To validate the diagnostic ability of six different scores to predict massive bleeding in a prehospital setting.
DesignRetrospective cohort.
SettingPrehospital attention of patients with severe trauma.
SubjectsSubjects with more than 15 years, a history of severe trauma (defined by code 15 criteria), that were initially assisted in a prehospital setting by the emergency services between January 2010 and December 2015 and were then transferred to a level one trauma center in Madrid.
VariablesTo validate: 1. Trauma Associated Severe Haemorrhage Score. 2. Assessment of Blood Consumption Score. 3. Emergency Transfusión Score. 4. Índice de Shock. 5. Prince of Wales Hospital/Rainer Score. 6. Larson Score.
Results548 subjects were studied, 76.8% (420) were male, median age was 38 (interquartile range [IQR]: 27–50). Injury Severity Score was 18 (IQR: 9–29). Blunt trauma represented 82.5% (452) of the cases. Overall, frequency of MB was 9.2% (48), median intensive care unit admission days was 2.1 (IQR: 0.8–6.2) and hospital mortality rate was 11.2% (59). Emergency Transfusión Score had the highest precisions (AUC 0.85), followed by Trauma Associated Severe Haemorrhage score and Prince of Wales Hospital/Rainer Score (AUC 0.82); Assessment of Blood Consumption Score was the less precise (AUC 0.68).
ConclusionIn the prehospital setting the application of any of the six scoring systems predicts the presence of massive hemorrhage and allows the activation of massive transfusion protocols while the patient is transferred to a hospital.
Validar a nivel extrahospitalario la capacidad diagnóstica de seis escalas de predicción para hemorragia masiva.
DiseñoCohorte retrospectiva.
ÁmbitoAtención extrahospitalaria del paciente con enfermedad traumática grave.
ParticipantesPacientes mayores de 15 años, que han sufrido un trauma grave (definido por los criterios de código 15), atendidos en el medio extrahospitalario por un servicio de atención sanitaria de emergencias desde enero de 2010 hasta diciembre de 2015 y trasladados a un centro hospitalario de alta complejidad en Madrid.
Variables de interés principalesSe validaron las siguientes escalas: 1. Trauma Associated Severe Haemorrhage score. 2. Assessment of Blood Consumption Score. 3. Emergency Transfusion Score. 4. Índice de Shock. 5. Prince of Wales Hospital/Rainer Score. 6. Larson Score.
ResultadosSe estudiaron 548 pacientes, el 76,8% (420) fueron hombres, una edad mediana de 38 (rango intercuartil [RIC]: 27-50). Injury Severity Score de 18 (RIC: 9-29). El trauma cerrado fue el 82,5% (452). La frecuencia global de HM fue de 9,2% (48), días de estancia en UCI de 2,1 (RIC: 0,8 - 6,2) y una mortalidad hospitalaria del 11,2% (59). La escala con mayor precisión fue la Emergency Transfusion Score (AUC 0,85), en segundo lugar se encuentran Trauma Associated Severe Haemorrhage y Prince of Wales Hospital/Rainer (AUC 0,82); la escala con menor precisión Assessment of Blood Consumption (AUC 0,68).
ConclusionesA nivel extrahospitalario la aplicación de cualquiera de las seis escalas predice la presencia de hemorragia masiva y permite la activación de los protocolos de transfusión masiva mientras el paciente es trasladado a un centro hospitalario.
Trauma is the main worldwide cause of death among people under 40 years of age (2010 – 5.1 million deaths, with 1 of every 10 being due to trauma), accounting for approximately 6% of overall mortality in the industrialized world and 12% in developing countries.1
Up until 2005, the mortality rate among patients requiring massive transfusion was 55–65%.2 The introduction of massive transfusion protocols (MTPs) reduced mortality to 45–50%.3 In turn, the incorporation of new strategies – resuscitation with damage control – further lowered the mortality rate to 30%.4 Recently, the Pragmatic Randomized Optimal Platelet and Plasma Ratios study has shown mortality to continue its downward trend: 26% in the case of plasma:platelet:packed red cell ratios of 1:1:2 and 22% for patients receiving ratios of 1:1:1.5 Despite such progress, however, massive bleeding (MB) remains the main cause of potentially avoidable mortality.6
Massive transfusion protocols are designed to facilitate a balanced supply of blood products in patients with MB and to allow the early start of resuscitation with damage control, even before the laboratory test results become available. The rapid identification of these patients with early activation of the MTP has been shown to be an independent predictor of patient survival.3,7
In recent years much effort has been dedicated to the identification of clinical, laboratory and imaging parameters capable of predicting MB and thus allowing early activation of the MTP. Different MB predicting scales have been developed in this respect, combining a broad range of variables and validated at both in-hospital level8,9 and individually at prehospital level10–15 – with the confirmation of good discriminating capacity.
