únicamente se identificaron la infección como causa del ingreso (OR: 3,3 [1,9-5,8]), el total de días con ventilación (OR 1,07 [1,01-1,12]), el total de días de hospitalización (OR 1,04 [1,01-1,07]), la inmunosupresión (OR 2,04 [1,2-3,5]), los antecedentes de hiperlipidemia (OR 2,2 [1,2-3,8]), los antecedentes quirúrgicos (OR 1,82 [1,05-3,14]) y la edad (OR 1,02 [1,002-1,04]) como factores clínicos asociados de manera independiente con los OMR, mientras que dicha asociación fue negativa en el caso de la raza blanca. La distribución de los días de ventilación, los días de ingreso hospitalario y los días de tratamiento con antibióticos antes de la infección fueron diferentes entre los grupos positivo para OMR y negativo para OMR. El grupo de pacientes positivos para OMR presentó una mayor mediana del número de días de hospitalización y de tratamiento con antibióticos antes de la infección, con una mayor mediana del número de días de hospitalización, de tratamiento con antibióticos y de ventilación tras la infección frente a los pacientes del grupo negativo para OMR. La diferencia en la tasa de mortalidad entre ambos grupos no fue estadísticamente significativa. Se prescribió un tratamiento empírico adecuado en el 82% de los casos positivos para OMR, un tratamiento que se inició en el plazo de las 24horas siguientes a la manifestación de la infección en el 68,5% de los casos.
ConclusiónLa definición de los factores clínicos asociados a las infecciones por OMR y la administración de un tratamiento antibiótico empírico adecuado y de manera oportuna puede ayudar a reducir la mortalidad asociada a estas infecciones. En nuestro hospital no restringimos los fármacos de amplio espectro como tratamiento empírico en pacientes con características clínicas asociadas con la infección por OMR. Por este motivo, nuestra tasa de administración de un tratamiento empírico adecuado ha sido elevada, lo que podría explicar la ausencia de una mortalidad excesiva elevada en el caso de los pacientes infectados con OMR.
Infections remain one of the most serious concerns in the critical care setting, where multidrug resistant organism (MDRO) outbreaks can jeopardize the chances for effective therapy. An organism is considered multi-drug resistant when in vitro drug – susceptibility testing shows resistance to one or more classes of antimicrobial agents recommended as first line therapy.1 Multidrug resistance has been demonstrated for a variety of organisms that are more common in ICUs than in other hospital wards, and the risk of infection increases with duration of hospitalization.2–4 The primary goal of this study was to compare the epidemiological and the clinical characteristics, of patients with and without MDRO in a medical ICU (MICU). The secondary goal is to determine the accuracy of the empiric antibiotic selection and the outcomes associated with MDRO infection. We also evaluated the timing of appropriate empiric therapy, and its impact on outcomes.
Prior studies have shown that critically ill patients harboring MDROs have an increased mortality,5–7 primarily because resistance is often associated with inappropriate empiric therapy. However, other reports8,9 have suggested that it is unclear whether using appropriate empiric antimicrobial therapy improves mortality in these patients. When MDROs are common, empiric antibiotic selection for at risk patients should be based on known susceptibility patterns of likely microorganisms, while reliance on culture data cannot correct initial errors of empiric therapy. Thus, defining clinical factors associated with MDRO infection can potentially lead to less inappropriate empiric therapy and improved outcomes.
Patients and methodsStudy design and settingThis retrospective case–control study was conducted in the medical intensive care unit (MICU) at Winthrop University Hospital, a 600-bed tertiary care university hospital, between January 1, 2010 and May 31, 2010. Only active and symptomatic infections, receiving antimicrobial treatment, were included. MDRO positive patients had organisms resistant to one or more classes of antimicrobial agents recommended as first line therapy and were identified from: blood, sputum, urine or tissue cultures, collected at the time of suspected infection. Included organisms were: Acinetobacter baumannii, Escherichia coli, Pseudomonas aeruginosa, Klebsiella pneumoniae, Proteus mirabilis, Morganella morganii and Serratia spp.
Other organisms identified, were vancomycin-resistant enterococcus spp. (VRE) and methicillin-resistant Staphylococcus aureus (MRSA) bacteria. Susceptibilities to all antimicrobial agents were determined and interpreted according to criteria of the Clinical and Laboratory Standards Institute (CLSI) by disk diffusion susceptibility.10 Intermediate susceptibility was considered as resistance, based on CLSI standards10 and because this approach has been used in other MDRO studies in the ICU.11–13 Only MDROs isolated more than 48h after MICU admission and within 15 days after MICU discharge were considered, and duplicates were excluded. All patients had cultures obtained at the time of suspected infection, but if cultures were negative, they were considered MDRO negative.
