To analyze mortality and functional outcome in patients with severe spontaneous intracerebral hemorrhage (ICH), and identify the clinical characteristics, radiological findings and therapeutic procedures predictive of mortality in the Intensive Care Unit (ICU) and during hospitalization, as well as of poor functional results at 6 months.
DesignA prospective, observational study was carried out.
SettingNeurocritical Care Unit of a university hospital.
PatientsPatients diagnosed with ICH were included over a period of 23 months.
Variables of interestDemographic characteristics, cardiovascular risk factors, regular medication, laboratory test parameters, cranial CT findings, therapeutic procedures and outcome data.
InterventionNone.
ResultsA total of 186 patients with ICH met the inclusion criteria. Surgery to evacuate ICH was performed in 25.8% of the patients. The mortality rate was 46.7%. The modified Rankin score at 6 months was 5 (RI: 4.6). Multivariate Cox regression analysis showed the presence of diabetes, prior anticoagulation, as well as APACHE II severity and the type of bleeding on the cranial CT scan to be predictors of mortality and poor functional outcomes. On the other hand, neurosurgical procedures and intracranial pressure (ICP) monitoring were associated with better outcomes.
ConclusionThe presence of comorbidities such as diabetes, or previous anticoagulation, as well as the CT findings were associated to poorer outcomes. In contrast, ICP monitoring and early neurosurgery were predictive of longer survival and better functional outcomes.
Evaluar la mortalidad y el resultado funcional final de los pacientes con hemorragia intraparenquimatosa espontánea grave (HIP). Determinar las características clínicas, radiológicas y terapéuticas con mayor poder predictivo sobre la mortalidad en la Unidad de Cuidados Intensivos (UCI) y durante el ingreso hospitalario, así como sobre los malos resultados funcionales a los 6 meses.
DiseñoEstudio prospectivo, observacional.
ÁmbitoUCI de un hospital universitario.
PacientesDurante 23 meses se incluyó a aquellos pacientes con diagnóstico de HIP.
Variables de interésDatos demográficos, factores de riesgo cardiovascular, medicación habitual, datos de laboratorio, tomografía craneal (TC), neuromonitorización, manejo terapéutico y evaluación pronóstica.
IntervenciónNinguna.
ResultadosSe incluyó a un total de 186 pacientes. En el 25,8% se realizó evacuación quirúrgica de la HIP. La mortalidad fue del 46,7%. La mediana del Rankin modificado a los 6 meses fue de 5 (RI: 4; 6). El análisis multivariante de regresión de Cox mostró que la presencia de diabetes, tratamiento previo con anticoagulantes, la gravedad según APACHE II y el tipo de hemorragia en la TC de cráneo se comportaron como variables predictoras de mortalidad y malos resultados funcionales, mientras que la realización de procedimientos neuroquirúrgicos y la monitorización de la presión intracraneal (PIC) asociaron una mayor supervivencia y mejores resultados.
ConclusiónLa presencia de comorbilidades, como la diabetes o el tratamiento previo con anticoagulantes, así como los hallazgos de la TC se asociaron a peores resultados. Por contra, el marcador de mayor supervivencia y mejor resultado funcional era la monitorización de la PIC, así como la cirugía precoz.
Cerebrovascular accident (CVA) or stroke is a common disorder. Specifically, spontaneous intracerebral hemorrhage (ICH) is the second leading cause of CVA, following ischemic stroke (10–15%).1 The annual incidence of ICH is estimated to be between 16 and 33 cases/100,000 inhabitants, and its prevalence has increased in recent years in relation to aging of the general population and the increasingly widespread use of antithrombotic therapy in younger patients.2–7
As a result of the increasing frequency of the disorder and the associated need for specialized treatment, affected patients often require admission to the Intensive Care Unit (ICU) for adequate management.1,6 This fact, and subsequent admission to the conventional hospital ward, imply an important use of social and economic resources.8 However, despite advances in specialized care in the ICU, the mortality rate associated to ICH is very high–one-half of all deaths occurring within the first 48h–and the functional outcomes remain very disappointing: only 12–39% of those patients who survive the episode are functionally independent. These considerations justify the different studies that have sought to establish predictive mortality and functional outcome models based on different demographic, clinical and biological variables, with the aim of optimizing the ulterior management of patients with ICH.8–13 However, many of these investigations only address short-term mortality in the ICU or in hospital. On the other hand, uncertainty remains regarding the influence of different invasive procedures such as immediate surgery or intracranial pressure (ICP) catheter monitoring and brain tissue oxygenation pressure (PtiO2) upon the final outcome.11,14
The main objective of this study is to determine mortality and functional outcome in patients with severe ICH. Likewise, an evaluation is made of the clinical, radiological and therapeutic variables with the strongest predictive capacity in relation to mortality in the ICU and during hospital admission, and to poor functional outcome after 6 months.
Materials and methodsStudy designA prospective observational study was carried out from October 2011 to August 2013 in the ICU of the Unit of Clinical Management and Critical and Emergency Care of Virgen del Rocío Hospital in Seville (Spain). The study was approved by the Clinical Research Ethics Committee of our center, and the obtainment of informed consent was not considered necessary, due to the strictly observational nature of the study.
We included all patients diagnosed with severe ICH. The inclusion criteria were: (1) individuals over 14 years of age (with no upper age limit); (2) ICH confirmed by brain computed tomography (CT); (3) admission to the ICU in the first 48h following the acute event; (4) patient level of consciousness upon arrival in hospital (in the absence of pharmacological, metabolic or hemodynamic interferences) defined by a Glasgow Coma Score (GCS) of ≤8; (5) bleeding volume >30ml at supratentorial level or >10ml at infratentorial level; and (6) need for external ventricular drainage (EVD) due to the presence of obstructive hydrocephalus. Patients with ICH secondary to aneurysms or vascular malformations were excluded, as were those with underlying tumor lesions or previous trauma, protuberance hemorrhage due to cerebral herniation (Duret hemorrhage), and secondary ischemic tissue reperfusion damage or hemorrhagic transformation. We also excluded patients with neurological sequelae due to previous congenital or acquired disorders and those in which short- to middle-term follow-up was not possible.