The objective of the present study is to validate the diagnostic capacity of six MB predicting scales at prehospital level.
Design and methodsA retrospective cohort study was carried out. The study population consisted of severe trauma patients over 15 years of age assisted in the prehospital setting by the Municipal Emergency and Rescue Service – Civil Protection (Servicio de Asistencia Municipal de Urgencia y Rescate – Protección Civil [SAMUR-PC]) from January 2010 to December 2015, and transferred to a high-complexity hospital center in Madrid (Spain).
We included patients with severe trauma defined according to the “Code 15” criteria (Appendix A). Patients presenting the following upon admission to hospital were excluded: (a) cardiorespiratory arrest or premortem conditions disadvising resuscitation maneuvering; and (b) rejection of blood product transfusion on the part of the patient or relatives.
Initial prehospital care is provided by a specialized team composed of two physicians, two nurses and two technicians. Care is provided following the Advanced Trauma Life Support criteria. The following information was recorded on a retrospective basis: demographic data (age and gender), clinical variables (mechanism of injury, suspicion of unstable pelvic or femoral fracture), physiological parameters (first heart rate [HR], systolic blood pressure [SBP] and diastolic blood pressure [DBP]), laboratory test parameters (hemoglobin, base excess [BE] and venous blood lactic acid [“epoc®” blood gas analyzer from Medio Medico S.R.L.]), imaging parameters (Focused Abdominal Sonography for Trauma (FAST), using a “Sonosite 180 plus®” portable system from Fujifilm [variables recorded from the structured report of the SAMUR-PC]); hospital follow-up data (anatomical lesions, severity [Injury Severity Score, ISS]), and the need for arteriography/surgery to control bleeding (variables compiled from the database of our Unit).
Our transfusion registry was consulted to document the number of packed red blood cell units (PRCs) transfused to each patient included in the study. Massive bleeding was defined by the administration of ≥10 PRCs in the first 24h of admission after trauma (≥2500ml) or ≥5 PRCs (>1250ml) in the first 4h.
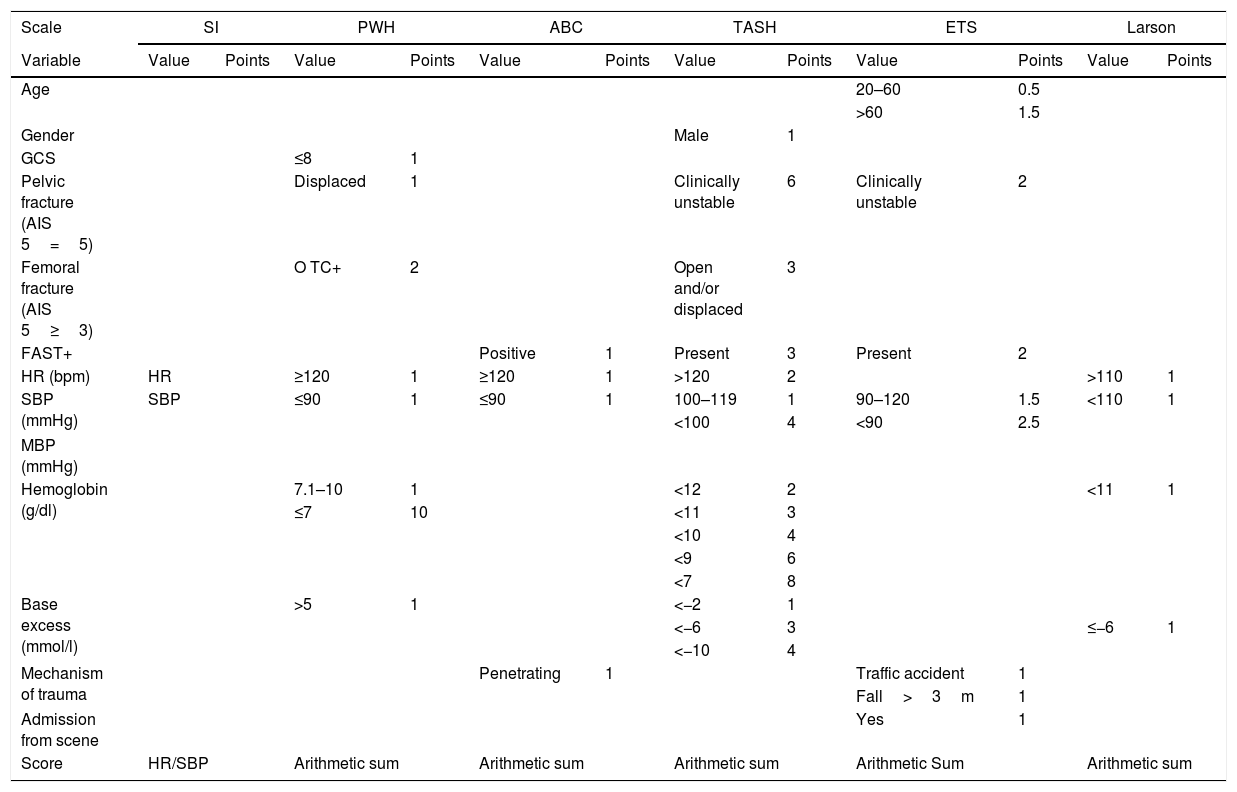
The prehospital physiological, anatomical, laboratory and imaging data were used to calculate the following MB predictive scales:1. Trauma Associated Severe Hemorrhage (TASH) Score16,17: This instrument was developed and validated on the basis of 6044 patients included in the TraumaRegister DGU® database. The TASH uses 7 variables: SBP, gender, hemoglobin, FAST, HR, BE and fracture of the pelvis or femur. Massive bleeding is defined as the transfusion of ≥10 PRCs in the first 24h. The score ranges from 0 to 29, with TASH ≥18 points indicating an MB probability of >50%. The maximum score of ≥27 points is associated to an MB risk of 100% (Table 1).2. Assessment of Blood Consumption (ABC) Score18: This instrument was developed on the basis of 596 civilian trauma patients. The ABC uses the following variables: penetrating trauma mechanism, SBP, HR and FAST. Massive bleeding is defined as the transfusion of ≥10 PRCs in the first 24h. The score ranges from 0 to 4. A score of ≥2 affords a sensitivity of 75% and a specificity of 86% in predicting MB (Table 