To audit the impact upon mortality of a massive bleeding management protocol (MBP) implemented in our center since 2007.
DesignA retrospective, single-center study was carried out. Patients transfused after MBP implementation (2007–2012, Group 2) were compared with a historical cohort (2005–2006, Group 1).
BackgroundMassive bleeding is associated to high mortality rates. Available MBPs are designed for trauma patients, whereas specific recommendations in the medical/surgical settings are scarce.
PatientsAfter excluding patients who died shortly (<6h) after MBP activation (n=20), a total of 304 were included in the data analysis (68% males, 87% surgical).
InterventionsOur MBP featured goal-directed transfusion with early use of adjuvant hemostatic medications.
Variables of interestPrimary endpoints were 24-h and 30-day mortality. Fresh frozen plasma-to-red blood cells (FFP:RBC) and platelet-to-RBC (PLT:RBC) transfusion ratios, time to first FFP unit and the proactive MBP triggering rate were secondary endpoints.
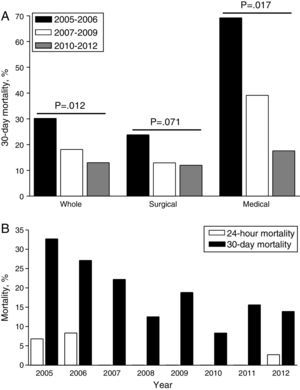
ResultsAfter MBP implementation (Group 2; n=222), RBC use remained stable, whereas FFP and hemostatic agents increased, when compared with Group 1 (n=82). Increased FFP:RBC ratio (p=0.053) and earlier administration of FFP (p=0.001) were also observed, especially with proactive MBP triggering. Group 2 patients presented lower rates of 24-h (0.5% vs. 7.3%; p=0.002) and 30-day mortality (15.9% vs. 30.2%; p=0.018) – the greatest reduction corresponding to non-surgical patients. Logistic regression showed an independent protective effect of MBP implementation upon 30-day mortality (OR=0.3; 95% CI 0.15–0.61).
ConclusionsThese data suggest that the implementation of a goal-directed MBP for prompt and aggressive management of non-trauma, massive bleeding patients is associated to reduced 24-h and 30-day mortality rates.
Auditar el impacto en la mortalidad de un protocolo de manejo de hemorragia masiva (PHM) en pacientes médico-quirúrgicos, implementado en nuestro centro desde el 2007.
DiseñoEstudio retrospectivo de cohortes. Grupo de intervención trasfundido con PHM (2007-2012, Grupo 2) comparado con cohorte histórica (2005-2006, Grupo 1).
ÁmbitoLos PHM existentes están diseñados para pacientes con politraumatismo, sin evidencia clara para pacientes médicos o quirúrgicos.
PacientesSe incluyeron 304 pacientes en el análisis (68% hombres, 87% quirúrgicos), tras la exclusión de aquellos con mortalidad inmediata (<6h) tras la activación del PHM (n=20).
IntervencionesEl PHM consta de transfusión dirigida por objetivos analíticos y uso precoz de adyuvantes hemostáticos.
Variables de interésMortalidad a las 24h y a los 30 días. Las tasas transfusionales «plasma fresco congelado:concentrado de hematíes (PFC:CH)» y «plaquetas:CH (PLT:CH)», el tiempo hasta la primera unidad de PFC y la tasa de alerta proactiva del PHM fueron objetivos secundarios.
ResultadosDespués de la implementación del PHM (Grupo 2; n=222), el uso de CH se mantuvo estable, mientras que el de PFC y el uso de hemostáticos aumentó comparativamente con respecto al Grupo 1 (n=82). La razón PFC:CH se incrementó (p=0,053) y la administración de PFC fue más precoz (p=0,001), especialmente en el grupo de alerta proactiva. El Grupo 2 mostró una menor mortalidad a las 24h (0,5 vs. 7,3%, p=0,002) y a 30 días (15,9 vs. 30,2%, p=0,018), con el mayor descenso para los pacientes no quirúrgicos. La regresión logística mostró un efecto protector independiente del PHM para mortalidad a 30 días (OR=0,3; IC 95% 0,15-0,61).
ConclusionesEstos datos evidencian que la implementación de un PHM con gestión rápida y activa de la hemorragia masiva en pacientes médico-quirúrgicos se asocia a una reducción de las tasas de mortalidad a 24h y 30 días.
Massive transfusion refers to the infusion of a large volume of blood products over a relatively short period of time, in response to a clinical situation of massive haemorrhage.1 Massive bleeding leads to high mortality rates, partly as a result of the “lethal triad” (hypothermia, acidosis and coagulopathy).2 In trauma patients, uncontrolled bleeding may account for 50% of deaths occurred within the first 24h.3,4
Several studies have shown that an early and aggressive transfusion policy has a direct impact on patient survival.5,6 In fact, European guidelines for management of traumatic bleeding and coagulopathy recommend the development and implementation of local massive bleeding protocols (MBP), which should be activated soon after the patient admittance, or the awareness of a massive bleeding situation (GRADE 1C recommendation). Such a protocol should include a damage control strategy, restrictive fluid resuscitation, frequent coagulation monitoring, rational but optimal blood product usage, and early use of adjunctive haemostatics agents.6–8 Local Spanish recommendations have also become available, addressing requirements for transfusion support in massive bleeding situations.9
Transfusion strategy within MBPs has been designed based on the military trauma experience, where whole blood is used for transfusion support.10 Since the use of whole blood is considered unsafe and impractical in the civilian scenario,11–13 guidelines recommend the combined use of blood products aiming at a specific fresh frozen plasma-to-red blood cells ratio (FFP:RBC), attempting to emulate whole blood. Although several randomized controlled trials and meta-analysis have been conducted on this matter, the optimal FFP:RBC ratio remains to be established.14–17
Complementing transfusion support, the adjunctive co-administration of fibrinogen concentrates and antifibrinolytics have been included in the protocols, as both have proven to reduce mortality in massive transfusion trials.18–21 Prothrombin complex concentrate (PCC) and recombinant activated factor VII (rFVII) may also be useful when FFP is insufficient to provide coagulation factors to revert coagulopathy.22–25
Specific studies or recommendations for management of massive hemorrhage in medical or surgical patients were scarce, until recently.14,26–28 We report the use of a non-trauma MBP protocol in a single tertiary center, which has significantly impacted blood product usage and patient outcome.