Controls were patients with infection requiring antimicrobial therapy, but with no MDROs in any type of cultures. We included culture negative patients in the control group because culture-negative sepsis is common in the United States, with 49% of hospitalized patients with sepsis being culture-negative.14 Patients could be included more than once, if there were different admissions for each infection. The study was approved by the Institutional Review Board of the Winthrop University Hospital. Patient consent was waived because data were collected and analyzed anonymously and retrospectively from patient records. All research data obtained were de-identified, handled, stored and shared with confidentiality.
Demographic dataPatient characteristics including, age, gender, race, body mass index, past medical and/or surgical history, comorbid illness and clinical course (total length of stay, and MICU length of stay) were recorded. Antimicrobial drug exposures were assessed during hospital admission and until discharge. Duration (days of use) of antimicrobial exposure, central venous catheter, and bladder catheter were also analyzed. Days of antibiotics, ventilation and hospitalization were further separated, into days prior and after the diagnosis of infection. The recorded outcomes were: mortality, discharge to home, or to a nursing home/long term facility.
DefinitionsThe CDC definitions were used for infections at different anatomic sites.15 The definition of MDRO used was adopted from the CDC recommendations for the management of MDR organisms in healthcare settings that defines MDR organisms as bacteria that are resistant to one or more classes of antimicrobial agents recommended as first line therapy.1
Vancomycin-resistant Enterococcus spp. (VRE) was defined as the Enterococcus faecium/facaelis species that were resistant to vancomycin by standard susceptibility testing methods or by results from any FDA-approved test for VRE detection from specific specimen sources. Methicillin-resistant Staphylococcus aureus (MRSA) was defined as Staphylococcus aureus cultured from any specimen that tested oxacillin-resistant, cefoxitin-resistant, or methicillin-resistant by standard susceptibility testing methods.
Antimicrobial classes were: penicillins (oxacillin), third-generation cephalosporins (ceftazidime, cefotaxime, ceftriaxone), fourth-generation cephalosporins (cefepime), carbapenems (meropenem, etrapenem), anti-pseudomonal penicillins (piperacillin-tazobactam), aminoglycosides (amikacin, gentamicin), monobactams (aztreonam), glycopeptide (vancomycin) quinolones (levofloxacin, moxifloxacin), and glycylcyclines (tigecycline). Susceptibility to colistin was not considered.
The distinction between colonization and infection when the site of infection was the urinary tract or the lungs, was made by evaluating clinical criteria such as the presence of fever and the adequacy of organ perfusion. All included patients and controls met criteria for infection.
Immunosuppression was defined, as active solid or hematologic malignancy, leukopenia (absolute neutrophil count <1500cells/μl blood), chronic immunosuppressive treatment, prior radiation and use of corticosteroids at a dose of least 10mg/d for 15 days.
Prior hospitalization, prior antimicrobial use and prior surgical history were defined as a hospital stay, antimicrobial administration and undergoing major surgery respectively, within 3 months before the index hospitalization.
Liver dysfunction was considered present in any patient with of a bilirubin concentration over 2.0mg/dl or with liver cirrhosis, while renal insufficiency was defined in a patient with a creatinine level above 2.0mg/dl or requirement for dialysis. Cardiovascular failure was defined when inotropic drugs were required. Respiratory dysfunction was defined as inadequate gas exchange requiring endotracheal intubation and mechanical ventilation.
Previous history of hyperlipidemia was defined in a patient who was diagnosed with an abnormal level of lipids in the blood and was currently being treated. Previous history of hypothyrodism was defined in a patient who was diagnosed with hypothyroidism and was receiving therapy.
Initial empiric antibiotic treatment was defined as appropriate if the antibiotic prescribed within 24h of obtaining cultures matched the in vitro susceptibility of the presumed etiologic pathogen, in a patient with a positive culture at the time of infection. The doses used were in accordance with current standards. For example, in patients with normal renal function, we prescribed ertapenem 1g q24h, meropenem 1g q8h, ceftriaxone 2g q24h, cefotaxime 1g q8h, ceftazidime 1g q8h, cefepime 1g q6h, aztreonam 2g q8h, vancomycin loading dose 25–30mg/kg, followed by 15mg/kg q12h, linezolid 600mg q12h, levofloxacin 750mg q24h, piperacillin-tazobactam 4.5g q8h, gentamicin 7mg/kg, daptomycin 6–8mg/kg IV q24h, tigecycline 100mg IV loading dose, followed by 50mg q12h. By 2010, the CLSI approved new breakpoints for meropenem, imipenem, and doripenem that define susceptibility as a MIC of ≤1μg/ml and resistance as ≥4μg/ml.16
Statistical analysisContinuous variables were tested for normality using histograms and Kolmogorov–Smirnov test. If the data were markedly non-normally distributed, they are presented as medians (interquartile range) and categorical data are presented as percentages. Fisher's exact test was used to compare MDRO (positive vs. negative) for binary variables and Wilcoxon rank-sum test for continuous variables. We used all variables that were significant in univariate logistic regression at p<0.05 in the multiple logistic regression model, with a stepwise selection method to find independent predictors of MDRO. Risk was assessed using odds ratios. All calculations were performed utilizing SAS 9.2 (SAS Institute, Cary, NC) for Windows and results were considered statistically significant when p<0.05.