Characteristics of the study populationThe variables recorded comprised patient age and gender; cardiovascular risk factors such as the presence of previous arterial hypertension (AHT) defined by a blood pressure of over 140/80mmHg, diabetes mellitus (DM), known dyslipidemia (DL), smoking defined by the World Health Organization (WHO) as the current and regular smoking of >10cigarettes/day (including ex-smokers of up to 6 months); toxic abuse including alcohol (defined as the daily consumption of four standard beverage units (SBU) in males and 2.5 in females [in Spain 1 SBU=10g of pure alcohol]); illegal drug use (e.g., cocaine or amphetamines) on a continuous or sporadic basis; chronic treatment for background disease in the form of anticoagulants (vitamin K antagonists) or antiplatelet drugs such as aspirin or clopidogrel; and the presence of previous diseases predisposing to intracranial bleeding, such as congenital coagulopathy (hemophilia, vasculitis, Moyamoya disease or amyloidosis).
The clinical situation of the patients was evaluated upon admission by means of the Acute Physiology and Chronic Health Evaluation (APACHE) II score, while level of consciousness was scored with the GCS (in the absence of pharmacological, metabolic or hemodynamic interferences), and neurological defects were assessed on the basis of clinical signs such as pupil response, focal neurological alterations, aphasia or Jacksonian seizures when the patient was not under the effects of sedation or neuromuscular blockers. In these latter situations, in the event of clinical suspicion, an electroencephalographic study was made.
Radiological and laboratory test dataA brain CT scan was made upon patient admission, and again after 24h in the case of clinical stability (for evolutive monitoring of the lesions) or at any time during the clinical course in the event of neurological worsening. The information from these studies was obtained by two specialists in neurocritical care blinded to the clinical condition and prognosis of the patients. The findings were subsequently contrasted with the corresponding radiological reports. The following data were recorded: location of hemorrhage; volume of ICH using the formula based on calculation of the volume of a sphere or ellipsoid (ABC/2); the presence, amount and severity of intraventricular hemorrhage (IVH) estimated from the Graeb scale; the existence of radiological signs of mass effect (defined as midline displacement >5mm, compression or obliteration of the third ventricle, or compression of the basal cisterns); and repeat bleeding signs at control CT (defined as an increase in hematoma volume of >33%).15–17
In the case of hematomas of deep supratentorial location we used the Modified Intracerebral Hemorrhage Score (MICH), which quantifies the level of consciousness according to the GCS, the volume of the hematoma in cubic centimeters (cc), and the presence of hydrocephalus or intraventricular hemorrhage.10
For prognostic purposes, and in addition to the routine parameters, we documented the hemoglobin level and blood glucose concentration upon admission and after 24h in all patients.
Procedures and treatment modalitiesThe patients were studied based on the diagnostic and treatment procedures contemplated in the 2010 clinical practice guides of the AHA/ASA referred to the management of ICH, and on the protocols of our own center, which include invasive neuromonitoring (recording of ICP and PtiO2) in patients with GCS≤8 or patients subjected to sedoanalgesia, and the use of imaging techniques for evidencing intracranial hypertension, provided no limitation of therapeutic effort was contemplated.9 In those cases in which an underlying secondary vascular cause (arteriovenous or aneurysmal malformation) was suspected, we requested a diagnostic arteriographic study.
The indication of neurosurgery was based on the established clinical practice guides or on the criterion of the neurosurgeon. In all cases surgery was performed within the first 24h following symptoms onset. A standard craniotomy was performed according to the location of the hematoma: superficial lobular hematomas measuring >3cm in diameter at <1cm from the cortical layer, in patients with diminished consciousness and GCS score 9–12 points; or cerebellar hematomas measuring >3cm in diameter with evidence of brainstem compression or secondary obstructive hydrocephalus. External ventricular drainage was indicated in patients with moderate to severe IVH or secondary obstructive hydrocephalus (pure IVH, deep hematomas with important intraventricular invasion or infratentorial hematomas of the cerebellum not amenable to craniotomy, with progressive worsening of level of consciousness clearly attributable to hydrocephalus). Craniotomy and EVD were performed in patients with deep supratentorial hematomas under 60 years of age with progressive worsening of level of consciousness and with >30cc of volume with >5mm midline displacement and a mean MICH score of 2–4.8
Intraventricular fibrinolysis was prescribed in those patients with moderate to severe IVH and a Graeb score >5 points, for the treatment of obstructive hydrocephalus secondary to the intraventricular clot, after discarding the presence of vascular malformations, and provided the coagulation tests proved normal and EVD was permeable. Fibrinolysis was carried out with urokinase 10,000IU/12h. The duration of the treatment was 5 days, with adjustment according to clinical criteria and the evolutive changes evaluated by CT every 48h.
Clinical evolutionThe following neurological complications were recorded: hydrocephalus, brain edema, seizures, repeat bleeding and infections.
The following general information was also collected: days of stay in the ICU and in hospital, and mortality during admission to the ICU and in hospital or at 6 months post-hemorrhage. The patients that were still alive at discharge from hospital were evaluated after 6 months using the modified Rankin scale.18 This scale comprises seven categories with scores that increase with patient severity and functional disability after stroke. The extreme scores are 0=asymptomatic and 6=death.
Statistical analysisA descriptive analysis was made of the numerical variables, which were summarized as the mean and standard deviation or–in the case of asymmetrical distributions–as the median and interquartile range (IQR). We subsequently performed an inferential analysis, establishing relationships between categorical variables. The chi-squared test or Fisher exact test was used to assess the relationship between two qualitative variables. The comparison of means was carried out to analyze the association between a dichotomic qualitative variable and a quantitative variable. The Student t-test was used, following validation of the requirements referred to randomness, independence, normality and equality of variance. In the event of a non-normal distribution, we used the Mann–Whitney U-test. The corresponding 95% confidence interval (95% CI) was calculated when significant differences were detected. An initial univariate inferential analysis was performed, followed by a multivariate analysis based on the Cox regression (proportional hazards) model. The multivariate model included those variables found to be significant (p<0.1) in the previous univariate analysis. We used this method to investigate the effect of different variables (e.g., age, gender, personal history, clinical presentation upon admission, CT findings, etc.) which hypothetically could be predictive over follow-up until death (or not) of the patient. In relation to the variables included in the final model, we analyzed the hazard ratio (HR) and 95% CI. This afforded incident hazard rates referred to the event of interest (mortality) adjusted to the effect of one or more predictive variables. In order to obtain the prognostic model at 6 months, we estimated the relative risk (RR) and 95% CI of the variables based on Cox regression analysis with fixed times (evaluation at 6 months) in all the subjects. In this sense, and for analytical purposes, the functional outcomes were classified into two groups according to the modified Rankin score obtained: good functional outcome (scores 1 and 2) or poor functional outcome (scores 3, 4 and 5).