Material and methodsPatientsAll patients over 18 years of age for whom a MBP was activated in our center between 2007 and 2012 were considered for analysis (Group 2). A retrospective cohort of patients, who were transfused in 2005 and 2006 and fulfilled the first two criteria of MBP, served as control group (Group 1; see below). Patients were included regardless they were on antiplatelet or anticoagulant therapy. Exclusion criteria were pregnancy, congenital and acquired coagulation factor deficiencies (with the exception of cirrhotic patients), and congenital platelet aggregation disorders. To avoid the effect of survival bias, patients who died shortly after MBP activation (<6h) were excluded from the final analysis. To analyze the impact of a MBP update made on 2009, patients from the Group 2 were divided in sub-groups for data analysis (Group 2A, 2007–2009 and Group 2B, 2010–2012; see below).
Massive bleeding criteriaMassive bleeding was identified with one or more of the following criteria: (1) replacement of whole blood volume in a 24-h period (7% of ideal body weight in adults); (2) replacement of 50% of blood volume in a 3-h period; or (3) blood loss >1500mL in 10min.
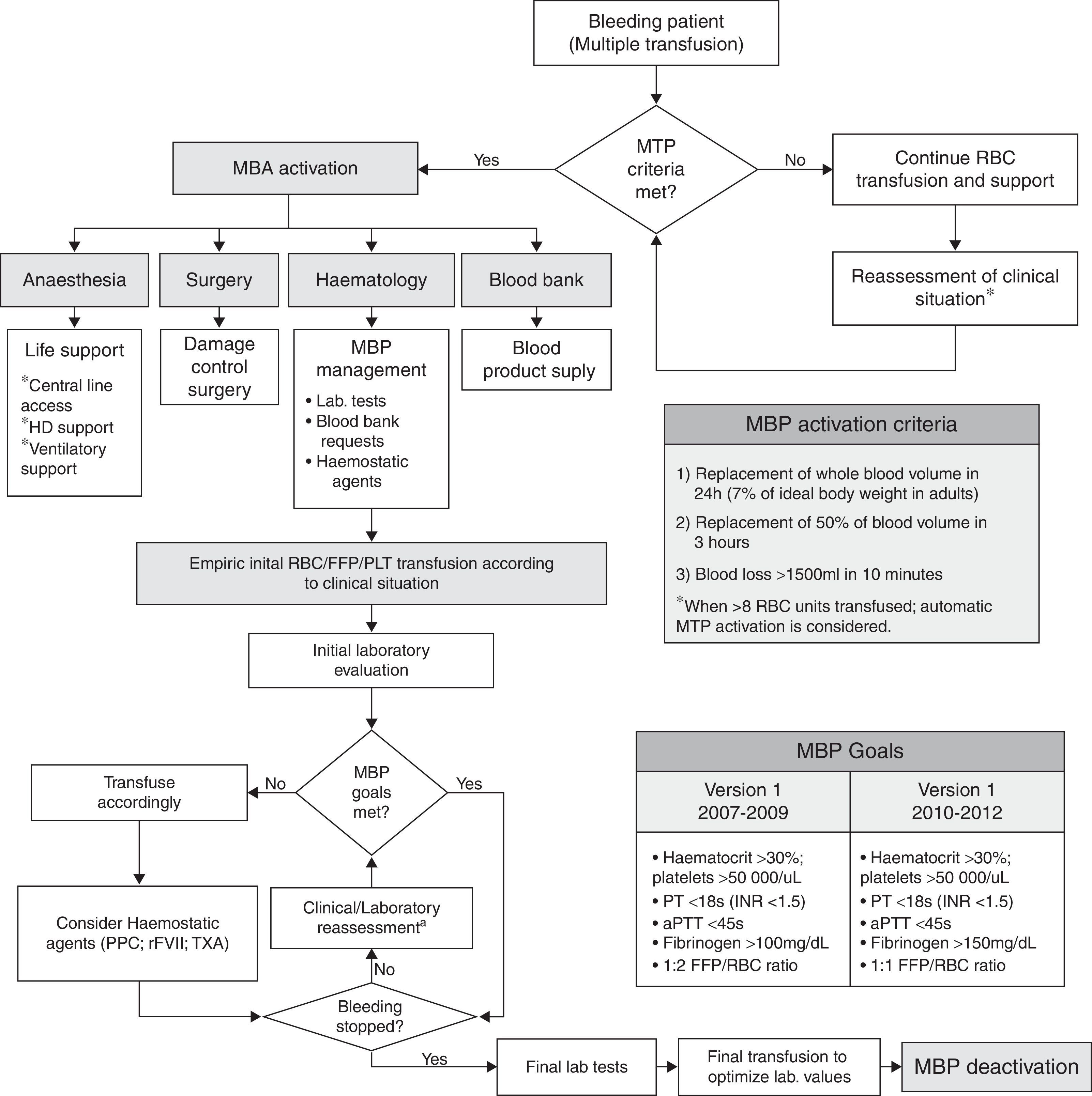
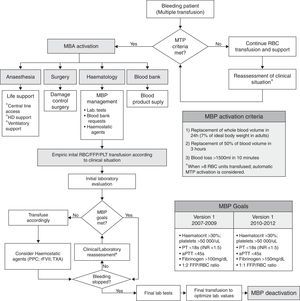
Massive bleeding management protocolThe MBP is summarized in Fig. 1. The 2009 update modified the targeted fibrinogen level (>1.5g/L), the targeted FFP:RBC transfusion ratio (1:1), and the use of PCC. For calculation of FFP:RBC ratio, 300mL of FFP was considered as 1 unit, whereas the platelet (PLT) to RBC transfusion ratio (PLT:RBC) was defined as the number of PLT pools per RBC unit (7 PLT units=1 PLT pool).