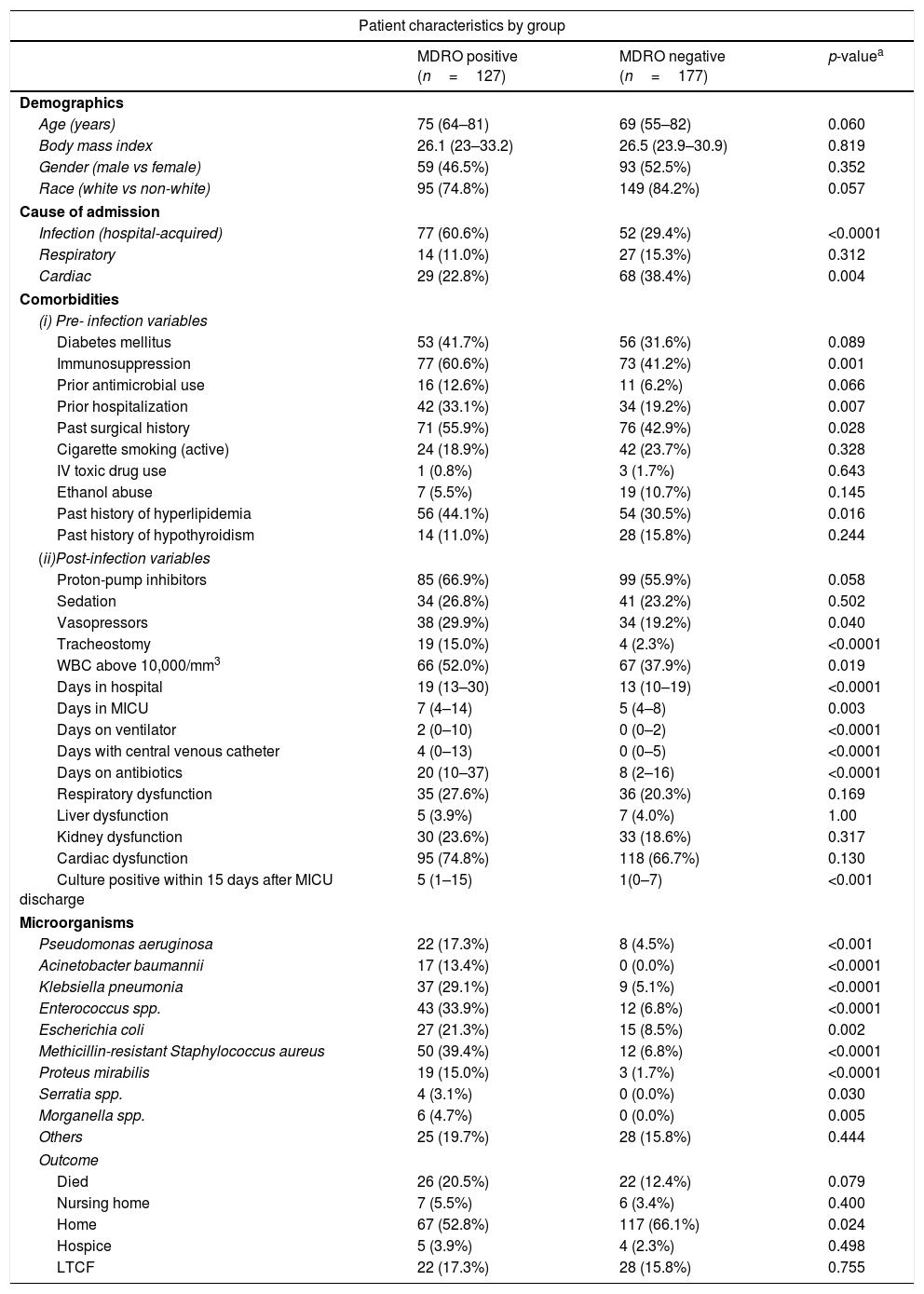
ResultsPatient characteristicsThree hundred and thirteen (313) patients were included in this study. Demographic and clinical characteristics comparing cases and controls are shown in Table 1. Although Coagulase-negative Staphylococci (CoNS) can cause true bacteraemia, repeated blood cultures (from a total of 9 subjects) did not isolate CoNS with either the same antibiogram or isolated another pathogen. Therefore these CoNs isolates were considered as a contaminant and not a pathogen. MDRO were found in 127 patients (41.7%), while 177 (58.2%) did not have MDRO (control population) present in cultures. Of the 177 control patients, a non-MDRO pathogen was present in 65 and cultures were negative in 112. Among the MDRO positive and negative patients, infection may have been incubating at the time of hospital admission in 21 (16%) and in 9 patients (12%) respectively, although all infections were diagnosed after 48h in the ICU, and thus met the criteria for nosocomial infection. Interestingly all these patients had healthcare exposure risk factors [history of prior hospitalization and one or two comorbidities (COPD, diabetes)] and thus were similar to HAIs in terms of various comorbid conditions, source of infection, pathogens and mortality rate and therefore we considered them as HAIs.