A level of statistical significance of p<0.05 was considered for all comparisons. The SPSS version 22.0 statistical package (IBM Corporation, NY, USA) was used throughout.
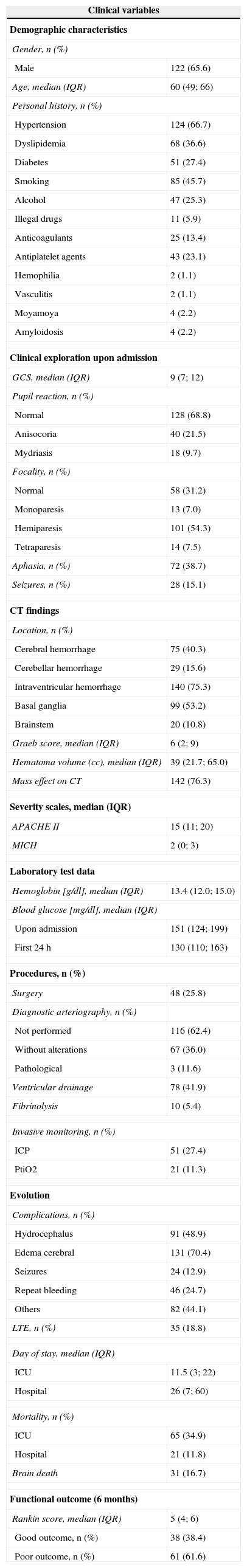
ResultsA total of 186 patients with ICH met the study inclusion criteria. The patient characteristics, clinical parameters, CT findings, procedures and evolutive data of the sample are shown in Table 1. The median age was 60 years (IQR 49; 66), and 122 patients were males (65.6%). The most common risk factor in our series was arterial hypertension (66.7%), followed by antiplatelet drug treatment (23.1%). The median GCS score upon admission was 9 points (IQR 7; 12), and the basal ganglia represented the most frequent location of ICH (53.2%). The median hematoma volume was 39cc (IQR 21.7; 65.0). A total of 25.8% of the patients included in the study underwent surgical evacuation of ICH. The overall mortality rate was 46.7% (89 patients); of these subjects, 65 (34.9%) died in the ICU, 11.8% (21 patients) during hospital stay, and three in the course of the 6 months following stroke.
General characteristics of the 186 patients included in the study.
| Clinical variables | |
|---|---|
| Demographic characteristics | |
| Gender, n (%) | |
| Male | 122 (65.6) |
| Age, median (IQR) | 60 (49; 66) |
| Personal history, n (%) | |
| Hypertension | 124 (66.7) |
| Dyslipidemia | 68 (36.6) |
| Diabetes | 51 (27.4) |
| Smoking | 85 (45.7) |
| Alcohol | 47 (25.3) |
| Illegal drugs | 11 (5.9) |
| Anticoagulants | 25 (13.4) |
| Antiplatelet agents | 43 (23.1) |
| Hemophilia | 2 (1.1) |
| Vasculitis | 2 (1.1) |
| Moyamoya | 4 (2.2) |
| Amyloidosis | 4 (2.2) |
| Clinical exploration upon admission | |
| GCS, median (IQR) | 9 (7; 12) |
| Pupil reaction, n (%) | |
| Normal | 128 (68.8) |
| Anisocoria | 40 (21.5) |
| Mydriasis | 18 (9.7) |
| Focality, n (%) | |
| Normal | 58 (31.2) |
| Monoparesis | 13 (7.0) |
| Hemiparesis | 101 (54.3) |
| Tetraparesis | 14 (7.5) |
| Aphasia, n (%) | 72 (38.7) |
| Seizures, n (%) | 28 (15.1) |
| CT findings | |
| Location, n (%) | |
| Cerebral hemorrhage | 75 (40.3) |
| Cerebellar hemorrhage | 29 (15.6) |
| Intraventricular hemorrhage | 140 (75.3) |
| Basal ganglia | 99 (53.2) |
| Brainstem | 20 (10.8) |
| Graeb score, median (IQR) | 6 (2; 9) |
| Hematoma volume (cc), median (IQR) | 39 (21.7; 65.0) |
| Mass effect on CT | 142 (76.3) |
| Severity scales, median (IQR) | |
| APACHE II | 15 (11; 20) |
| MICH | 2 (0; 3) |
| Laboratory test data | |
| Hemoglobin [g/dl], median (IQR) | 13.4 (12.0; 15.0) |
| Blood glucose [mg/dl], median (IQR) | |
| Upon admission | 151 (124; 199) |
| First 24h | 130 (110; 163) |
| Procedures, n (%) | |
| Surgery | 48 (25.8) |
| Diagnostic arteriography, n (%) | |
| Not performed | 116 (62.4) |
| Without alterations | 67 (36.0) |
| Pathological | 3 (11.6) |
| Ventricular drainage | 78 (41.9) |
| Fibrinolysis | 10 (5.4) |
| Invasive monitoring, n (%) | |
| ICP | 51 (27.4) |
| PtiO2 | 21 (11.3) |
| Evolution | |
| Complications, n (%) | |
| Hydrocephalus | 91 (48.9) |
| Edema cerebral | 131 (70.4) |
| Seizures | 24 (12.9) |
| Repeat bleeding | 46 (24.7) |
| Others | 82 (44.1) |
| LTE, n (%) | 35 (18.8) |
| Day of stay, median (IQR) | |
| ICU | 11.5 (3; 22) |
| Hospital | 26 (7; 60) |
| Mortality, n (%) | |
| ICU | 65 (34.9) |
| Hospital | 21 (11.8) |
| Brain death | 31 (16.7) |
| Functional outcome (6 months) | |
| Rankin score, median (IQR) | 5 (4; 6) |
| Good outcome, n (%) | 38 (38.4) |
| Poor outcome, n (%) | 61 (61.6) |
APACHE, Acute Physiology and Chronic Health Evaluation; cc, cubic centimeters; GCS, Glasgow Coma Scale; H, hemorrhage; LTE, limitation of therapeutic effort; MICH, Modified Intracerebral Hemorrhage Score; ICP, intracranial pressure; PtiO2, brain tissue oxygenation pressure; IQR, interquartile range; CT, computed tomography.
