To investigate the applications and effects of neuromuscular electrical stimulation (NMES) in critically ill patients in ICU by means of a systematic review.
Materials and methodsElectronic searches were conducted in the databases Medline, CINAHL, Cochrane Central Register of Controlled Trials, Web of Science, Embase, ProQuest Health and Medical Complete, AMED, and PEDro. The PEDro score was used to assess the methodological quality of the eligible studies.
ResultsThe search yielded a total of 9759 titles and nine articles satisfied the eligibility criteria. These studies showed that NMES can maintain or increase muscle mass, strength and volume, reduce time in mechanical ventilation and weaning time, and increase muscle degradation in critically ill patients in ICU. Two studies allowed a meta-analysis of the effects of NMES on quadriceps femoris strength and it showed a significant effect in favor of NMES in the Medical Research Council (MRC) Scale (standardized mean difference 0.77 points; p=0.02; 95% CI: 0.13–1.40).
ConclusionsThe selected studies showed that NMES has good results when used for the maintenance of muscle mass and strength in critically ill patients in ICU. Future studies with high methodological quality should be conducted to provide more evidence for the use of NMES in an ICU setting.
Investigar las aplicaciones y los efectos de la estimulación eléctrica neuromuscular (EENM) en pacientes en estado crítico en la UCI a través de una revisión sistemática.
Materiales y métodosSe hicieron búsquedas electrónicas en las bases de datos MEDLINE, CINAHL, Registro Cochrane Central de Ensayos Controlados, Web Of Science, Embase, ProQuest Health and Medical Complete, AMED y PEDro. La escala PEDro se utilizó para evaluar la calidad metodológica de los estudios elegibles.
ResultadosLa búsqueda arrojó un total de 9.759 títulos, y 9 artículos cumplían los criterios de elegibilidad. Estos estudios demostraron que la EENM puede mantener o aumentar la masa muscular, la fuerza y el volumen, reducir el tiempo de ventilación mecánica y el tiempo de destete, y aumentar la degradación muscular en pacientes en estado crítico en la UCI. Dos estudios permitieron un metaanálisis de los efectos de la EENM en la fuerza del cuádriceps femoral y mostraron un efecto significativo a favor de la EENM en la escala Medical Research Council (diferencia media estandarizada de 0,77 puntos; p=0,02; IC 95%: 0,13-1,40).
ConclusionesLos estudios seleccionados mostraron que la EENM tiene buenos resultados cuando se utiliza para el mantenimiento de la masa y la fuerza muscular en pacientes en estado crítico en la UCI. Futuros estudios con alta calidad metodológica deben llevarse a cabo para proporcionar más pruebas para el uso de la EENM en la UCI.
Critically ill patients in intensive care units (ICUs) are subject to various complications resulting from the underlying disease and from being immobilized.1–3 A few examples of these complications are systemic inflammation, atelectasis, metabolic and vascular dysfunction, joint contracture, pressure ulcers, and loss of muscle mass.1–3 The reduction in muscle mass is one of the most debilitating complications in critically ill patients and it hinders their recovery after discharge from ICU due to loss of function.4,5 Many factors contribute to muscle mass reduction in the critical patients, e.g. use of medicines, presence of sepsis, prolonged mechanical ventilation (MV), and bed rest.1,6,7 One study investigated the effects of 7 days of restricted bed rest in healthy individuals and found a significant reduction of 3% in thigh muscle volume using magnetic resonance imaging.2 Therefore, preventing loss of muscle mass in critical patients is one of the main objectives of ICU professionals.4 Several therapeutic measures are employed to that end, including range of motion exercises, positioning, and resisted exercises.4
One of the techniques used in ICUs to stimulate muscle function is neuromuscular electrical stimulation (NMES). It is widely used in the rehabilitation of patients who need to maintain or increase muscle mass, strength and function, and it has shown promising results in the rehabilitation of immobilized muscles.8,9 By definition, NMES is the application of non-invasive sensitive electrical stimulation that causes muscle contraction independent of the patient's effort, i.e. without the need for neural stimuli for the recruitment of muscle fibers.10 However, there is no consensus on whether NMES alone can increase muscle strength. Nevertheless, some positive results from the technique have been found when it was applied to the muscles of individuals with spinal cord injury.11–13
Thus, NMES can be used both in clinical practice and in a hospital setting to increase muscle mass and strength and to increase tolerance to future exercise.14 For patients in ICU specifically, NMES has been suggested to improve microcirculation and to minimize the deleterious effects of prolonged bed rest, thereby preventing the development of neuromuscular complications after recovery from the underlying disease.15 Because patients in ICU are extremely debilitated, the use of NMES must have a solid theoretical basis that is grounded on quality studies so that the team involved in the treatment can safely apply the technique.10 Therefore, the aim of the present study was to investigate the applications and effects of neuromuscular electrical stimulation in critically ill patients in intensive care by means of a systematic review.