1).3. Emergency Transfusion Score (ETS)19: This instrument is based on the analysis of 1103 civilian trauma patients. The ETS uses 6 variables: age, unstable pelvic fracture, SBP, FAST, mechanism of trauma (traffic accident, fall from >3m), and admission from the scene of the accident. Massive bleeding is defined as the transfusion of ≥10 PRCs in the first 24h. The score ranges from 0 to 9.5, with scores of 1, 3 and 9.5 points implying a probability of MB of 0.7%, 5% and 97%, respectively (Table 1).4. Shock Index (SI)20,21: This index is defined as heart rate divided by arterial pressure. Massive bleeding is defined as the transfusion of ≥10 PRCs in the first 24h. An SI score of >0.9 affords a sensitivity of 91% and a specificity of 80% in predicting MB (Table 1).5. Prince of Wales Hospital/Rainer Score (PWH)22: This model was developed from the analysis of 1891 civilian trauma patients (PWH Trauma Registry). The PWH uses 7 variables: HR, SBP, the Glasgow Coma Score (GCS), pelvic fracture, free abdominal fluid (FAST or computed tomography [CT]), BE and hemoglobin. Massive bleeding is defined as the transfusion of ≥5 PRCs in the first 4h. The score ranges from 0 to 10. A score of ≥6 affords a sensitivity of 36% and a specificity of 97% (Table 1).6. Larson Score (LS)23: This instrument is based on the analysis of 1124 patients included in a military database. The LS uses four variables: SBP, HR, hemoglobin and BE. Massive bleeding is defined as the transfusion of ≥10 PRCs in the first 24h. The score ranges from 0 to 4, with scores of 1, 3 and 4 points implying a probability of MB of 44%, 64% and 74%, respectively (Table 1).
Variables and scores of the predictive scales.
| Scale | SI | PWH | ABC | TASH | ETS | Larson | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Value | Points | Value | Points | Value | Points | Value | Points | Value | Points | Value | Points |
| Age | 20–60 | 0.5 | ||||||||||
| >60 | 1.5 | |||||||||||
| Gender | Male | 1 | ||||||||||
| GCS | ≤8 | 1 | ||||||||||
| Pelvic fracture (AIS 5=5) | Displaced | 1 | Clinically unstable | 6 | Clinically unstable | 2 | ||||||
| Femoral fracture (AIS 5≥3) | O TC+ | 2 | Open and/or displaced | 3 | ||||||||
| FAST+ | Positive | 1 | Present | 3 | Present | 2 | ||||||
| HR (bpm) | HR | ≥120 | 1 | ≥120 | 1 | >120 | 2 | >110 | 1 | |||
| SBP (mmHg) | SBP | ≤90 | 1 | ≤90 | 1 | 100–119 | 1 | 90–120 | 1.5 | <110 | 1 | |
| <100 | 4 | <90 | 2.5 | |||||||||
| MBP (mmHg) | ||||||||||||
| Hemoglobin (g/dl) | 7.1–10 | 1 | <12 | 2 | <11 | 1 | ||||||
| ≤7 | 10 | <11 | 3 | |||||||||
| <10 | 4 | |||||||||||
| <9 | 6 | |||||||||||
| <7 | 8 | |||||||||||
| Base excess (mmol/l) | >5 | 1 | <−2 | 1 | ||||||||
| <−6 | 3 | ≤−6 | 1 | |||||||||
| <−10 | 4 | |||||||||||
| Mechanism of trauma | Penetrating | 1 | Traffic accident | 1 | ||||||||
| Fall>3m | 1 | |||||||||||
| Admission from scene | Yes | 1 | ||||||||||
| Score | HR/SBP | Arithmetic sum | Arithmetic sum | Arithmetic sum | Arithmetic Sum | Arithmetic sum | ||||||
SI: Shock Index, PWH: Prince of Wales Hospital/Rainer, ABC: Assessment of Blood Consumption, TASH: Trauma Associated Severe Hemorrhage, ETS: Emergency Transfusion Score, GCS: Glasgow Coma Score, AIS: Abbreviated Injury Scale, FAST: Focused Abdominal Sonography for Trauma, HR: heart rate, SBP: systolic blood pressure, MBP: mean blood pressure.