Massive transfusion protocol (MTP) algorithm. The boxes show the MTP activation criteria and MTP goals, used within the algorithm. RBC: red blood cells; FFP: fresh frozen plasma; PLT: platelet units; ICU: intensive-care unit; PT: prothrombin time; aPTT: activated partial thromboplastin time; PPC: prothrombin complex concentrate; rFVII: recombinant factor VII; TXA: tranexamic acid.
Globally, the goal of the MBP is the maintenance of an adequate blood volume with IV fluids and blood products, together with a timely control of bleeding source and an optimization of the coagulation status. Implementation of MBP required the cooperation between the on-call anaesthesiologists, hematologists and intensive care physicians, to provide hemodynamic, respiratory and blood product support, as soon as massive bleeding was detected.
Blood coagulation parameters were monitored using standard prothrombin time (PT), activated partial thromboplastin time (aPTT), and fibrinogen laboratory tests (Clauss method). At our institution, point-of-care coagulations tests (e.g. TEG/ROTEM) were not available for clinical decision-making within MBP.
The activation of MBP could be triggered by the bedside medical staff, when major bleeding criteria were fulfilled (proactive triggering), or by the blood bank personnel (remote or automatic triggering), after a pre-established threshold of requests was surpassed (>8 RBC). Activation of the MBP had to be confirmed by the on-call hematologist, who acted as team leader and also decided upon MBP deactivation once the bleeding was controlled. No changes in the activation criteria were introduced after the MBP protocol update. Additionally, a specific software environment was developed for the blood bank; it provided a computer-generated alert after the dispensation of the 8th consecutive RBC unit and allowed for a quick management of requests/dispensation of blood products.
Blood productsAll blood products were supplied by the regional blood bank of Navarra. RBC units were standard-leukocyte reduced. Cross-matching was done for RBC whenever possible, however, non-cross matched and/or ABO incompatible RBC units were allowed if needed, under mandatory hematologist authorization and supervision during transfusion.
Fresh frozen plasma was always from single-donor origin, inactivated with riboflavin–UVA and a typical volume of 300±20mL. ABO group compatibility was always maintained.
Platelets units were transfused according to availability; single-donor apheresis units were most frequently used in Groups 1 and 2A. From 2009, pooled platelet units became available and preferred for transfusion, therefore were more frequently used in the final period of the study (Group 2A). Platelets where leukocyte-plasma-free and suspended in saline solution, pooled platelet units contained an average of 7-units per pool. ABO/Rh compatibility was maintained if possible, however, incompatible ABO and Rh platelets were permitted. Anti-D gammaglobulin was administered according to local blood bank guidelines.
Haemostatic agentsThe use of haemostatic agents was considered when coagulopathy worsened despite FFP transfusion. Permitted haemostatic agents were recombinant activated factor VII (rFVIIa; Novoseven®, Novo Nordisk, Bagsværd, Denmark), 4-factor prothrombin complex concentrate (PCC; Octaplex® Octapharma, Ottawa, Canada), and fibrinogen (Haemocomplettan®, CSL Behring GmbH, Marburg, Germany). No cryoprecipitates were allowed. Doses were used according to physician criteria and prescribing information for each product. Repeated doses were allowed after a minimum of 3h if MBP was still active. PCC and rFVIIa were never used in combination, and typically rFVIIa administration was restricted to critical situations as a last resort for haemostatic control.
As for antifibrinolytics, ¿-aminocaproic acid and tranexamic acid were both allowed, in continuous or intermittent perfusion and without dosing limit. Antifibrinolytics were never used in combination with PPC or rFVII, their administration was maintained for a maximum of 48h, and was tapered down upon reversion of hyperfibrinolysis.
OutcomesThe primary outcome was the impact of the MBP implementation on 24-h and 30-day mortality. Secondary endpoints were frequency of proactive MBP triggering, transfusion ratios (FFP:RBC and PLT:RBC), time to first FFP unit administration, and the frequency of haemostatic agents use (Fibrinogen, rFVIIa, antifibrinolytics or PCC). Safety endpoint was the incidence of clinically evident thromboembolic complications 24h after MBP deactivation.
Statistical analysisKruskal–Wallis test was used for median comparison; Mann–Whitney U test was used for the remaining comparisons with Bonferroni adjustment for post hoc tests. McNemar test or Pearson's Chi square test was used for frequency analysis. Multinomial logistic regression analyses was performed using 30-day mortality as the dependent variable, PLT/RBC ratio, FFP/RBC ratio, time to first FFP unit (<2h), fibrinogen, antifibrinolytics, rFVIIa and PCC use, gender, age >65y, CV surgery, oncological surgery, non-surgical bleeding, and massive transfusion (>12 RBC units) were introduced as dichotomic co-variables, after excluding co-linearity.