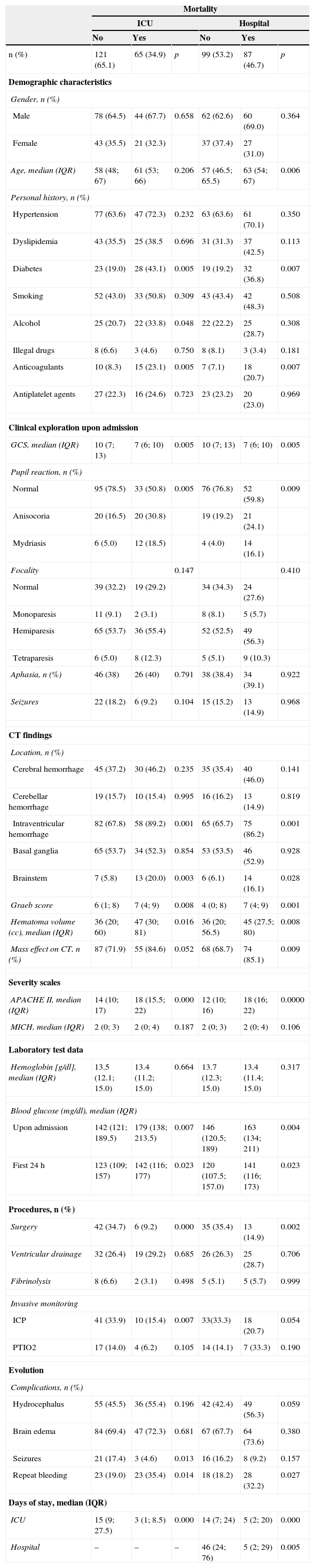
Table 2 shows the differences of the different variables between the patients that died and the survivors. Of note is the observation that elevated blood glucose upon admission and after 24h, diabetes, and oral anticoagulant therapy were associated to a higher mortality rate both in the ICU and in hospital. Among other variables, the level of consciousness upon admission, pupil response, bleeding location at brainstem level, the presence of IVH assessed by the Graeb scale, and the APACHE II score all showed differences between the patients that died in the ICU or in hospital and the survivors. Neurosurgical intervention also resulted in differences between the two groups, since survival was found to be greater among the patients that underwent surgical treatment. These data are summarized in Table 3.
Distribution of the variables according to mortality in the ICU and in hospital.
| Mortality | ||||||
|---|---|---|---|---|---|---|
| ICU | Hospital | |||||
| No | Yes | No | Yes | |||
| n (%) | 121 (65.1) | 65 (34.9) | p | 99 (53.2) | 87 (46.7) | p |
| Demographic characteristics | ||||||
| Gender, n (%) | ||||||
| Male | 78 (64.5) | 44 (67.7) | 0.658 | 62 (62.6) | 60 (69.0) | 0.364 |
| Female | 43 (35.5) | 21 (32.3) | 37 (37.4) | 27 (31.0) | ||
| Age, median (IQR) | 58 (48; 67) | 61 (53; 66) | 0.206 | 57 (46.5; 65.5) | 63 (54; 67) | 0.006 |
| Personal history, n (%) | ||||||
| Hypertension | 77 (63.6) | 47 (72.3) | 0.232 | 63 (63.6) | 61 (70.1) | 0.350 |
| Dyslipidemia | 43 (35.5) | 25 (38.5 | 0.696 | 31 (31.3) | 37 (42.5) | 0.113 |
| Diabetes | 23 (19.0) | 28 (43.1) | 0.005 | 19 (19.2) | 32 (36.8) | 0.007 |
| Smoking | 52 (43.0) | 33 (50.8) | 0.309 | 43 (43.4) | 42 (48.3) | 0.508 |
| Alcohol | 25 (20.7) | 22 (33.8) | 0.048 | 22 (22.2) | 25 (28.7) | 0.308 |
| Illegal drugs | 8 (6.6) | 3 (4.6) | 0.750 | 8 (8.1) | 3 (3.4) | 0.181 |
| Anticoagulants | 10 (8.3) | 15 (23.1) | 0.005 | 7 (7.1) | 18 (20.7) | 0.007 |
| Antiplatelet agents | 27 (22.3) | 16 (24.6) | 0.723 | 23 (23.2) | 20 (23.0) | 0.969 |
| Clinical exploration upon admission | ||||||
| GCS, median (IQR) | 10 (7; 13) | 7 (6; 10) | 0.005 | 10 (7; 13) | 7 (6; 10) | 0.005 |
| Pupil reaction, n (%) | ||||||
| Normal | 95 (78.5) | 33 (50.8) | 0.005 | 76 (76.8) | 52 (59.8) | 0.009 |
| Anisocoria | 20 (16.5) | 20 (30.8) | 19 (19.2) | 21 (24.1) | ||
| Mydriasis | 6 (5.0) | 12 (18.5) | 4 (4.0) | 14 (16.1) | ||
| Focality | 0.147 | 0.410 | ||||
| Normal | 39 (32.2) | 19 (29.2) | 34 (34.3) | 24 (27.6) | ||
| Monoparesis | 11 (9.1) | 2 (3.1) | 8 (8.1) | 5 (5.7) | ||
| Hemiparesis | 65 (53.7) | 36 (55.4) | 52 (52.5) | 49 (56.3) | ||
| Tetraparesis | 6 (5.0) | 8 (12.3) | 5 (5.1) | 9 (10.3) | ||
| Aphasia, n (%) | 46 (38) | 26 (40) | 0.791 | 38 (38.4) | 34 (39.1) | 0.922 |