Qualitative variables were reported as frequencies and proportions, while quantitative variables were presented as the mean (±standard deviation [SD]) and median (interquartile range [IQR]). The relationship between two categorical variables was explored using the chi-squared test and Fisher exact test. The comparison of two means was based on the Student t-test, after confirming normal data distribution with the Kolmogorov–Smirnov test and the homogeneity of variances with the Levene test. The Wilcoxon test was used in the case of a non-normal distribution. Statistical significance was considered for p<0.05.
We calculated the sensitivity (Se) and specificity (Sp) of each scale, plotted the receiver operating characteristic (ROC) curves, and calculated the area under the receiver operating characteristic (AUROC) curves with their respective 95% confidence intervals (95%CI). Use was made of the macro ROC for SPSS Statistics to determine the optimum cut-off point together with the corresponding Se and Sp values and likelihood ratios (LR+ and LR−). The positive and negative predictive values (PV+ and PV−) were also calculated. The SPSS® version 19.0 statistical package for MS Windows was used throughout.
The study was evaluated and approved by the Ethics Committee of our hospital.
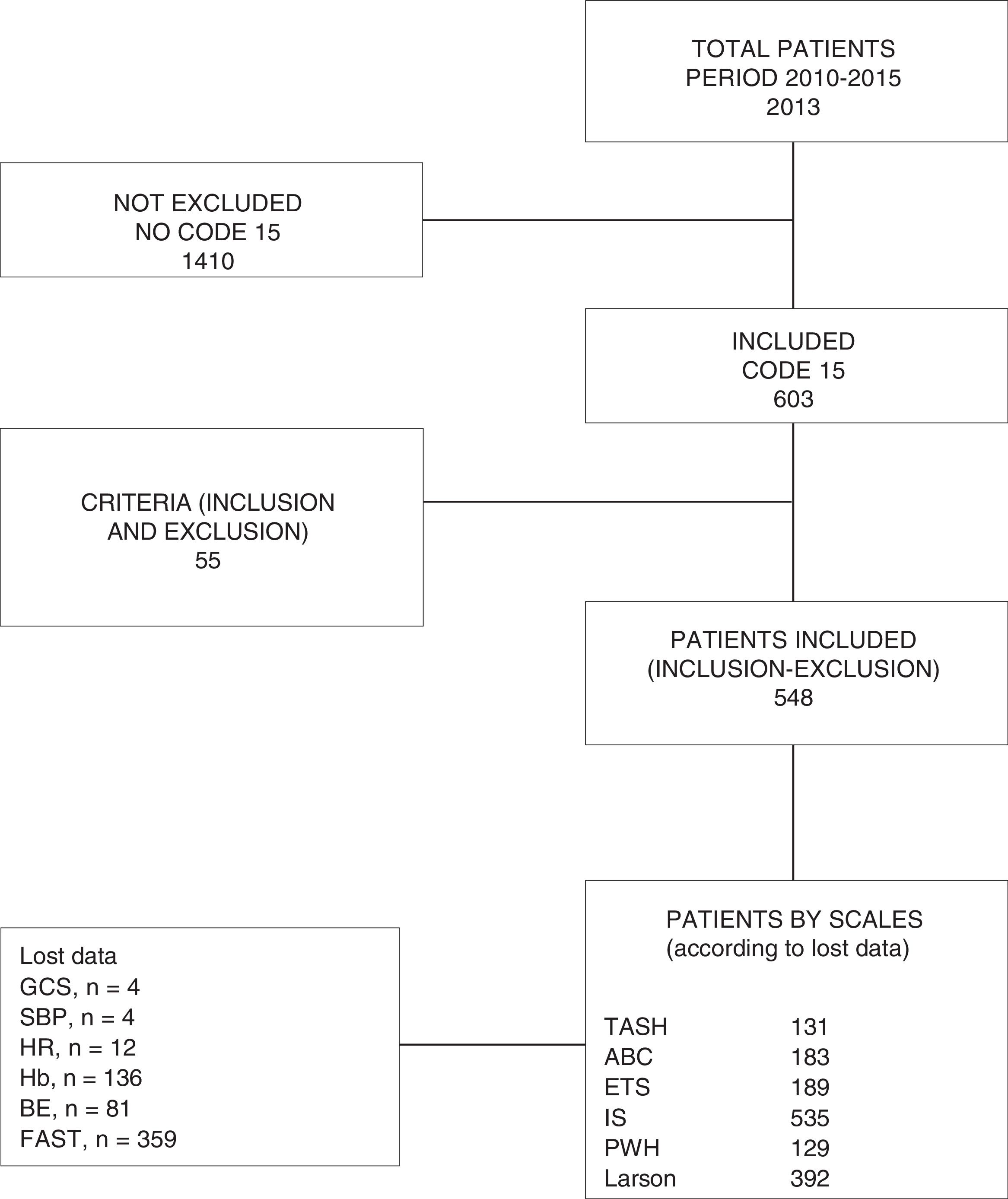
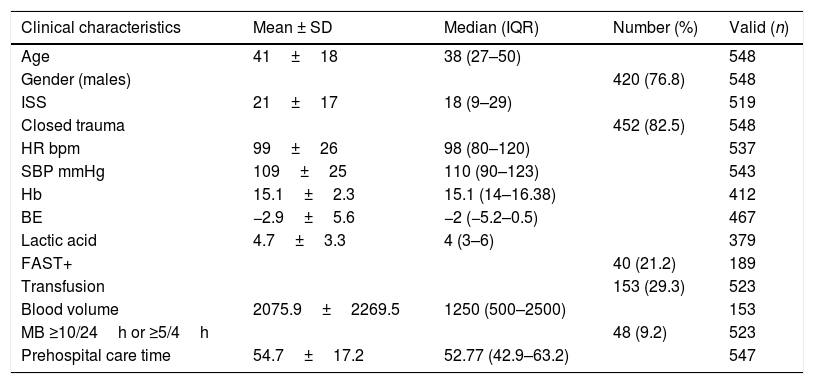
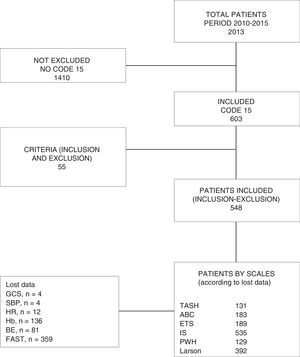