Demographic and clinical characteristics comparisons for case and controls patients. Pre-infection and post-infection variables are also shown.
| Patient characteristics by group | |||
|---|---|---|---|
| MDRO positive (n=127) | MDRO negative (n=177) | p-valuea | |
| Demographics | |||
| Age (years) | 75 (64–81) | 69 (55–82) | 0.060 |
| Body mass index | 26.1 (23–33.2) | 26.5 (23.9–30.9) | 0.819 |
| Gender (male vs female) | 59 (46.5%) | 93 (52.5%) | 0.352 |
| Race (white vs non-white) | 95 (74.8%) | 149 (84.2%) | 0.057 |
| Cause of admission | |||
| Infection (hospital-acquired) | 77 (60.6%) | 52 (29.4%) | <0.0001 |
| Respiratory | 14 (11.0%) | 27 (15.3%) | 0.312 |
| Cardiac | 29 (22.8%) | 68 (38.4%) | 0.004 |
| Comorbidities | |||
| (i) Pre- infection variables | |||
| Diabetes mellitus | 53 (41.7%) | 56 (31.6%) | 0.089 |
| Immunosuppression | 77 (60.6%) | 73 (41.2%) | 0.001 |
| Prior antimicrobial use | 16 (12.6%) | 11 (6.2%) | 0.066 |
| Prior hospitalization | 42 (33.1%) | 34 (19.2%) | 0.007 |
| Past surgical history | 71 (55.9%) | 76 (42.9%) | 0.028 |
| Cigarette smoking (active) | 24 (18.9%) | 42 (23.7%) | 0.328 |
| IV toxic drug use | 1 (0.8%) | 3 (1.7%) | 0.643 |
| Ethanol abuse | 7 (5.5%) | 19 (10.7%) | 0.145 |
| Past history of hyperlipidemia | 56 (44.1%) | 54 (30.5%) | 0.016 |
| Past history of hypothyroidism | 14 (11.0%) | 28 (15.8%) | 0.244 |
| (ii)Post-infection variables | |||
| Proton-pump inhibitors | 85 (66.9%) | 99 (55.9%) | 0.058 |
| Sedation | 34 (26.8%) | 41 (23.2%) | 0.502 |
| Vasopressors | 38 (29.9%) | 34 (19.2%) | 0.040 |
| Tracheostomy | 19 (15.0%) | 4 (2.3%) | <0.0001 |
| WBC above 10,000/mm3 | 66 (52.0%) | 67 (37.9%) | 0.019 |
| Days in hospital | 19 (13–30) | 13 (10–19) | <0.0001 |
| Days in MICU | 7 (4–14) | 5 (4–8) | 0.003 |
| Days on ventilator | 2 (0–10) | 0 (0–2) | <0.0001 |
| Days with central venous catheter | 4 (0–13) | 0 (0–5) | <0.0001 |
| Days on antibiotics | 20 (10–37) | 8 (2–16) | <0.0001 |
| Respiratory dysfunction | 35 (27.6%) | 36 (20.3%) | 0.169 |
| Liver dysfunction | 5 (3.9%) | 7 (4.0%) | 1.00 |
| Kidney dysfunction | 30 (23.6%) | 33 (18.6%) | 0.317 |
| Cardiac dysfunction | 95 (74.8%) | 118 (66.7%) | 0.130 |
| Culture positive within 15 days after MICU discharge | 5 (1–15) | 1(0–7) | <0.001 |
| Microorganisms | |||
| Pseudomonas aeruginosa | 22 (17.3%) | 8 (4.5%) | <0.001 |
| Acinetobacter baumannii | 17 (13.4%) | 0 (0.0%) | <0.0001 |
| Klebsiella pneumonia | 37 (29.1%) | 9 (5.1%) | <0.0001 |
| Enterococcus spp. | 43 (33.9%) | 12 (6.8%) | <0.0001 |
| Escherichia coli | 27 (21.3%) | 15 (8.5%) | 0.002 |
| Methicillin-resistant Staphylococcus aureus | 50 (39.4%) | 12 (6.8%) | <0.0001 |
| Proteus mirabilis | 19 (15.0%) | 3 (1.7%) | <0.0001 |
| Serratia spp. | 4 (3.1%) | 0 (0.0%) | 0.030 |
| Morganella spp. | 6 (4.7%) | 0 (0.0%) | 0.005 |
| Others | 25 (19.7%) | 28 (15.8%) | 0.444 |
| Outcome | |||
| Died | 26 (20.5%) | 22 (12.4%) | 0.079 |
| Nursing home | 7 (5.5%) | 6 (3.4%) | 0.400 |
| Home | 67 (52.8%) | 117 (66.1%) | 0.024 |
| Hospice | 5 (3.9%) | 4 (2.3%) | 0.498 |
| LTCF | 22 (17.3%) | 28 (15.8%) | 0.755 |
Continuous variables are presented as median (1srt quartile–3rd quartile) as they were not normally distributed and categorical variables are presented as n (%).