| Seizures | 22 (18.2) | 6 (9.2) | 0.104 | 15 (15.2) | 13 (14.9) | 0.968 |
| CT findings | ||||||
| Location, n (%) | ||||||
| Cerebral hemorrhage | 45 (37.2) | 30 (46.2) | 0.235 | 35 (35.4) | 40 (46.0) | 0.141 |
| Cerebellar hemorrhage | 19 (15.7) | 10 (15.4) | 0.995 | 16 (16.2) | 13 (14.9) | 0.819 |
| Intraventricular hemorrhage | 82 (67.8) | 58 (89.2) | 0.001 | 65 (65.7) | 75 (86.2) | 0.001 |
| Basal ganglia | 65 (53.7) | 34 (52.3) | 0.854 | 53 (53.5) | 46 (52.9) | 0.928 |
| Brainstem | 7 (5.8) | 13 (20.0) | 0.003 | 6 (6.1) | 14 (16.1) | 0.028 |
| Graeb score | 6 (1; 8) | 7 (4; 9) | 0.008 | 4 (0; 8) | 7 (4; 9) | 0.001 |
| Hematoma volume (cc), median (IQR) | 36 (20; 60) | 47 (30; 81) | 0.016 | 36 (20; 56.5) | 45 (27.5; 80) | 0.008 |
| Mass effect on CT, n (%) | 87 (71.9) | 55 (84.6) | 0.052 | 68 (68.7) | 74 (85.1) | 0.009 |
| Severity scales | ||||||
| APACHE II, median (IQR) | 14 (10; 17) | 18 (15.5; 22) | 0.000 | 12 (10; 16) | 18 (16; 22) | 0.0000 |
| MICH, median (IQR) | 2 (0; 3) | 2 (0; 4) | 0.187 | 2 (0; 3) | 2 (0; 4) | 0.106 |
| Laboratory test data | ||||||
| Hemoglobin [g/dl], median (IQR) | 13.5 (12.1; 15.0) | 13.4 (11.2; 15.0) | 0.664 | 13.7 (12.3; 15.0) | 13.4 (11.4; 15.0) | 0.317 |
| Blood glucose (mg/dl), median (IQR) | ||||||
| Upon admission | 142 (121; 189.5) | 179 (138; 213.5) | 0.007 | 146 (120.5; 189) | 163 (134; 211) | 0.004 |
| First 24h | 123 (109; 157) | 142 (116; 177) | 0.023 | 120 (107.5; 157.0) | 141 (116; 173) | 0.023 |
| Procedures, n (%) | ||||||
| Surgery | 42 (34.7) | 6 (9.2) | 0.000 | 35 (35.4) | 13 (14.9) | 0.002 |
| Ventricular drainage | 32 (26.4) | 19 (29.2) | 0.685 | 26 (26.3) | 25 (28.7) | 0.706 |
| Fibrinolysis | 8 (6.6) | 2 (3.1) | 0.498 | 5 (5.1) | 5 (5.7) | 0.999 |
| Invasive monitoring | ||||||
| ICP | 41 (33.9) | 10 (15.4) | 0.007 | 33(33.3) | 18 (20.7) | 0.054 |
| PTIO2 | 17 (14.0) | 4 (6.2) | 0.105 | 14 (14.1) | 7 (33.3) | 0.190 |
| Evolution | ||||||
| Complications, n (%) | ||||||
| Hydrocephalus | 55 (45.5) | 36 (55.4) | 0.196 | 42 (42.4) | 49 (56.3) | 0.059 |
| Brain edema | 84 (69.4) | 47 (72.3) | 0.681 | 67 (67.7) | 64 (73.6) | 0.380 |
| Seizures | 21 (17.4) | 3 (4.6) | 0.013 | 16 (16.2) | 8 (9.2) | 0.157 |
| Repeat bleeding | 23 (19.0) | 23 (35.4) | 0.014 | 18 (18.2) | 28 (32.2) | 0.027 |
| Days of stay, median (IQR) | ||||||
| ICU | 15 (9; 27.5) | 3 (1; 8.5) | 0.000 | 14 (7; 24) | 5 (2; 20) | 0.000 |
| Hospital | – | – | – | 46 (24; 76) | 5 (2; 29) | 0.005 |
APACHE, Acute Physiology and Chronic Health Evaluation; cc, cubic centimeters; GCS, Glasgow Coma Scale; H, hemorrhage; LTE, limitation of therapeutic effort; MICH, Modified Intracerebral Hemorrhage Score; ICP, intracranial pressure; PtiO2, brain tissue oxygenation pressure; IQR, interquartile range; CT, computed tomography.
Univariate Cox regression analysis referred to mortality in ICU and in hospital.