ResultsWe studied a total of 548 patients (Fig. 1), of which 76.8% (n=420) were males. The median age was 38 years (IQR: 27–50). The ISS score was 18 (IQR: 9–29). A total of 82.5% of the patients (n=452) suffered closed trauma. The overall frequency of MB was 9.2% (n=48), with an ICU stay of 2.1 days (IQR: 0.8–6.2). The in-hospital mortality rate was 11.2% (n=59). Table 2 describes the demographic and clinical characteristics of the study sample.
Demographic and clinical characteristics of the study population.
| Clinical characteristics | Mean ± SD | Median (IQR) | Number (%) | Valid (n) |
|---|---|---|---|---|
| Age | 41±18 | 38 (27–50) | 548 | |
| Gender (males) | 420 (76.8) | 548 | ||
| ISS | 21±17 | 18 (9–29) | 519 | |
| Closed trauma | 452 (82.5) | 548 | ||
| HR bpm | 99±26 | 98 (80–120) | 537 | |
| SBP mmHg | 109±25 | 110 (90–123) | 543 | |
| Hb | 15.1±2.3 | 15.1 (14–16.38) | 412 | |
| BE | −2.9±5.6 | −2 (−5.2–0.5) | 467 | |
| Lactic acid | 4.7±3.3 | 4 (3–6) | 379 | |
| FAST+ | 40 (21.2) | 189 | ||
| Transfusion | 153 (29.3) | 523 | ||
| Blood volume | 2075.9±2269.5 | 1250 (500–2500) | 153 | |
| MB ≥10/24h or ≥5/4h | 48 (9.2) | 523 | ||
| Prehospital care time | 54.7±17.2 | 52.77 (42.9–63.2) | 547 |
BE: base excess; FAST: Focused Abdominal Sonography for Trauma; HR: heart rate; Hb: hemoglobin; MB: massive bleeding; ISS: Injury Severity Score; SBP: systolic blood pressure.