Materials and methodsThis systematic review was carried out in accordance with PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) recommendations16 by five investigators, two of whom have large experience in conducting systematic reviews.
Eligibility criteriaFor this systematic review, we included randomized and quasi-randomized controlled trials that assessed the effects of non-invasive NMES applied to lower and/or upper limbs of critical patients in ICU. No limit was placed with regard to time of publication or original language. The studies that used NMES outside ICU or in individuals under age of 18 were excluded. We also excluded studies that applied NMES to patients admitted to ICU for less than 48h.
Search strategyAn electronic search was conducted in the databases Medline via OVID, CINAHL (Cumulative Index to Nursing and Allied Health Literature) via EBSCO, Cochrane Central Register of Controlled Trials, Web of Science, Embase via OVID, ProQuest Health and Medical Complete, AMED (Allied and Complementary Medicine Database) via EBSCO, and PEDro (Physiotherapy Evidence Database). The searches were conducted until November 26th, 2013.
The search filters developed by the Scottish Intercollegiate Guidelines Network (SIGN, http://www.sign.ac.uk/methodology/filters.html) for searching randomized clinical trials were combined with the strategy developed by the authors to find studies on NMES in critical patients in ICU. This strategy was used in all databases and was adapted to the specific characteristics of each database (Table 1).
Medline search via Ovid.a
| 1. | Randomized Controlled Trials as Topic/ |
| 2. | randomized controlled trial/ |
| 3. | Random Allocation/ |
| 4. | Double Blind Method/ |
| 5. | Single Blind Method/ |
| 6. | clinical trial/ |
| 7. | clinical trial, phase i.pt |
| 8. | clinical trial, phase ii.pt |
| 9. | clinical trial, phase iii.pt |
| 10. | clinical trial, phase iv.pt |
| 11. | controlled clinical trial.pt |
| 12. | randomized controlled trial.pt |
| 13. | multicenter study.pt |
| 14. | clinical trial.pt |
| 15. | exp Clinical Trials as topic/ |
| 16. | or/1–15 |
| 17. | (clinical adj trial$).tw |
| 18. | ((singl$ or doubl$ or treb$ or tripl$) adj (blind$3 or mask$3)).tw |
| 19. | PLACEBOS/ |
| 20. | placebo$.tw |
| 21. | randomly allocated.tw |
| 22. | (allocated adj2 random$).tw |
| 23. | or/17–22 |
| 24. | 16 or 23 |
| 25. | case report.tw |
| 26. | letter/ |
| 27. | historical article/ |
| 28. | or/25–27 |
| 29. | not 28 |
| 30. | Critical Illness/ |
| 31. | critic$ adj2 (patient or ill$ or care).mp |
| 32. | Critical Care/ |
| 33. | APACHE/ |
| 34. | Terminally Ill/ |
| 35. | Intensive Care Units/ |
| 36. | ICU.mp |
| 37. | intensive adj3 care.mp |
| 38. | Chronic Disease/ |
| 39. | Coma/ |
| 40. | Multiple Trauma/ |
| 41. | Pulmonary Disease, Chronic Obstructive/ |
| 42. | Respiratory Insufficiency/ |
| 43. | Polyneuropathies/ |
| 44. | Pneumonia/ |
| 45. | Respiration, Artificial/ |
| 46. | Positive-Pressure Respiration/ |
| 47. | Respiratory Therapy/ |
| 48. | Immobilization/ |
| 49. | Intubation, Intratracheal/ |
| 50. | or/30–49 |
| 51. | 29 and 50 |
| 52. | Electric Stimulation Therapy/ |
| 53. | Electric Stimulation/ |
| 54. | electr$ adj2 stimulation.mp |
| 55. | EMS.mp |
| 56. | Muscle Contraction/ |
| 57. | Muscle Weakness/ |
| 58. | Musculoskeletal Development/ |
| 59. | muscle adj2 strength.mp |
| 60. | muscle adj (recruit$ or activ$).mp |
| 61. | (muscle or neur$) adj2 stimul$.mp |
| 62. | early adj3 mobili$.mp |
| 63. | Electricity/ |
| 64. | Transcutaneous Electric Nerve Stimulation/ |
| 65. | (interferential or russian) adj current.mp |
| 66. | neuromuscular electrical stimulation.mp |
| 67. | NMES.mp |
| 68. | functional electric stimulation.mp |
| 69. | FES.mp |
| 70. | or/52–69 |
| 71. | 51 and 70 |
After searching the databases, two independent assessors selected the articles based on the titles and then the abstracts. Disagreements between the assessors were resolved by consensus. When no consensus could be reached, a third assessor was consulted to decide on the eligibility of the study. Only studies that would potentially satisfy the inclusion criteria were analyzed in full.