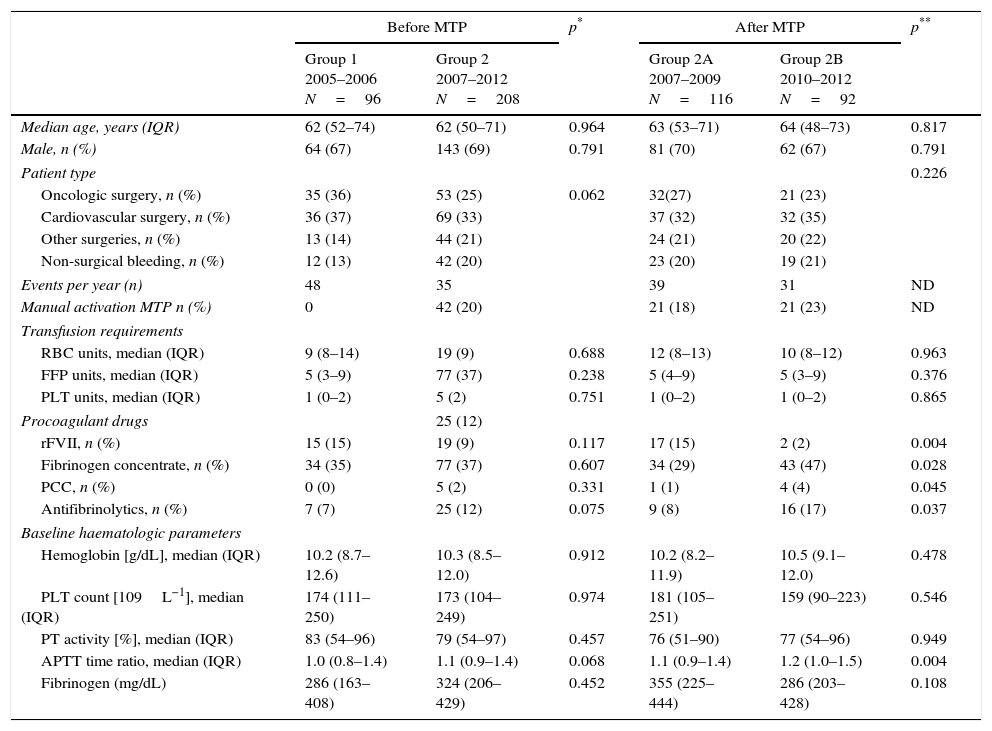
ResultsFrom a total of 324 patients with massive hemorrhage, 20 died shortly after MBP activation and were excluded from the study. For the remaining 304, there were virtually no differences in demographic and clinical data or baseline laboratory parameters between groups or sub-groups (Table 1).
Subgroup characteristics. Demographics, clinical information, transfusion requirements and procoagulant agents use.
| Before MTP | p* | After MTP | p** | |||
|---|---|---|---|---|---|---|
| Group 1 2005–2006 N=96 | Group 2 2007–2012 N=208 | Group 2A 2007–2009 N=116 | Group 2B 2010–2012 N=92 | |||
| Median age, years (IQR) | 62 (52–74) | 62 (50–71) | 0.964 | 63 (53–71) | 64 (48–73) | 0.817 |
| Male, n (%) | 64 (67) | 143 (69) | 0.791 | 81 (70) | 62 (67) | 0.791 |
| Patient type | 0.226 | |||||
| Oncologic surgery, n (%) | 35 (36) | 53 (25) | 0.062 | 32(27) | 21 (23) | |
| Cardiovascular surgery, n (%) | 36 (37) | 69 (33) | 37 (32) | 32 (35) | ||
| Other surgeries, n (%) | 13 (14) | 44 (21) | 24 (21) | 20 (22) | ||
| Non-surgical bleeding, n (%) | 12 (13) | 42 (20) | 23 (20) | 19 (21) | ||
| Events per year (n) | 48 | 35 | 39 | 31 | ND | |
| Manual activation MTP n (%) | 0 | 42 (20) | 21 (18) | 21 (23) | ND | |
| Transfusion requirements | ||||||
| RBC units, median (IQR) | 9 (8–14) | 19 (9) | 0.688 | 12 (8–13) | 10 (8–12) | 0.963 |
| FFP units, median (IQR) | 5 (3–9) | 77 (37) | 0.238 | 5 (4–9) | 5 (3–9) | 0.376 |
| PLT units, median (IQR) | 1 (0–2) | 5 (2) | 0.751 | 1 (0–2) | 1 (0–2) | 0.865 |
| Procoagulant drugs | 25 (12) | |||||
| rFVII, n (%) | 15 (15) | 19 (9) | 0.117 | 17 (15) | 2 (2) | 0.004 |
| Fibrinogen concentrate, n (%) | 34 (35) | 77 (37) | 0.607 | 34 (29) | 43 (47) | 0.028 |
| PCC, n (%) | 0 (0) | 5 (2) | 0.331 | 1 (1) | 4 (4) | 0.045 |
| Antifibrinolytics, n (%) | 7 (7) | 25 (12) | 0.075 | 9 (8) | 16 (17) | 0.037 |
| Baseline haematologic parameters | ||||||
| Hemoglobin [g/dL], median (IQR) | 10.2 (8.7–12.6) | 10.3 (8.5–12.0) | 0.912 | 10.2 (8.2–11.9) | 10.5 (9.1–12.0) | 0.478 |
| PLT count [109L−1], median (IQR) | 174 (111–250) | 173 (104–249) | 0.974 | 181 (105–251) | 159 (90–223) | 0.546 |
| PT activity [%], median (IQR) | 83 (54–96) | 79 (54–97) | 0.457 | 76 (51–90) | 77 (54–96) | 0.949 |
| APTT time ratio, median (IQR) | 1.0 (0.8–1.4) | 1.1 (0.9–1.4) | 0.068 | 1.1 (0.9–1.4) | 1.2 (1.0–1.5) | 0.004 |
| Fibrinogen (mg/dL) | 286 (163–408) | 324 (206–429) | 0.452 | 355 (225–444) | 286 (203–428) | 0.108 |
MTP: massive bleeding protocol; IQR: interquartile range; RBC: red-blood cells; FFP: fresh frozen plasma; PLT: platelets; rFVII: recombinant factor VII; PCC: prothrombin complex concentrate; PT: prothrombin time; APTT: activated partial thromboplastin time.