The MDRO patients had a median age of 75 years vs. 69 years in controls (p=0.06). There were no statistically significant differences between the two groups with respect to gender and body mass index. The comorbidities were similar, except that more MDRO positive patients had immunosuppression (60.0% vs 41.2%, p=0.001), prior antimicrobial use (12.6% vs 6.2%, p=0.06) and a history of prior hospitalization (33.1% vs 19.2%, p=0.007).
Among the 127 patients with MDRO infection, 38 (29.9%) had septic shock,. Septic shock was present in 35 (19.7%) of the 177 patients with non-MDRO infection. All other patients had active infection with sepsis. Of the 127 MDRO positive patients, previous colonization with the same MDRO was not identified, although routine surveillance cultures were not collected.
In those with MDRO infection, the site of infection was: the urinary tract in 55%, the lung in 35% and other sites (abdomen, skin and soft tissues) in 10%. For those with non-MDRO infection, the site of infection was: the urinary tract in 32%, the lung in 38%, and other sites in 30%.
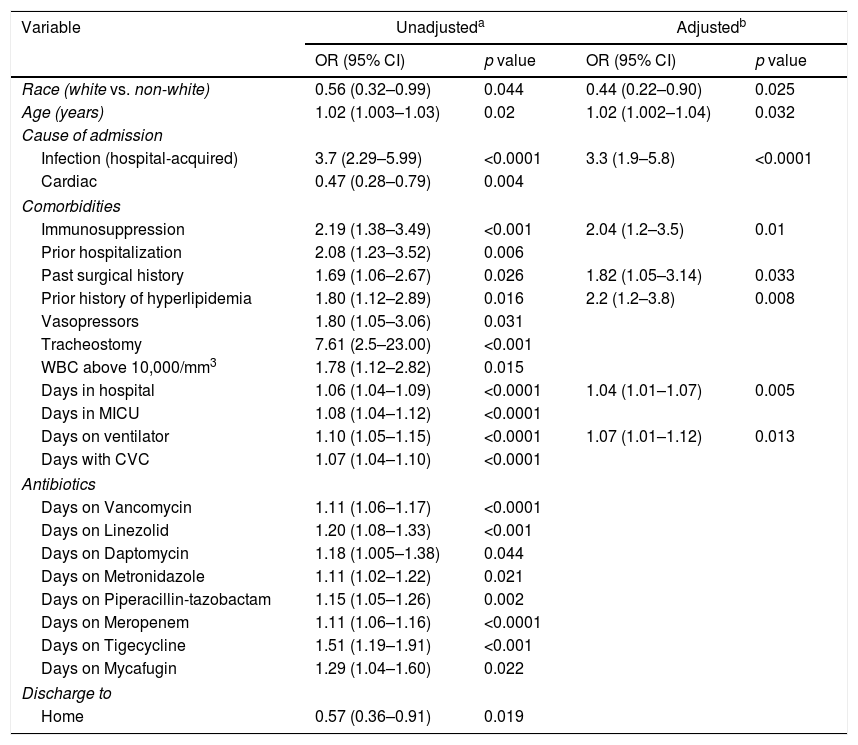
The identity and frequency of the pathogens are shown in Table 1, with methicillin-resistant Staphylococcus aureus, Enterococcus faecium, Acinetobacter baumannii, Klebsiella pneumoniae, Escherichia coli and Pseudomonas aeruginosa being the most common. After univariate analysis (Table 2), the clinical factors associated with MDRO were: infection and cardiac disease as a cause of admission, total days in hospital, days in MICU, days on the ventilator, days with a central venous catheter, tracheostomy, prior hospitalization, prior antibiotic use (before hospitalization), immunosuppression, past history of hyperlipidemia, use of vasopressors, past history of surgery and white blood cell count >10,000/mm3 on admission.