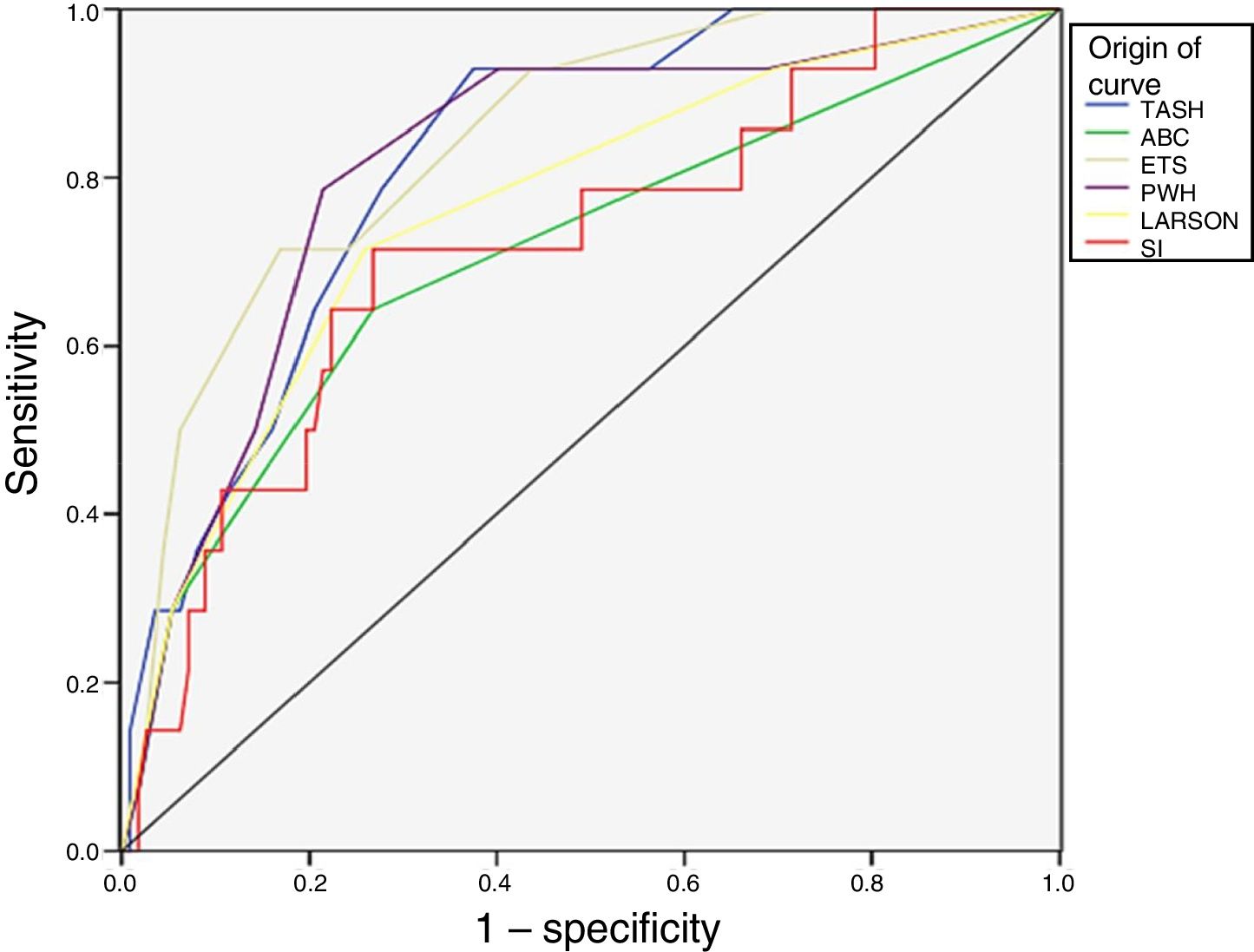
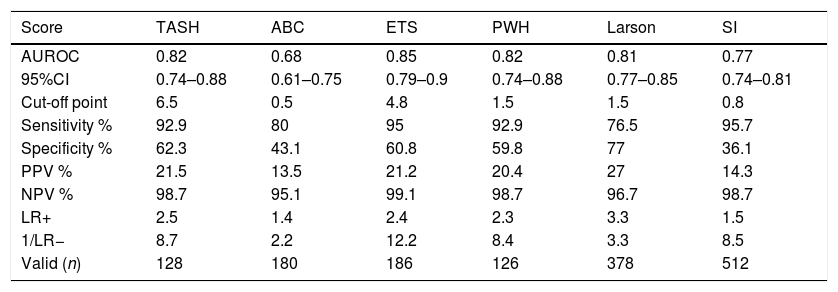
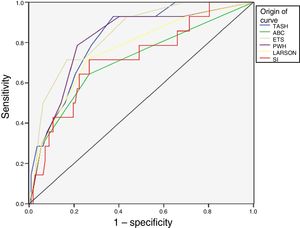
The scale with the greatest validity was found to be the ETS (AUC 0.85), followed by the TASH and PWH (AUC 0.82). The least precise instrument was the ABC (AUC 0.68). Fig. 2 shows the ROC curves of the 6 predictive scales.