A summary of the characteristics and the quantity of blood products used are summarized in supplementary Table 1. From the safety point of view, only 2 cases of transient pulmonary edema were reported during the study period, no transfusion related-lung injury (TRALI) was demonstrated based on anti-HLA antibody testing. Non-haemolytic transfusion-related reactions were seen in approximately 0.2% of patients, mostly hyperthermia and urticarial reactions that resolved quickly with symptomatic treatment (supplementary Table 1).
Globally, 82% of massive bleeding events occurred in surgical patients. The remaining 18% of cases were non-surgical, mostly gastro-intestinal bleeding in cirrhotic patients. Among patients from Group 2, MBP was proactively triggered in 42 out 22 (20%), without differences between sub-groups (Table 1).
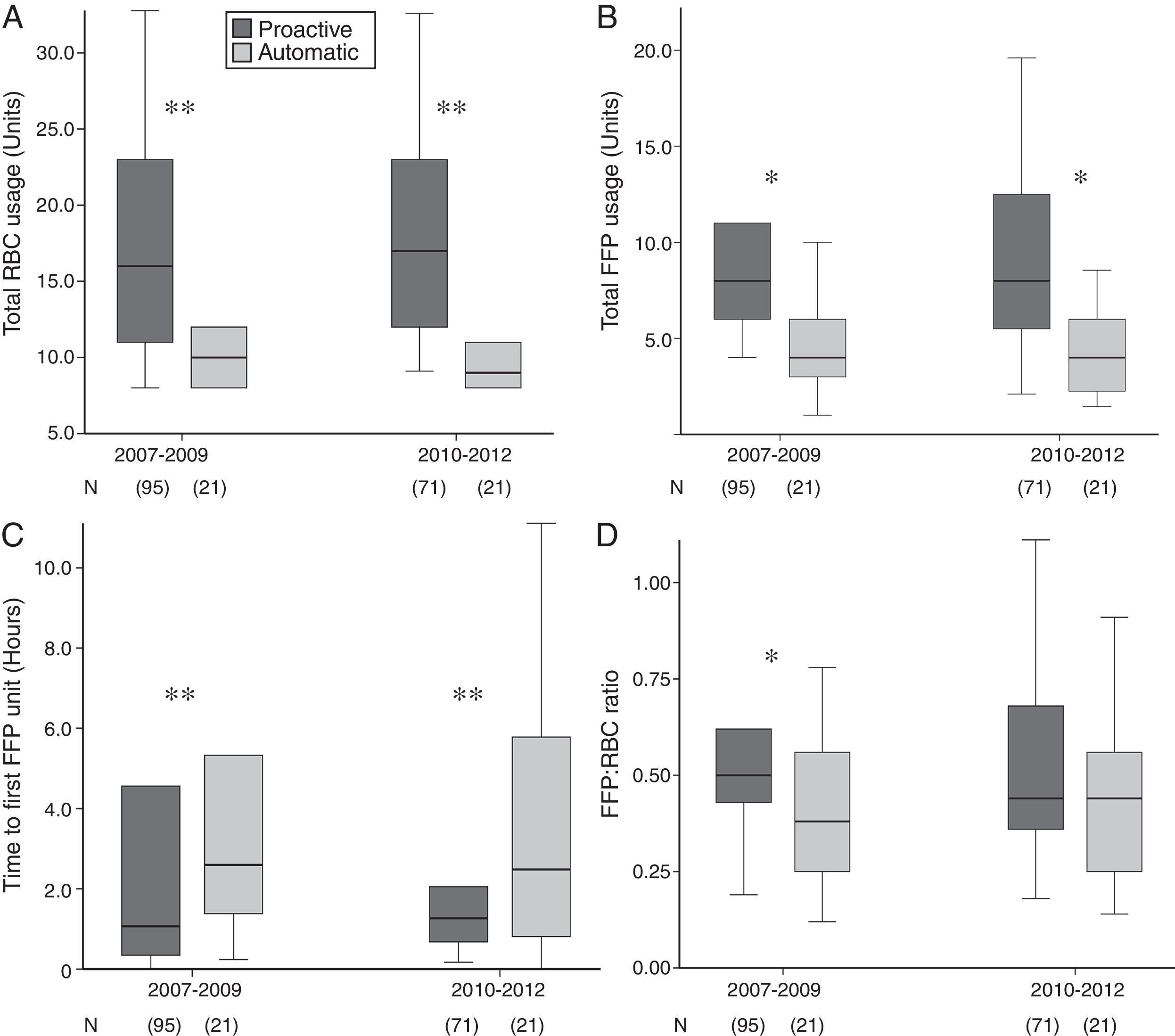
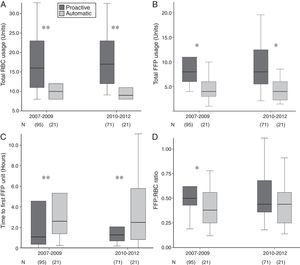
For the whole series, the absolute frequency of haemostatic agents used was high (60%), without group differences in median doses used. A significant time-dependent increase in fibrinogen, antifibrinolytics and PCC usage, together with a reduction in rFVIIa usage, was observed since MBP implementation (Table 1). As shown in Table 2, blood component requirements were comparable among groups and subgroups. Overall, an increase in FFP:RBC ratio (p=0.053) and a significant decrease in time to transfusion of the first FFP unit (p=0.001) were found after MBP implementation, without differences in PLT:RBC ratio.