Unadjusted and adjusted analysis for clinical features associated with MDRO pathogens. Multivariable model revealed eight clinical and demographics factors that were independently associated with MDRO after removing co-linear variables.
| Variable | Unadjusteda | Adjustedb | ||
|---|---|---|---|---|
| OR (95% CI) | p value | OR (95% CI) | p value | |
| Race (white vs. non-white) | 0.56 (0.32–0.99) | 0.044 | 0.44 (0.22–0.90) | 0.025 |
| Age (years) | 1.02 (1.003–1.03) | 0.02 | 1.02 (1.002–1.04) | 0.032 |
| Cause of admission | ||||
| Infection (hospital-acquired) | 3.7 (2.29–5.99) | <0.0001 | 3.3 (1.9–5.8) | <0.0001 |
| Cardiac | 0.47 (0.28–0.79) | 0.004 | ||
| Comorbidities | ||||
| Immunosuppression | 2.19 (1.38–3.49) | <0.001 | 2.04 (1.2–3.5) | 0.01 |
| Prior hospitalization | 2.08 (1.23–3.52) | 0.006 | ||
| Past surgical history | 1.69 (1.06–2.67) | 0.026 | 1.82 (1.05–3.14) | 0.033 |
| Prior history of hyperlipidemia | 1.80 (1.12–2.89) | 0.016 | 2.2 (1.2–3.8) | 0.008 |
| Vasopressors | 1.80 (1.05–3.06) | 0.031 | ||
| Tracheostomy | 7.61 (2.5–23.00) | <0.001 | ||
| WBC above 10,000/mm3 | 1.78 (1.12–2.82) | 0.015 | ||
| Days in hospital | 1.06 (1.04–1.09) | <0.0001 | 1.04 (1.01–1.07) | 0.005 |
| Days in MICU | 1.08 (1.04–1.12) | <0.0001 | ||
| Days on ventilator | 1.10 (1.05–1.15) | <0.0001 | 1.07 (1.01–1.12) | 0.013 |
| Days with CVC | 1.07 (1.04–1.10) | <0.0001 | ||
| Antibiotics | ||||
| Days on Vancomycin | 1.11 (1.06–1.17) | <0.0001 | ||
| Days on Linezolid | 1.20 (1.08–1.33) | <0.001 | ||
| Days on Daptomycin | 1.18 (1.005–1.38) | 0.044 | ||
| Days on Metronidazole | 1.11 (1.02–1.22) | 0.021 | ||
| Days on Piperacillin-tazobactam | 1.15 (1.05–1.26) | 0.002 | ||
| Days on Meropenem | 1.11 (1.06–1.16) | <0.0001 | ||
| Days on Tigecycline | 1.51 (1.19–1.91) | <0.001 | ||
| Days on Mycafugin | 1.29 (1.04–1.60) | 0.022 | ||
| Discharge to | ||||
| Home | 0.57 (0.36–0.91) | 0.019 | ||
The multivariate analysis included all variables with a univariate p-value of <0.05 (Table 2), and the only clinical features significantly associated with MDRO were: infection as a cause of admission (p<0.0001, OR=3.3), total days in hospital (p=0.005, OR=1.04) total days of ventilation (p=0.013, OR=1.07), immunosuppression (p=0.01, OR=2.04), past history of hyperlipidemia (p=0.008, OR=2.2), past surgical history (p=0.033, OR=1.82), age (p=0.032, OR=1.02) and white race (p=0.025, OR=0.44). Days of ventilation and days in hospital were further separated, into days before and after the onset of infection.
Relationship of MDROs to hospital days, ventilator days, prior and after the diagnosis of infectionThe median time for acquiring an MDRO infection while in the hospital was 8 days (range of 1–48 days). Each additional hospital day increased the risk of having an MDRO by about 5%.
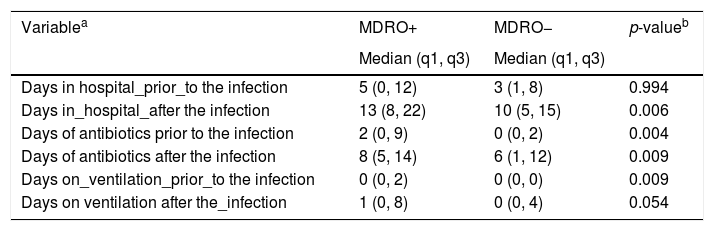
Days of hospitalization (the day of hospital admission was calendar day 1) were separated, into days prior and days after the diagnosis of infection. Both groups spent a similar number of days in the hospital prior to infection, but the MDRO positive patients spent a greater median number of days after infection than the MDRO negative patients. The median number of days in hospital after the diagnosis of infection was 13 and 10 days, respectively (p=0.006) for the MDRO positive and negative patients (Table 3). Culture negative control patients were not included in this analysis.