The highest sensitivities corresponded to the ETS and SI (95% and 95.7%, respectively), with high negative predictive values in both cases (99.1% and 98.7%). Table 3 shows the calculated values (optimum cut-off point, Se and Sp, PV+ and PV−, LR+ and LR−) for each scale.
Comparison of the predictive scales.
| Score | TASH | ABC | ETS | PWH | Larson | SI |
|---|---|---|---|---|---|---|
| AUROC | 0.82 | 0.68 | 0.85 | 0.82 | 0.81 | 0.77 |
| 95%CI | 0.74–0.88 | 0.61–0.75 | 0.79–0.9 | 0.74–0.88 | 0.77–0.85 | 0.74–0.81 |
| Cut-off point | 6.5 | 0.5 | 4.8 | 1.5 | 1.5 | 0.8 |
| Sensitivity % | 92.9 | 80 | 95 | 92.9 | 76.5 | 95.7 |
| Specificity % | 62.3 | 43.1 | 60.8 | 59.8 | 77 | 36.1 |
| PPV % | 21.5 | 13.5 | 21.2 | 20.4 | 27 | 14.3 |
| NPV % | 98.7 | 95.1 | 99.1 | 98.7 | 96.7 | 98.7 |
| LR+ | 2.5 | 1.4 | 2.4 | 2.3 | 3.3 | 1.5 |
| 1/LR− | 8.7 | 2.2 | 12.2 | 8.4 | 3.3 | 8.5 |
| Valid (n) | 128 | 180 | 186 | 126 | 378 | 512 |
ABC: Assessment of Blood Consumption; AUROC: area under the receiver operating characteristic curve; ETS: Emergency Transfusion Score; CI: confidence interval; SI: Shock Index; PWH: Prince of Wales Hospital/Rainer; LR: likelihood ratio; TASH: Trauma Associated Severe Hemorrhage; NPV: negative predictive value; PPV: positive predictive value.
It is crucial to be able to identify MB patients (prediction) following severe trauma and to thus ensure early MTP activation.
Our study is the first to validate six scales for the prediction of MB after severe trauma at prehospital level. It includes particularly patients with closed trauma (82.5%), with a median ISS score of 18 – the overall frequency of MB being 9.2%. Three predictive scales in particular stand out: the ETS with an AUROC of 0.85, followed by the TASH and PWH scales (AUROC 0.82). Interestingly, although the ETS scale includes fewer parameters (laboratory test variables), it affords greater precision, and it comes as no surprise that the TASH and PWH instruments offer similar precision, since they include almost the same parameters. In relation to the optimum cut-off points, we recorded high sensitivities, with PV− values close to 100%, which would avoid losing patients with MB despite over-triage (false-positive results). Simple scales without laboratory test and imaging parameters, such as the Shock Index, offer good precision (AUROC 0.77) – thereby allowing their utilization on the part of prehospital services that lack point-of-care devices.
Different groups have attempted to improve the prediction of MB at prehospital level. Olaussen et al.11 validated the prehospital Shock Index as a predictor of MB and underscored that its simplicity facilitates application at prehospital level. Walcher et al.13 conducted a prospective study to establish the precision of FAST in the detection of free abdominal fluid at prehospital level (pFAST), demonstrating that it improves the triage and management of trauma patients. Goodman et al.14 in turn evaluated the capacity of the ABC scale at prehospital level (pABC) in predicting the need for massive transfusion. During air transfer, nurses performed the pFAST and calculated pABC using the variables of the standard ABC before reaching the hospital center. The diagnostic usefulness was evidenced by an AUC of 0.85 and a negative predictive value of 96%. Holcomb et al.15 conducted a prehospital study with the purpose of starting PRC and fresh frozen plasma transfusion in patients meeting MB criteria as established by the ABC scale.
Our study has many limitations, including its retrospective design and the loss of data this implies (Fig. 1). A fundamental limitation in studies on the prediction of MB is the lack of a universally accepted definition of MB. The current definition of ≥10 PRCs in 24h introduces bias by excluding early death cases and patients with active bleeding and early control of bleeding (arteriography and/or surgery) that do not receive 10 PRCs, and by including patients with no need for acute transfusion (administration of the tenth PRC 20h after admission, and without clinical or complementary data indicative of active bleeding). With the aim of reducing survival bias caused by the classical diagnostic criterion (≥10 PRCs/24h), our group included a second criterion (≥5 PRCs/4h)24 (the first criterion yielding a frequency of MB of 8.2%, versus 9.2% on adding the second criterion), thus including early death cases or patients with early control of active bleeding.