| Mortality | ||||||
|---|---|---|---|---|---|---|
| ICU | Hospital | |||||
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Age | 1.017 | 0.996; 1.039 | 0.106 | 1.027 | 1.007; 1.046 | 0.007 |
| Diabetes | 1.947 | 1.189; 3.188 | 0.008 | 1.879 | 1.210; 2.918 | 0.005 |
| Anticoagulants | 2.614 | 1.461; 4.679 | 0.001 | 2.813 | 1.658; 4.771 | <0.001 |
| GCS | 0.870 | 0.800; 0.945 | 0.001 | 0.896 | 0.836; 0.960 | 0.002 |
| Pupil reaction | ||||||
| Normal | 1 | Ref. | 1 | Ref. | ||
| Anisocoria | 2.071 | 1.182; 3.628 | 0.011 | 1.361 | 0.817; 0.279 | 0.237 |
| Mydriasis | 3.994 | 2.046; 7.797 | <0.001 | 3.376 | 1.857; 6.137 | <0.001 |
| Location | ||||||
| Intraventricular hemorrhage | 2.898 | 1.323; 6.350 | 0.008 | 2.681 | 1.423; 5.050 | 0.002 |
| Brainstem | 2.313 | 1.255; 4.262 | 0.007 | 1.887 | 1.062; 3.352 | 0.030 |
| Graeb score | 1.071 | 1.002; 1.144 | 0.042 | 1.084 | 1.024; 1.148 | 0.005 |
| Hematoma volume (cc) | 1.664 | 1.020; 2.713 | 0.041 | 1.004 | 0.999; 1.010 | 0.120 |
| APACHE II | 1.154 | 1.102; 1.210 | <0.001 | 1.151 | 1.105; 1.199 | <0.001 |
| Hemoglobin (g/dl) | 0.928 | 0.844; 1.020 | 0.123 | 0.914 | 0.839; 0.996 | 0.040 |
| Blood glucose upon admission (mg/dl) | 1.006 | 1.001; 1.010 | 0.009 | 1.001 | 0.998; 1.005 | 0.366 |
| Surgery | 0.229 | 0.099; 0.532 | 0.001 | 0.322 | 0.177; 0.585 | <0.001 |
| Ventricular drainage | 0.492 | 0.279; 0.869 | 0.014 | 0.621 | 0.382; 1.012 | 0.056 |
| Invasive monitoring, n (%) | ||||||
| ICP | 0.369 | 0.188; 0.725 | 0.004 | 0.484 | 0.286; 0.818 | 0.007 |
cc, cubic centimeters; GCS, Glasgow Coma Scale; H, hemorrhage; HR, hazard ratio; CI, confidence interval; p, significance level; ICP, intracranial pressure.
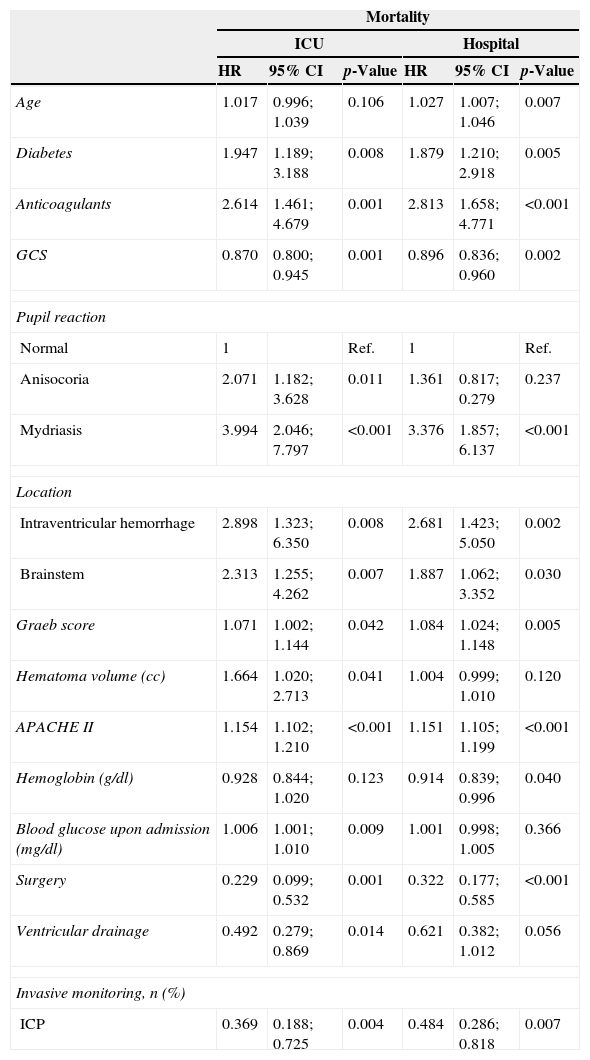
Table 4 shows the results of the multivariate Cox regression analysis of those variables found to be independently associated to mortality after ICH both in the ICU and during admission to hospital. Of note is the fact that the radiological findings associated to increased mortality in the ICU and in hospital were the presence of IVH, bleeding location at brainstem level, and the presence of radiological evidence of repeat bleeding. On the other hand, hematoma volume upon admission and the presence of a mass effect were associated to mortality in the ICU and in hospital, respectively.
Multivariate Cox regression (proportional hazards) analysis referred to mortality in the ICU and in hospital.
| Mortality | ||||||
|---|---|---|---|---|---|---|
| ICU | Hospital | |||||
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Diabetes mellitus | 2.629 | 1.515; 4.561 | 0.001 | 1.978 | 1.235; 3.167 | 0.005 |
| APACHE II | 1.137 | 1.081; 1.196 | <0.001 | 1.166 | 1.117; 1.217 | <0.001 |
| Pupil reaction | ||||||
| Normal | 1 | Ref. | ||||
| Anisocoria | 2.306 | 1.269; 4.191 | 0.006 | – | ||
| Mydriasis | 2.688 | 1.243; 5.814 | 0.012 | |||
| CT location | ||||||
| Intraventricular hemorrhage | 2.358 | 1.323; 6.350 | 0.047 | 3.215 | 1.549; 6.673 | 0.002 |
| Brainstem | 3.734 | 1.807; 7.716 | <0.001 | 2.263 | 1.234; 4.152 | 0.008 |
| Hematoma volume (cc) | 1.992 | 1.147; 3.461 | 0.014 | – | – | |
| Surgery | 0.295 | 0.170; 0.510 | <0.001 | 0.348 | 0.184; 0.656 | <0.001 |
| Monitoring ICP | 0.374 | 0.184; 0.760 | 0.007 | 0.358 | 0.203; 0.630 | <0.001 |
cc, cubic centimeters; H, hemorrhage; HR, hazard ratio; CI, confidence interval; p, significance level; ICP, intracranial pressure; CT, computed tomography.