Pre- and post-infection comparisons between MDRO positive and negative group.
| Variablea | MDRO+ | MDRO− | p-valueb |
|---|---|---|---|
| Median (q1, q3) | Median (q1, q3) | ||
| Days in hospital_prior_to the infection | 5 (0, 12) | 3 (1, 8) | 0.994 |
| Days in_hospital_after the infection | 13 (8, 22) | 10 (5, 15) | 0.006 |
| Days of antibiotics prior to the infection | 2 (0, 9) | 0 (0, 2) | 0.004 |
| Days of antibiotics after the infection | 8 (5, 14) | 6 (1, 12) | 0.009 |
| Days on_ventilation_prior_to the infection | 0 (0, 2) | 0 (0, 0) | 0.009 |
| Days on ventilation after the_infection | 1 (0, 8) | 0 (0, 4) | 0.054 |
In the analysis of days on ventilation prior and after the diagnosis of infection we observed that more days on ventilation (prior to infection) were present for MDRO+ versus MDRO− patients (p=0.009). Additionally MDRO patients spent more median days on ventilation after infection than MDRO negative patients (p=0.054). Culture negative control patients were not included in this analysis.
Relationship of MDRO to antibiotic use, prior and after the diagnosis of the infectionMDRO positive patients had a greater median number of antibiotic days before infection and after infection, compared to MDRO negative patients. Among MDRO positive and negative patients the median number of days of antibiotics prescribed prior to the infection was 2 and 0 days respectively (p=0.004) (Table 3). After the diagnosis of the infection among the MDRO positive and negative patients the median number of days of antibiotics prescribed was 8 and 6 days respectively (p=0.009) (Table 3).
Multivariable analysis did not identify any specific antibiotic as independently associated with MDROs.
Appropriateness of initial antibiotic therapy and the impact of MDRO on outcomesAppropriate empiric antibiotic therapy (matching in vitro susceptibility to the isolated pathogen) was administrated in 105 out of 127 (82.6%) of MDRO positive cases. 72/105 (68.5%) patients received appropriate therapy within 24h of the onset of infection. Among MDRO negative patients with an identified pathogen, 60/65 received antibiotic therapy, which was appropriate in 54/60. The use of appropriate therapy was not associated with reduced mortality in either the MDRO positive or MDRO negative patients.
No statistically significant difference in 28-day ICU mortality rate was observed between MDRO positive and MDRO negative patients (20% vs 12.4%, p=0.07). In addition, there was no difference in the percent of patients discharged to home, nursing home, hospice or other long term care facilities (Table 1).
DiscussionIn this study, clinical features associated with MDRO pathogen infection in the ICU were identified, along with the impact of resistance and appropriate initial antimicrobial therapy on outcomes, including mortality. Only infection as a cause of admission to the hospital, total days in hospital, total days of mechanical ventilation, immunosuppression, past surgical history, past history of hyperlipidemia and age were independently associated with resistance. Moreover, white race was independently associated with protection against the MDRO infection.
Total days on ventilation may have been either a risk factor for, or a consequence of MDRO infection. To clarify this issue, we separated ventilator days into those prior and after the onset of infection. Those patients with MDRO had a greater median number of ventilator days after the onset of infection than those without MDRO. We also found, as did others, that one of the most important determinants of MDRO pathogen emergence was the duration of mechanical ventilation prior to the onset of pneumonia.17
Previous investigators have shown that prolonged hospitalization can predispose patients to infection with antibiotic-resistant bacteria,18 but they have not analyzed the relationship between hospital stay and antibiotic resistance in the same way as in this study. In the present study, each day of hospitalization increased the risk of MDRO by 5%, and the onset of MDRO infection was at a median of 8 days. However, we also found that MDRO infection itself prolonged hospitalization. Patients with MDRO spent a similar number of hospital days before infection and those without MDRO, but more hospital days after the onset of infection than those without MDRO.
Similarly, we examined the relationship of exposure to antibiotics to MDRO infection. While those with MDRO infection received more total antibiotics than those without, we also found that antibiotics were used more often and for a longer time both before and after the onset of ICU infection in those with MDROs compared to those without. Thus, antibiotic use was not only a necessary response to infection with resistant organisms, but also a risk factor for their acquisition.
No statistically significant difference was observed between MDRO positive and control patients for mortality. Although this is in contrast to many prior studies, in most instances, the mechanism for increased mortality was the frequent use of inappropriate empiric therapy. In our study, the use of appropriate empiric antibiotic therapy (matching in vitro susceptibility for the isolated pathogen) was common and most patients received the antibiotics within 24h of the onset of the infection. It seems possible that if resistance is anticipated and initial therapy is timely, and correct, then mortality may not be increased by the presence of MDRO pathogens.