Assessment of the methodological quality of the studiesThe selected articles were assessed according to the PEDro scale.17–19 Two assessors analyzed the selected articles and generated an independent score that were later compared. In cases where the final score was different between assessors, a third assessor was consulted to generate the final score on the PEDro scale. This is an 11-item scale that assesses the methodology of randomized controlled trials (internal validity and statistical information). Each item is worth one point with the exception of the first item, which is not scored, for a total of 10 points. For this review, scores over 7 were considered high methodological quality; 6 and 5, intermediate; and 4 or less, low quality.
Data extraction and analysisThe data extraction and analysis were conducted by at least two authors independently. When similarities were found between studies regarding intervention, patient characteristics and analyzed variables, a meta-analysis was conducted. The data for mean and standard deviation of the studies included in the meta-analysis were extracted and converted into weighted mean differences (treatment effect) and 95% confidence intervals (95% CI). The statistical homogeneity of the studies was assessed using the I2 value and, considering that for the only possible meta-analysis the I2 value was over 25%, the meta-analysis was performed using the random effects model in the software package Review Manager (RevMan) (Version 5.2, Copenhagen, Denmark).20 The studies that did not allow a meta-analysis were qualitatively analyzed considering only comparisons between groups and when it was possible, central tendency data (mean and median), dispersion data (standard deviation, interquartile range [IR] and minimal–maximal score [range]), and comparison data (95% CI and p value) were extracted.
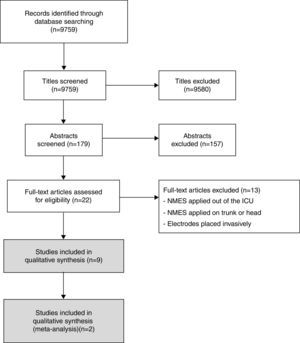
ResultsThe search yielded 9759 titles and nine articles satisfied the eligibility criteria for inclusion in the present review21–29 (Fig. 1). After the studies were analyzed, it was ascertained that the studies by Bouletreau et al.21,22 refer to the same research. Therefore, we analyzed the results of these studies as if they were a single article. The study by Karatzanos et al.25 is a post hoc analysis of the study by Routsi et al.28; however they have different variables and were analyzed separately, except for meta-analysis.
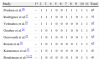
Thus, the present review analyzed the results of eight studies. According to the PEDro scale, two studies ranked as high methodological quality, four with intermediate quality and two with low methodological quality. Although two studies had high methodological quality, most of the studies had intermediate to low methodological quality (PEDro scale≤5 points) (Table 2).
Classification of articles in the PEDro Scale.