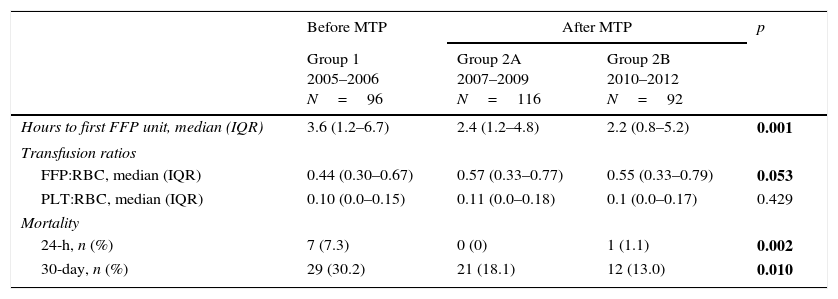
MTP effect on transfusion requirements and mortality.
| Before MTP | After MTP | p | ||
|---|---|---|---|---|
| Group 1 2005–2006 N=96 | Group 2A 2007–2009 N=116 | Group 2B 2010–2012 N=92 | ||
| Hours to first FFP unit, median (IQR) | 3.6 (1.2–6.7) | 2.4 (1.2–4.8) | 2.2 (0.8–5.2) | 0.001 |
| Transfusion ratios | ||||
| FFP:RBC, median (IQR) | 0.44 (0.30–0.67) | 0.57 (0.33–0.77) | 0.55 (0.33–0.79) | 0.053 |
| PLT:RBC, median (IQR) | 0.10 (0.0–0.15) | 0.11 (0.0–0.18) | 0.1 (0.0–0.17) | 0.429 |
| Mortality | ||||
| 24-h, n (%) | 7 (7.3) | 0 (0) | 1 (1.1) | 0.002 |
| 30-day, n (%) | 29 (30.2) | 21 (18.1) | 12 (13.0) | 0.010 |
MTP: massive transfusion protocol; IQR: interquartile range; RBC: red-blood cells; FFP: fresh frozen plasma; PLT: platelets.
Compared to automatic activation, proactive MBP triggering was consistently associated with increased use of RBC, FFP, higher FFP:RBC transfusion ratio and shorter to first FFP unit transfusion (Fig. 2).
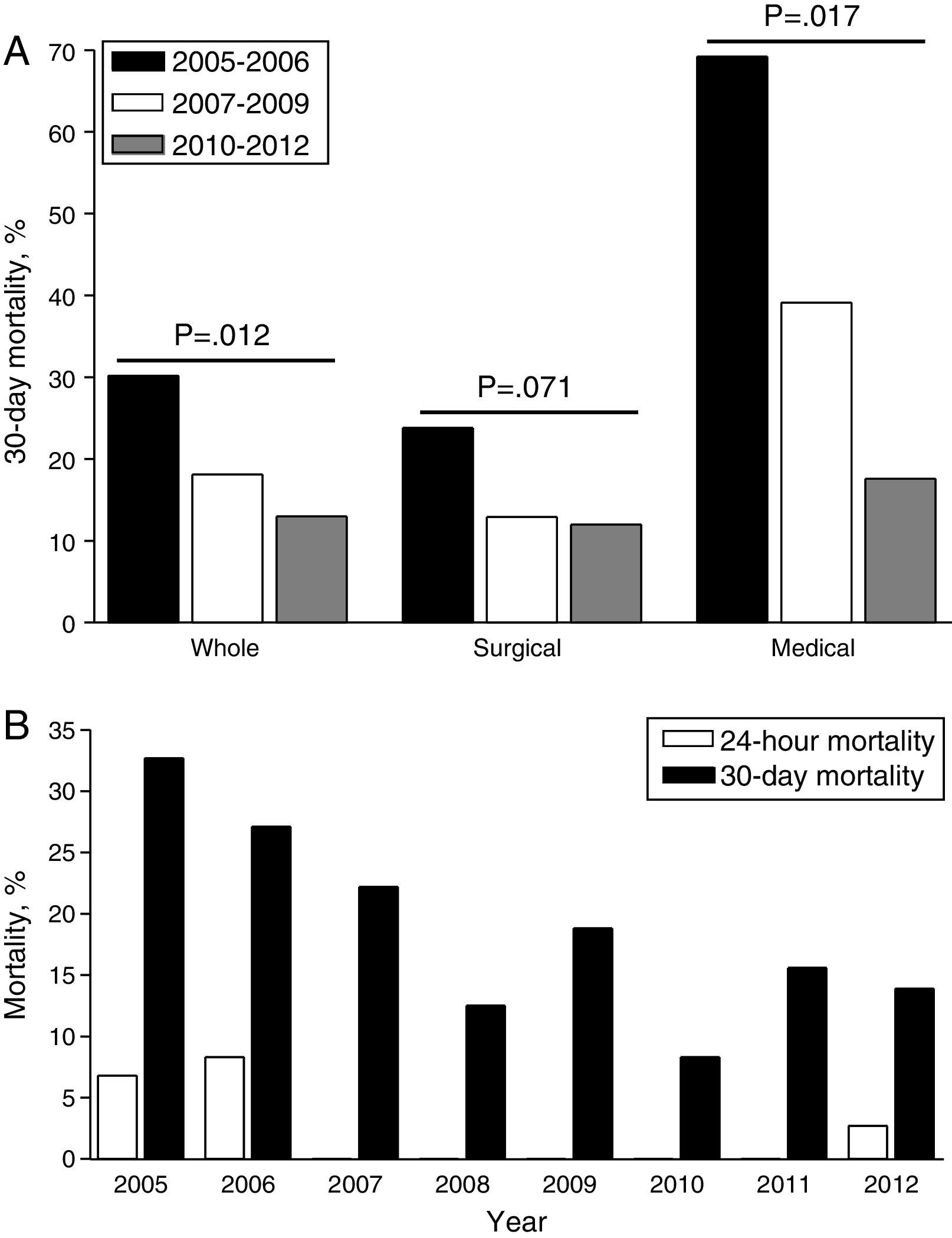