Infection as an indication for admission was more frequently observed in the MDRO patients than in the control patients. Infection on ICU admission has been identified as a predisposing factor for Acinetobacter baumannii acquisition in a number of reports.19–21 The presence of an infection on ICU admission was also an independent risk factor for Acinetobacter baumannii bacteremia.22 Colonization or infection by MDRO and/or carbapenem-resistant Acinetobacter baumannii was also found in patients who had prior infection in a study from Brazil.23 It is likely that the presence of an infection at the time of admission led the physicians to use broad-spectrum antibiotics, which favored the selection of multidrug-resistant microorganisms as the cause of their next, nosocomial, infection. In a retrospective case–control study, Nseir et al. found that immune suppression was not associated with ICU-acquired MDRO bacteria,24 while we found it to be an independently associated risk factor. However, the concept of immune suppression is broad, and includes a heterogeneous population, only some of whom are at high risk.25
Our data also showed that in the multivariable analysis, past history of hyperlipidemia was associated with MDRO, and all these individuals were receiving cholesterol lowering therapy. An older agent, neomycin, can increase the risk of resistant flora,26 but statins may have an immune modulating effect, although their impact on bacteriology has not been carefully examined.27,28 Multivariable analysis also identified that for every one year increase in age, holding other independent predictors constant, the odds of having MDRO increased by 2%. Age above or equal to 65 years was identified by other investigators as an independent risk factor for harboring MDR-GNB at hospital admission.29
Most of our MDRO patients had undergone surgery in the previous 6 months (56%). Extended-spectrum beta-lactamases (ESBL) – producing bacteria have been shown to be more frequent in patients with a past history of urogenital surgery history.30 Prior surgery may also indirectly reflect the effect of antibiotic use. Moreover white race was independently associated with protection against the MDROs, although the validity of this finding is uncertain because the majority of our patients were white. However, previous studies have shown that VRE and MRSA are more common in African Americans than in white patients, and that ESBL positive E. coli are more common in Asian speaking patients than in others.31–33
Several limitations of this study merit discussion. In our control group many patients were culture negative and thus we could not be certain that they did not have an MDRO infection, however, we did not include any patients who had no cultures collected. Culture negative sepsis is a well-documented problem, and thus we believed that it was important to include these patients.4,14 In addition, in these patients, we could not determine if they received appropriate therapy, so they were excluded from any analysis of this factor. One other limitation is that, our sample size lacked the power to show a difference in mortality, so the observed lack of difference may have not have been present in a larger study. It is also possible that the lack of significant difference in mortality between the two groups may be partially explained by differences in infection sites. The high frequency of urinary tract infections may reflect the time period of this study, and its having been conducted prior to a change in the hospital policy for daily assessment of the need for bladder catheters. Finally this study is limited by its retrospective design and it was conducted at a single center in a single ICU over a few months and thus may not be fully representative of other hospitals. Moreover the study was performed seven years ago and this may affect the applicability and the validity of our findings, particularly with regard to the site of infection, as stated above.
ConclusionOur findings suggest that, knowing the risk factors for MDRO pathogens could lead to early appropriate antimicrobial administration, which could prevent mortality from these organisms. In our hospital, we did not withhold broad spectrum agents as empiric therapy in patients with clinical features associated with MDRO infection, and thus our rate of appropriate empiric therapy was high, which could explain the lack excess mortality in patients infected with MDRO pathogens.
Our findings are unique because of our breakdown of days before and after the onset of infection. With this approach, we found that, prolonged antibiotic therapy before infection is a risk factor for MDRO acquisition, reinforcing the need for judicious antimicrobial stewardship. However, after the onset of infection, patients with MDROs required a longer duration of antibiotic therapy than those without these organisms. In addition, we found that patients with MDRO pathogens had a longer duration of ventilation, both prior and after the onset of infection than MDRO negative patients. In addition, days of ventilation was an independent risk factor associated with MDRO infection. Interestingly, we found that although those with MDRO pathogens had a longer length of stay than those without, most of the prolonged length of stay was the consequence of this infection and not the cause of the emergence of MDRO pathogens.
FundingThis study was supported by internal institutional funds. No external funds were utilized for this study.
Authors’ contributionsEEM and MSN contributed to the design of the study. EEM executed the study, collected the data and wrote the first draft. SI performed the statistical analysis. MSN conceived the study, participated in its design and in analyzing data, provided overall supervision of this project and revised the article for important intellectual content. All authors read and approved the final manuscript.
Conflict of interestThe authors declare that they have no conflict of interest.
The study has been presented in 2011 at the European Society of Intensive Care Medicine.