| Study | 1* | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | Total |
| Poulsen et al.26 | – | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 8a |
| Rodriguez et al.27 | – | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 1 | 7a |
| Velmahos et al.29 | – | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 5b |
| Gruther et al.24 | – | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 5b |
| Gerovasili et al.23 | – | 1 | 0 | 1 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 5b |
| Rousti et al.28 | – | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 5b |
| Karatzanos et al.25 | – | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 4c |
| Bouletreau et al.21,22 | – | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 4c |
Items PEDro Scale: (1) eligibility criteria were specified (* – this item is not used to calculate the PEDro score); (2) subjects were randomly allocated to groups; (3) allocation was concealed; (4) the groups were similar at baseline regarding the most important prognostic indicators; (5) there was blinding of all subjects; (6) there was blinding of all therapists who administered the therapy; (7) there was blinding of all assessors who measured at least one key outcome; (8) measures of at least one key outcome were obtained from more than 85% of the subjects initially allocated to groups; (9) all subjects for whom outcome measures were available received the treatment or control condition as allocated or, where this was not the case, data for at least one key outcome was analyzed by “intention to treat”; (10) the results of between-group statistical comparisons are reported for at least one key outcome; (11) the study provides both point measures and measures of variability for at least one key outcome.
Abbreviation: 1=item satisfied; 0=item not satisfied.
The analyses and description of the studies included in this review are shown below and in Table 3.
Summary of the studies included in the review.
| Study | Patients | Number of participantsa | Neuromuscular electrical stimulation | Outcomes | ||||||
| Treatment | Location of the electrodes | PW (μs) | F (Hz) | On (s) | Off (s) | Intensity | ||||
| Poulsen et al.26 | Septic shock | IG=8CG=8b | 60min a day for 7 days | Vastus medialis and lateralis and 5cm inguinal fold | 300 | 35 | 4 | 6 | Visible contraction | Volume of the quadriceps femoris muscle (CT) |
| Velmahos et al.29 | Major trauma | IG=26CG=21 | 30min, twice a day for 7–14 days | Hamstrings and thighs bilaterally | 300 | 1.75 | 5 | 5 | Visible contraction | Presence of DVT, velocity of venous blood flow and maximum venous diameter |
| Gruther et al.24 | Polytrauma, cardiovascular disease, transplantation, pneumonia and cancer:1 – admitted for less than one week2 – admitted for less than two weeks | 1IG=8CG=92IG=8CG=8* | 30min a day, 5 sessions a week for a period of 4 weeks. From the 2nd week, 60min. | Quadriceps and 5cm distal to the inguinal fold | 350 | 50 | 8 | 24 | IG: Maximum tetanic contraction of the quadricepsCG: stimulation in visible or palpable contraction. | Layer thickness of the quadriceps muscle (US)* |
| Gerovasili et al.23 | Sepsis, trauma and neurological diseases | IG=13CG=13 | 55min a day for 7 days | Quadriceps and fibularis longus, bilaterally | 400 | 45 | 12 | 6 | Visible contraction | Cross-sectional diameter of the quadriceps muscle (US)* |
| Rodriguez et al.27 | Sepsis | IG=14CG=14b | 30min, twice a day until extubation | Biceps brachii and vastus medialis, unilaterally | 300 | 100 | 2 | 4 | Visible contraction | Quadriceps and biceps muscle strength (MRC)*; thigh and leg circumference; thickness of biceps brachii (US) |
| Bouletreau et al.21,22 | Acute cerebral infarction and postoperative ventilatory failure | IG=10CG=10c | 30min, twice a day for 4 days | Hamstrings and thighs | 300 | 1.75 | 5 | 5 | Visible contraction | Elimination of nitrogen, urea, creatinine* and 3-methyl histidine* |
| Rousti et al.28 | Sepsis, trauma, brain injury, respiratory failure and post-surgical | IG=24CG=28 | 55min daily until discharge from ICU | Vastus medialis, fibularis lateralis and longus, bilaterally | 400 | 45 | 12 | 6 | Visible contraction | Muscle strength (MRC)*; development of polyneuromyopathy; duration of MV*; weaning time*; days without MV*; time spent in ICU |
| Karatzanos et al.25 | Sepsis, trauma, brain injury, respiratory failure and post-surgical | IG=24CG=28 | 55min daily until discharge from ICU | Vastus medialis, fibularis lateralis and longus, bilaterally | 400 | 45 | 12 | 6 | Visible contraction | Muscle strength (MRC)*; hand grip |
Abbreviations: PD=pulse width; F=frequency; On=time with stimulation in each cycle; Off=pause between cycles; IG=intervention group; CG=control group; CT=computerized tomography; DVP=deep vein thrombosis; US=ultrasound; MRC=Medical Research Council scale; MV=mechanical ventilation.