Overall, 24-h mortality was 2.6% and 30-day mortality 20.4%. The greatest reduction of 30-day mortality was observed at the end of the observation period (Group 2B), particularly for non-surgical patients (Fig. 3A); yearly evolution of 24-h and 30-day mortality is also depicted in Fig. 3B. After MBP implementation, a significant time-dependent decrease in 24-h mortality rate (7.3–1.1%) and 30-day mortality rate (30.2–13.0%, p=0.02) were observed (Table 2).
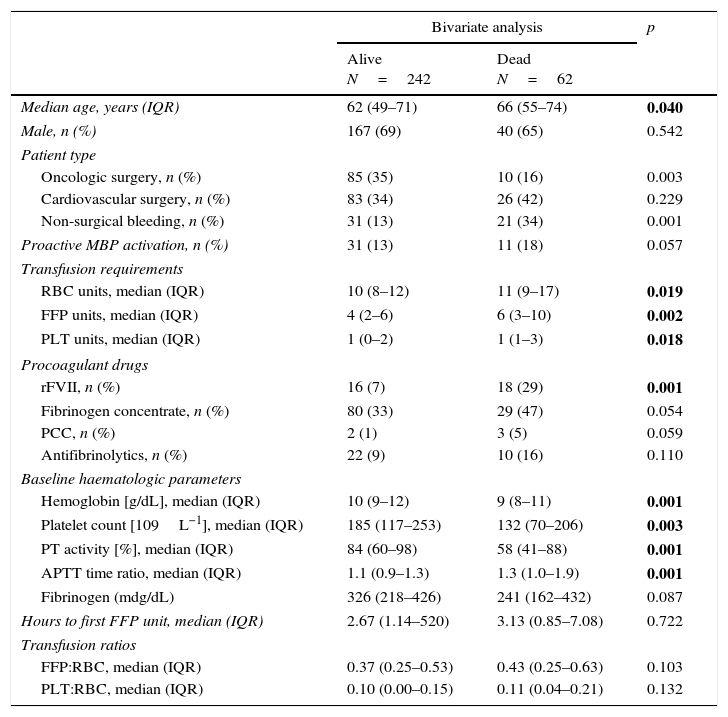
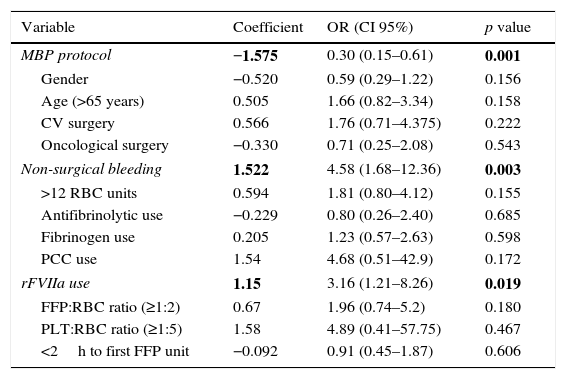
In the bivariate analysis, non-surgical bleeding, oncological surgery, lower hematimetric values, prolonged coagulation times, total blood products, and rFVIIa usage were associated with 30-day mortality, whereas MBP use appeared as protective factor (Table 3). Noteworthy, FFP:RBC ratio (≥1:2) and PLT:RBC ratio (≥1:5) were not associated with 30-day mortality in the bivariate analysis (Table 3). In the multinomial logistic regression analysis only non-surgical bleeding and rFVII use were independently associated with an increased OR for 30-day mortality, whereas MBP implementation was associated with a reduction in OR for 30-day mortality (Table 4).
Summary of bivariate analysis of 30-day mortality.