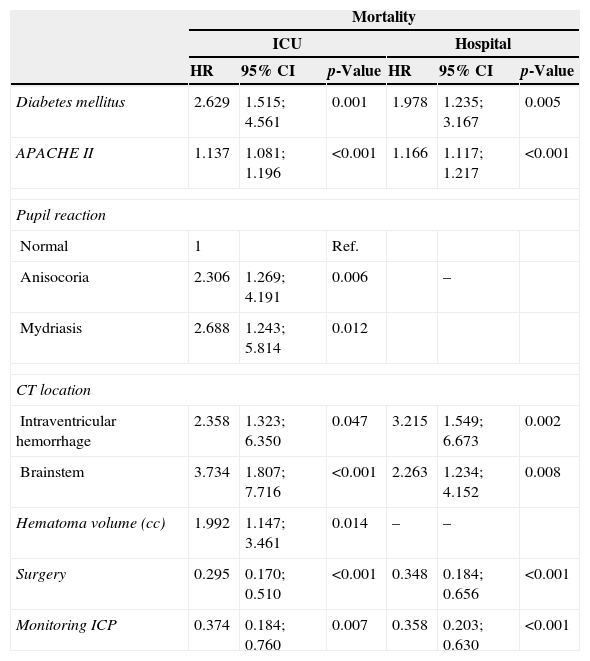
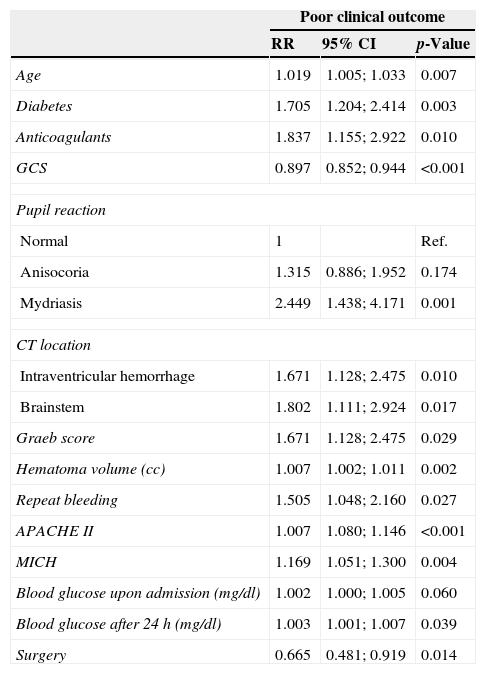
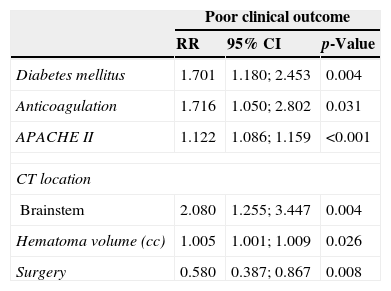
Lastly, the functional evaluation of the patients after 6 months is shown in Table 5. The multivariate analysis corresponding to Table 6 shows the presence of diabetes or previous anticoagulant treatment and the type of hemorrhage on the CT scan to be predictors of mortality and of poor functional outcome, while surgery was associated to increased survival and better functional outcomes.
Univariate Cox regression analysis referred to poor functional outcome (modified Rankin score ≥3) at 6 months.
| Poor clinical outcome | |||
|---|---|---|---|
| RR | 95% CI | p-Value | |
| Age | 1.019 | 1.005; 1.033 | 0.007 |
| Diabetes | 1.705 | 1.204; 2.414 | 0.003 |
| Anticoagulants | 1.837 | 1.155; 2.922 | 0.010 |
| GCS | 0.897 | 0.852; 0.944 | <0.001 |
| Pupil reaction | |||
| Normal | 1 | Ref. | |
| Anisocoria | 1.315 | 0.886; 1.952 | 0.174 |
| Mydriasis | 2.449 | 1.438; 4.171 | 0.001 |
| CT location | |||
| Intraventricular hemorrhage | 1.671 | 1.128; 2.475 | 0.010 |
| Brainstem | 1.802 | 1.111; 2.924 | 0.017 |
| Graeb score | 1.671 | 1.128; 2.475 | 0.029 |
| Hematoma volume (cc) | 1.007 | 1.002; 1.011 | 0.002 |
| Repeat bleeding | 1.505 | 1.048; 2.160 | 0.027 |
| APACHE II | 1.007 | 1.080; 1.146 | <0.001 |
| MICH | 1.169 | 1.051; 1.300 | 0.004 |
| Blood glucose upon admission (mg/dl) | 1.002 | 1.000; 1.005 | 0.060 |
| Blood glucose after 24h (mg/dl) | 1.003 | 1.001; 1.007 | 0.039 |
| Surgery | 0.665 | 0.481; 0.919 | 0.014 |
APACHE, Acute Physiology and Chronic Health Evaluation; cc, cubic centimeters; GCS, Glasgow Coma Scale; CI, confidence interval; MICH, Modified Intracerebral Hemorrhage Score; p, significance level; RR, relative risk; CT, computed tomography.
Multivariate Cox regression (proportional hazards) analysis referred to poor functional outcome (modified Rankin score ≥3) at 6 months after cerebral hemorrhage.
| Poor clinical outcome | |||
|---|---|---|---|
| RR | 95% CI | p-Value | |
| Diabetes mellitus | 1.701 | 1.180; 2.453 | 0.004 |
| Anticoagulation | 1.716 | 1.050; 2.802 | 0.031 |
| APACHE II | 1.122 | 1.086; 1.159 | <0.001 |
| CT location | |||
| Brainstem | 2.080 | 1.255; 3.447 | 0.004 |
| Hematoma volume (cc) | 1.005 | 1.001; 1.009 | 0.026 |
| Surgery | 0.580 | 0.387; 0.867 | 0.008 |
APACHE, Acute Physiology and Chronic Health Evaluation; cc, cubic centimeters; CI, confidence interval; p, significance level; RR, relative risk; CT, computed tomography.
In our series, ICH was associated to high mortality and poor functional outcome. A history of diabetes, previous anticoagulation treatment, the location and volume of the hematoma, and patient severity upon admission were associated to poor patient prognosis, while monitoring of ICP or the application of surgical treatment was associated to improved outcome.