Three studies investigated muscle strength after NMES application by using the Medical Research Council (MRC) scale and all three found an increase in muscle strength for the NMES group.25,27,28 Rodriguez et al.27 found an increase in the MRC score for the quadriceps on the day of extubation on the side that received NMES compared to the side that did not receive it (median 3 points, IR 3–4 vs median 3 points, IR 2–3, respectively; p=0.034) and for the biceps brachii (median 4 points, IR 3–4 vs median 3 points, IR 2–4, respectively; p=0.005). The study by Rousti et al.28 reported a significant difference for MRC (whole body measurement) between NMES group and control group (median 58 points, range 33–60 vs median 52 points, range 2–60, respectively; p=0.04). Also, Karatzanos et al.25 complemented the data by Rousti et al.28 by presenting the IR (NMES group: 51–60; control group: 40–58).
Muscle strength meta-analysisThe meta-analysis was conducted with the outcome of NMES on quadriceps femoris assessed using the MRC scale for muscle strength. The MRC is a widely used scale for assessing strength in critically ill patients.30–32 It consists of six points (0–5), where 0 refers to total absence of muscle contraction and 5 refers to active movement against gravity and great resistance. The MRC score is usually calculated by adding up the points for the following movements: shoulder abduction, elbow flexion, wrist flexion, hip flexion, knee extension, and ankle dorsiflexion. This generates a maximum score of 60 points; however, it is common to use the scores separately for each joint.30–32
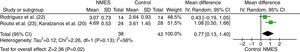
The effect of NMES on strength was verified in the studies by Rodriguez et al.,27 Karatzanos et al.,25 and Routsi et al.28 (Fig. 2). The studies by Routsi et al.28 and Karatzanos et al.25 show the data for both limbs submitted to stimulation. For the meta-analysis, the data were randomized to obtain results from one limb per participant. The study by Rodriguez et al.27 compared the stimulated side to the non-stimulated side. For the meta-analysis, the sides were considered as independent groups. The data were obtained from the authors. The meta-analysis pooled data from 66 participants and showed a significant effect in favor of the group that received NMES over the control group, with a standardized mean difference of 0.77 (p=0.02; 95% CI: 0.13–1.40; Fig. 2).
Meta-analysis of the effect of neuromuscular electrical stimulation (NMES) on quadriceps femoris strength assessed by the Medical Research Council (MRC) scale. Abbreviations: SD=standard deviation; IV=inverse variance method; Random=random effects model; CI=confidence interval; df=degrees of freedom.
Six studies presented data on the effects of NMES on muscle structure. Poulsen et al.26 investigated the decrease in muscle volume and found that NMES did not prevent such decrease when compared to the contra-lateral muscle group. Similarly, Rodriguez et al.27 did not find any difference between sides after one side received NMES, measured by ultrasound and limb circumference. Gruther et al.24 used only the ultrasound to evaluate muscle thickness after NMES application and found no difference between the NMES group and the control group in the short term.
Other studies found some significant differences between groups for other muscle structure variables.21–23 Gerovasili et al.23 investigated the diameter for the rectus femoris and the vastus intermedius of both limbs by means of ultrasound and found that there was a smaller decrease in diameter for the NMES group when compared to a control group for all muscles, except for the left rectus femoris (−0.13±0.10cm vs −0.19±0.16cm, respectively; p=0.07).
Bouletreau et al.21,22 measured the level of muscle degradation during NMES application periods and compared it to a control period by monitoring the elimination of urea, nitrogen balance, creatinine, and 3-methyl histidine. They reported a smaller elimination during NMES application only for creatinine (79.2±25μmol/kg/day vs 92.4±6.8μmol/kg/day, respectively; p<0.01) and 3-methyl histidine (3.15±0.32μmol/kg/day vs 3.78±0.37μmol/kg/day, respectively; p<0.01).
Time in intensive care and in mechanical ventilationOnly one study investigated the effect of NMES on the time spent in ICU and in MV. Rousti et al.28 reported no difference between a NMES group and a control group for the variables average time spent in ICU and average time in MV; however, they reported a better performance for the weaning period in the NMES group when compared to the control group (median 1 day, range 0–10 vs 3 days, range 0–44, respectively; p=0.003) and a shorter period between effective extubation until ICU discharge (days off MV) for the NMES group than the control group (median 4 days, range 0–16 vs 6 days, range 0–41, respectively; p=0.003).