New concepts are emerging, such as: (a) “substantial bleeding”, which is more related to the severity of bleeding than to transfusion volume. It defines patients who have received at least one PRC within the first 2h and 5 or more subsequent PRCs or early death due to bleeding in the first 4h. With this definition we reduce survival bias, including patients requiring large transfusion volumes as well as those that die before receiving large transfusion volumes; (b) “resuscitation intensity”. The investigators of the Prospective Observational Multicenter Major Trauma Transfusion study have proposed this new concept, which includes all the fluid therapy received in the context of initial resuscitation. One liter of crystalloids, half a liter of colloids, one PRC, one plasma unit and one platelet unit are regarded as a resuscitation unit. A study was made, evidencing an increase in mortality among patients administered more than three resuscitation units in the first 30min of care; (c) “critical administration threshold” (CAT). This new concept has been proposed as an indicator of bleeding severity. It was developed to minimize survival bias and to offer a tool capable of better reflecting the resuscitation rate than volume alone. In this context, CAT (+) is defined as a patient who has received three or more PRCs in some hour within the first 24h after trauma. Patients with CAT (+) were at a four-fold higher risk of death than those with CAT (−).25–28
Other limitations of the studied scales are: (a) some of them are difficult to apply in the prehospital setting (TASH), since they not only require more time to apply but also need complementary tests that are often not available at prehospital level; (b) it remains to be determined which of the different measurements of the physiological variables (the first SBP recording, the poorest HR) should be included in the calculation.
In sum: (a) we need a new definition of massive bleeding/massive transfusion, applying new concepts such as resuscitation intensity and critical administration threshold – concepts that allow a more precise diagnosis and thus facilitate the conduction of reliable and reproducible studies; (b) dynamic values (expressing a trend) should be included, not simply point values referred to the evolution of the patient.
ConclusionsThis study has validated six scales for the prediction of massive bleeding at prehospital level, of which three in particular stand out in terms of performance: ETS (AUC 0.85), TASH and PWH (AUROC 0.82). At prehospital level, the application of any of these six scales predicts the presence of massive bleeding and allows activation of the massive transfusion protocols while the patient is being moved to a hospital center.
Authorship/CollaboratorsAll the authors have participated in the investigation and in the preparation of the article. Likewise, all of them have reviewed and approved the final manuscript version.
Luis Juan Terceros-Almanza: literature search, database, data compilation, statistical analysis and drafting of the manuscript.
Carlos García-Fuentes: literature search, database, data compilation, statistical analysis and drafting of the manuscript.
Susana Bermejo-Aznárez: drafting of the manuscript.
Isidro Javier Prieto del Portillo: data compilation.
Carolina Mudarra-Reche: data compilation.
Helena Domínguez-Aguado: data compilation.
Rubén Viejo-Moreno: data compilation.
Jesús Barea-Mendoza: data compilation.
Roberto Gómez-Soler: literature search, database, data compilation, statistical analysis and drafting of the manuscript.
Isabel Casado-Flores: drafting of the manuscript.
Mario Chico-Fernández: drafting of the manuscript.
Conflicts of interestThe authors declare that they have no conflicts of interest.
- 1.
Hemodynamic instability: systolic blood pressure (SBP) <90mmHg, heart rate (HR) >120bpm.
- 2.
Respiratory distress, need for mechanical ventilation and/or unstable airway.
- 3.
Altered level of consciousness due to trauma, with GCS<12.
- 4.
Penetrating wounds of the head, neck, torso or extremities above the knee and elbow, or which imply neurovascular damage.
- 5.
Traumatic amputations proximal to the wrist or ankle.
- 6.
Burns affecting over 15% of the body surface and/or facial burns, suspected inhalation injury or respiratory distress.
- 7.
Open or unstable pelvic fracture.
- 8.
Positive Focused Abdominal Sonography for Trauma (FAST).
- 9.
Patients over 65 years of age with some of the following clinical profiles: altered level of consciousness due to trauma with GCS<14; stable patient with multiple trauma; open fractures of the extremities; multiple closed fractures (more than two long bones) of the extremities; stable pelvic fractures; high-energy trauma mechanism.
- 10.
Criterion of the physician in charge of primary assessment.
Please cite this article as: Terceros-Almanza LJ, García-Fuentes C, Bermejo-Aznárez S, Prieto del Portillo IJ, Mudarra-Reche C, Domínguez-Aguado H, et al. Predicción de hemorragia masiva a nivel extrahospitalario: validación de seis escalas. Med Intensiva. 2019;43:131–138.