In recent decades the incidence of ICH has dropped by up to 31%.2 We consider that this can be explained by improved control of arterial hypertension, though the mortality rate remains high and the functional outcomes are still poor, as evidenced by our own findings. This situation in turn is probably attributable to the increased use of anticoagulants and antiplatelet drugs.1 Recent studies have found no changes in immediate or long-term mortality despite advances in primary prevention, adherence to the current clinical management guides and the increasingly widespread practice of treating these patients in specialized neurocritical units.6,19
In our series the mortality rates were consistent with those reported in the literature. The systematic review and metaanalysis published by Van Asch et al.4 documented a mortality rate of 35–52% after 30 days, and one-half of these deaths occurred in the first 48–72h after the bleeding event. In our series mortality was in the upper range (46.7%), probably due to a number of reasons. Firstly, our sample exclusively consisted of critical patients requiring admission to the ICU. Secondly, the median age of our patients was 60 years, and in this regard it is known that mortality in the 45–59 years age interval is even higher than among patients over 75 years of age.2 The growing use of anticoagulants in younger populations as treatment for their background diseases or for prophylactic purposes has been shown to be an independent risk factor for mortality in ICH, and is associated to expansion of the hematoma or the recurrence of ICH.7,20 In our series, 23.1% of the patients had received prior antiplatelet medication, and 13.4% were receiving oral anticoagulants. On the other hand, the duration of follow-up in our study was longer than in other series. Most publications analyze mortality after 30 days. In our study we extended this period, assessing also usually occult mortality occurring after discharge from the ICU and frequently beyond the mentioned 30-day interval. In our case this mortality rate was 11.8%, which would also explain our higher mortality versus other series. However, methodological reasons precluded a more exhaustive and isolated analysis of this subgroup.
In concordance with other studies, a total of 66.7% of the patients included in our series had high blood pressure–the latter being identified as the leading risk factor for ICH. Recent studies have underscored the importance of adequate blood pressure control in these patients, though such control has only been shown to offer benefits in terms of functional outcome, with no demonstrated impact upon mortality–in coincidence with our own observations.5
On relating the study variables to mortality in the ICU and in hospital, diabetic patients were seen to have a twofold higher risk of death, in coincidence with the observations in other neurocritical conditions.21 Other independent mortality factors in both cases were the APACHE II severity score upon admission, the presence of IVH, and hematomas located at brainstem level. In contrast, mortality was found to be lower in patients subjected to surgical treatment or with ICP monitoring–both of these factors being found to exert a protective effects against mortality. One of the main mechanisms implicated in mortality and in functional outcome after ICH refers to hematoma expansion in the first 24h. In this regard, prompt surgical intervention could have an important impact upon outcome. On the other hand, although invasive multimodal neuromonitoring was initially designed for use in patients with severe traumatic brain injury, we consider that it may offer useful information for the management of neurocritical patients in general.19,22,23 Close monitoring of the neurological situation would allow early therapeutic actions and interventions, and thus could improve the functional outcome, as suggested by our findings referred to invasive ICP monitoring.
Many studies have evaluated the risk/benefit ratio of early surgical treatment (in the first 12h) versus initial conservative management.13 In this regard, the STICH trial reported no benefit of surgery versus medical treatment.3 Probably in this case the time from ICH symptoms onset to surgery was too long to avoid expansion of the hematoma (median time 30h). On the other hand, 26% of the patients assigned to the conservative treatment arm subsequently experienced neurological worsening and were crossed over to the surgical treatment arm–increasing the median time to 60h.3 In coincidence with our own findings, a metaanalysis published in 2008, with the inclusion of 10 articles, documented a lesser mortality and dependency risk in those patients who underwent surgery.11 Recently, the STICH-2 trial, designed to compare the results of early surgery versus conservative management in patients with lobar hematomas,14 likewise recorded no benefit from surgery following ICH. Probably these results were due to the fact that the patients had a normal level of consciousness and specifically presented lobar bleeding; there consequently was no intracranial hypertension, in contrast to our own series, where the patients probably would benefit from surgery. In any case, it should be noted that the European guides on the management of ICH support the existence of improved functional outcomes in patients subjected to neurosurgical treatment.24
In coincidence with Ruijiun et al.,8 we identified a number of variables related to functional outcome after 6 months: diabetes, the APACHE II score upon admission, initial hematoma volume, and hematoma location at brainstem level. In turn, we identified anticoagulation as a risk factor, in coincidence with other Spanish series, and found early hematoma evacuation to improve the patient prognosis.20
Our study has a number of limitations, including its single-center observational design. Nevertheless, our hospital is a third-level reference center for neurocritical patients. On the other hand, despite adherence to the clinical practice guides, the criterion for surgery in some cases was decided by the neurosurgical team on duty. In turn, the number of deaths during hospital stay, after passing through the ICU, did not allow exhaustive analysis of the factors associated to occult mortality. Lastly, we did not collect precise information on the type of surgical technique employed, and did not include signs of active intracranial bleeding among the radiological criteria.25 Nevertheless, we wish to point out that these aspects will be addressed in future studies. At present, we are conducting a multicenter trial designed to include these variables and contrast the findings obtained.
As strong points of our study, we followed-up on patients at discharge from the ICU and from hospital. In this respect, some studies limit analysis to a 30-day time window, thus preventing the obtainment of data on the evolution of the patients after they leave the hospital center. In turn, although patient recruitment was conducted on a single-center basis and during the period of the study, we consider that the sample size was adequate for a descriptive and exploratory analysis of the characteristics of our population.
ConclusionsIn our series, ICH remains a disease with high mortality and poor patient functional outcome. The presence of comorbidities such as diabetes, or previous treatment with anticoagulants, as well as the type of brain CT findings (bleeding at brainstem or intraventricular level, and the presence of repeat bleeding, among others) are predictors of mortality and poor functional outcome, while ICP monitoring and early surgical treatment are associated to improved survival and better functional outcome. In any case, we consider that further research is needed, with the conduction of a multicenter trial, in order to validate our findings and subsequently assess strategies designed to improve the management of patients with severe ICH.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Ferrete-Araujo AM, Egea-Guerrero JJ, Vilches-Arenas Á, Godoy DA, Murillo-Cabezas F. Predictores de mortalidad y mal resultado funcional en la hemorragia intraparenquimatosa espontánea grave: estudio prospectivo observacional. Med Intensiva. 2015;39:422–432.