Complications from immobilization and bed restVelmahos et al.29 investigated the effectiveness of NMES in preventing deep vein thrombosis (DVT) in patients with major trauma and not under heparinization and found no difference between groups for DVT occurrence. They also investigated the venous flow velocity and venous diameter and found a higher venous flow velocity for the NMES group when compared to the control group in the superficial femoral left vein (21±6cm/min vs 16±5cm/min, respectively; p=0.02) and in the left popliteal vein (22±10cm/min vs 15±9cm/min, respectively; p=0.03). Rousti et al.28 compared the development of critical illness polyneuropathy between the NMES group and the control group and found an odds ratio=0.22 (95% CI=0.05–0.92; p=0.04) in favor of the NMES group.
DiscussionIn the nine selected studies, we found that NMES can be applied to different kinds of patients and objectives within the ICU. Among these, NMES was used in patients with septic shock to maintain muscle volume and strength, trauma patients to prevent DVT, and critical patients to verify: (a) the effects of NMES on muscle mass in short and long-term hospital stay; (b) the deleterious effects of NMES on biological markers related to muscle degradation; and (c) its benefits to muscle functionality and prevention of polyneuromyopathy.21–29
The methodological quality of the selected articles shows the lack of high-quality studies that measure the effects of NMES applied to critically ill patients. The best studies were the ones by Poulsen et al.26 and Rodriguez et al.27 They did not reach the highest possible score due to the difficulty in blinding the therapist and the patient. Additionally, the study by Rodriguez et al.27 did not report whether the analysis was by intention to treat. The studies with the lowest ranking were the studies by Karatzanos et al.25 and Bouletreau et al.,21,22 which scored 4 on the PEDro scale and were considered to have low methodological quality. The study by Karatzanos et al.25 only received points for random allocation to groups, similarity at baseline, between-group comparison after intervention, and presentation of measures of variability for at least one key outcome. The study by Bouletreau et al.21,22 was different from the study by Karatzanos et al.25 only because it satisfied the requirement of measures of at least one key outcome obtained from more than 85% of the subjects initially allocated to groups; however it did not meet the requirement of similarity between groups at baseline.
Regarding the form of application, different methods were used with no particular pattern in the selection of the modulation parameters of the NMES. Frequency ranged from 1.75Hz to 100Hz, with pulse duration varying from 300 and 400μs. According to Kesar and Binder-Macleod,33 the least muscle fatigue is produced when medium frequency (around 30Hz) and medium pulse duration (150±21μs) are used. Most of the selected studies used frequencies close to the range suggested by Kesar and Binder-Macleod33; however, the pulse durations were at least twice as long. This may explain why some of the studies did not find positive results after NMES due to the fatigue it produced.
Just as in the frequency and pulse duration parameters, the times of application also had high variability, ranging from 30 to 60min, one or two times a day. Additionally, NMES was applied 4–5 days a week, and the treatment varied from 1 week to discharge from ICU or effective extubation. Electrode positioning did not follow a pattern, with unilateral positioning in some studies,21,22,24,26 bilateral positioning in lower limbs in others23,25,28,29 or in lower and upper limbs unilaterally.27 Regarding stimulation intensity, with the exception of the study by Gruther et al.24 who applied NMES until a maximum tetanic contraction was achieved, all studies used NMES with sufficient intensity to obtain at least visible contraction during application. As there was important methodological heterogeneity among the studies, it seems that the same heterogeneity was reflected in statistical terms, as the value for I2 (56%) showed high statistical heterogeneity in the meta-analysis. This could be interpreted as an indication of lack of agreement among researchers on what is important in terms of procedures and outcome measures when investigating the effects of NMES in critically ill patients.
According to the data obtained from the selected studies, NMES presents good results when used to maintain lower limb muscle volume. Two studies assessed muscle mass by ultrasound and concluded that NMES preserves the muscle mass of critical patients on bed rest.23,24 Furthermore, Poulsen et al.26 used computerized tomography and found the least amount of loss of muscle mass in the stimulated limb compared to the non-stimulated limb in the same patient. While assessing peripheral muscle strength using the MRC scale, three studies showed significant improvement in global muscle strength with the use of NMES.25,27,28 This technique showed effect for maintenance of muscle mass and strength, but the study by Bouletreau et al.21,22 showed that electrical stimulation in patients on bed rest must be used with caution as it influences the degradation of muscle protein. However, the study by Bouletreau et al.21,22 used the lowest frequency of all the studies (1.75Hz), which may have led to greater fatigue than the others.
Also, it is unclear whether there is a relationship between muscle strength of limbs and MV. However, the results from De Jonghe et al.31 raise the question whether such relationship actually exists, as they present a relationship between limb muscle strength and respiratory muscle strength; also, they show a relationship between respiratory muscle strength and time in MV for critically ill patients. Therefore, it is possible that muscle weakness in lower limbs may represent the need of longer stay in MV. Among the included studies, only Routsi et al.28 investigated the effects of NMES on muscle strength and also looked at time in MV. They found positive effects for the NMES group in regards to MV and weaning period. However, there was a large variability for the control group in regards to weaning period (median 3 days, range 0–44) and time between extubation and ICU discharge (median 6 days, range 0–41). Thus, this large variability seen in the control group could have affected the statistical comparisons, reinforcing the need for further investigation.
Among the selected studies, two used the MRC scale to verify the effect of NMES on the strength of these patients, allowing a meta-analysis of this outcome for the quadriceps femoris muscle.25,27,28 The meta-analysis included one study with high methodological quality27 and another with intermediate quality25,28 and showed a significant effect in favor of the group that received NMES over the control. This shows that NMES was able to maintain or increase quadriceps strength in critically ill patients in ICU. Although the MRC scale uses ordinal numbers, the studies included in the meta-analysis used continuous data. For that reason and because of the difficulty in dichotomizing the MRC data, continuous data were also used in the meta-analysis.34
A possible limitation of the meta-analysis is the statistical approach used for the study by Rodriguez et al.27 A non-parametric test was applied in this study to compare the stimulated side to the non-stimulated side. It is a less robust test, which may explain the significance presented in the study. For this meta-analysis, we considered the stimulated and non-stimulated sides as independent groups.34 Another possible limitation is that the study with the most weight in the meta-analysis was the study with intermediate methodological quality. Thereby, the results from the meta-analysis should be analyzed with caution. Unfortunately, only two studies were included in the meta-analysis and these studies measured the patients’ strength by a manual test which is considered reliable for ICU patients,35 however a scale with low precision as the results for a single joint assessment are limited to only 5 possible scores, as opposed to a dynamometer, for example, that presents continuous data.
Some previous literature reviews, with similar aims to the present have being recently published.36–38 The conclusions from these publications corroborate our findings that NMES is likely to be a useful tool for the maintenance of muscle strength and muscle mass in critically ill patients. However, some differences among these studies can be noted. The study by Williams and Flynn38 did not limit their analysis to best evidence as they included studies that were not randomized controlled trials and Maffiuletti et al.37 included studies that applied NMES out of ICU. These reviews also had different search strategies as the numbers of studies retrieved from the respective searchers are substantially different to the current study. Despite the limitations of the meta-analysis, the present systematic review brings objective information regarding the application of NMES in ICUs. The search in eight databases and the detailed analysis of the long list of titles confirm the data of the present review. Although most of the selected studies did not use adequate methodology, they showed that NMES can maintain or increase muscle strength, maintain muscle mass and volume, reduce time in MV and weaning time, and increase muscle degradation in critically ill patients in ICU. Therefore, studies with high methodological quality are still needed. Future studies should include patients with similar diseases. One possible design would be the investigation of the effects of NMES applied once a day for 30min (35Hz, pulse width 150μs, time on 5s and time off 5s) on the quadriceps femoris of ICU patients. Different to most studies seen here, allocation should be concealed, assessors should be blinded and intention-to-treat should be implemented in analysis.
ConclusionThe studies included in the present systematic review showed that NMES could have good results when used to maintain muscle mass and strength of critical patients in ICU and this was reinforced by the small meta-analysis presented. Future high-quality studies should establish a standard for the use of this therapy, with well-defined parameters and times of stimulation per day of treatment, so that if its efficacy confirmed, it can be incorporated into clinical practice.
Conflicts of interestThe authors declare that they have no conflicts of interest.
The authors wish to thank Pablo O. Rodriguez, Lefteris Karatzanos, and Serafim Nanas for providing access to the data from their studies, thus allowing us to perform the meta-analysis.