| Bivariate analysis | p | ||
|---|---|---|---|
| Alive N=242 | Dead N=62 | ||
| Median age, years (IQR) | 62 (49–71) | 66 (55–74) | 0.040 |
| Male, n (%) | 167 (69) | 40 (65) | 0.542 |
| Patient type | |||
| Oncologic surgery, n (%) | 85 (35) | 10 (16) | 0.003 |
| Cardiovascular surgery, n (%) | 83 (34) | 26 (42) | 0.229 |
| Non-surgical bleeding, n (%) | 31 (13) | 21 (34) | 0.001 |
| Proactive MBP activation, n (%) | 31 (13) | 11 (18) | 0.057 |
| Transfusion requirements | |||
| RBC units, median (IQR) | 10 (8–12) | 11 (9–17) | 0.019 |
| FFP units, median (IQR) | 4 (2–6) | 6 (3–10) | 0.002 |
| PLT units, median (IQR) | 1 (0–2) | 1 (1–3) | 0.018 |
| Procoagulant drugs | |||
| rFVII, n (%) | 16 (7) | 18 (29) | 0.001 |
| Fibrinogen concentrate, n (%) | 80 (33) | 29 (47) | 0.054 |
| PCC, n (%) | 2 (1) | 3 (5) | 0.059 |
| Antifibrinolytics, n (%) | 22 (9) | 10 (16) | 0.110 |
| Baseline haematologic parameters | |||
| Hemoglobin [g/dL], median (IQR) | 10 (9–12) | 9 (8–11) | 0.001 |
| Platelet count [109L−1], median (IQR) | 185 (117–253) | 132 (70–206) | 0.003 |
| PT activity [%], median (IQR) | 84 (60–98) | 58 (41–88) | 0.001 |
| APTT time ratio, median (IQR) | 1.1 (0.9–1.3) | 1.3 (1.0–1.9) | 0.001 |
| Fibrinogen (mdg/dL) | 326 (218–426) | 241 (162–432) | 0.087 |
| Hours to first FFP unit, median (IQR) | 2.67 (1.14–520) | 3.13 (0.85–7.08) | 0.722 |
| Transfusion ratios | |||
| FFP:RBC, median (IQR) | 0.37 (0.25–0.53) | 0.43 (0.25–0.63) | 0.103 |
| PLT:RBC, median (IQR) | 0.10 (0.00–0.15) | 0.11 (0.04–0.21) | 0.132 |
MTP: massive bleeding protocol; IQR: interquartile range; RBC: red-blood cells; FFP: fresh frozen plasma; PLT: platelets; rFVII: recombinant factor VII; PCC: prothrombin complex concentrate; PT: prothrombin time; APTT: activated partial thromboplastin time.
Multinomial logistic regression analysis of 30-day mortality.
| Variable | Coefficient | OR (CI 95%) | p value |
|---|---|---|---|
| MBP protocol | −1.575 | 0.30 (0.15–0.61) | 0.001 |
| Gender | −0.520 | 0.59 (0.29–1.22) | 0.156 |
| Age (>65 years) | 0.505 | 1.66 (0.82–3.34) | 0.158 |
| CV surgery | 0.566 | 1.76 (0.71–4.375) | 0.222 |
| Oncological surgery | −0.330 | 0.71 (0.25–2.08) | 0.543 |
| Non-surgical bleeding | 1.522 | 4.58 (1.68–12.36) | 0.003 |
| >12 RBC units | 0.594 | 1.81 (0.80–4.12) | 0.155 |
| Antifibrinolytic use | −0.229 | 0.80 (0.26–2.40) | 0.685 |
| Fibrinogen use | 0.205 | 1.23 (0.57–2.63) | 0.598 |
| PCC use | 1.54 | 4.68 (0.51–42.9) | 0.172 |
| rFVIIa use | 1.15 | 3.16 (1.21–8.26) | 0.019 |
| FFP:RBC ratio (≥1:2) | 0.67 | 1.96 (0.74–5.2) | 0.180 |
| PLT:RBC ratio (≥1:5) | 1.58 | 4.89 (0.41–57.75) | 0.467 |
| <2h to first FFP unit | −0.092 | 0.91 (0.45–1.87) | 0.606 |
MTP: massive transfusion protocol; PCC: prothrombin complex concentrate; rFVII: recombinant factor VII; RBC: red blood cells; FFP: fresh frozen plasma; PLT: platelets; CV: cardiovascular; OR: odds-ratio.
No clinically evident thromboembolic complications were seen 24h after MTP deactivation.
DiscussionWe describe the experience of a single-center where the implementation of a MBP in non-trauma patients was associated with a significant reduction of 30-day mortality (an absolute 14% reduction compared to historical controls) together with improved transfusion dynamics and better transfusion ratios.
The frequency of massive hemorrhage events was 40.5 cases per year (0.75 cases per week), similar to previous reports with comparable patient characteristics.29 Overall, our results show lower 24-h (2.6%) and 30-day mortality rates (20.4%) compared to similar series.30,31 This is likely to depend on differences in MBP activation criteria (e.g., remote activation after transfusion of 8 RBC units at our center) and patient characteristics (mostly oncological and cardiovascular surgery in our series). A previous study compared the use of an MBP between trauma and non-trauma bleeding events, reporting higher 24-h mortality in non-trauma patients (59% vs. 35%), without differences in 30-day mortality (59% vs. 51%).31 Another retrospective study compared the results of MBP activation in trauma vs. non-trauma patients, and found no differences in blood product usage or in-hospital mortality between groups (33.9% vs. 36.5%). However, trauma patients were more likely to die in the first 24h than non-trauma patients (27.4% vs. 11.1%).30 Although no definitive conclusion can be made regarding the utility of MBP in non-trauma patients, the use of a non-MBP cohort in our study strengthen the results obtained regarding the potential role of a MBP in non-surgical bleeding scenarios.
It has been proposed that beneficial effects of any MBP are mainly derived from the improvement in transfusion ratios and the early administration of FFP.11,12,16,17,32,33 In our series, there was a noticeable effect of MBP implementation on transfusion practices (FFP:RBC ratio and time to first FFP unit), but multivariate regression analysis showed that 30-day mortality reduction (survival effect) was only independently associated with MBP implementation. Although improvements in transfusion support might have contributed to mortality reduction, many other unmeasured variables associated with MBP may have contributed equally to the observed results (e.g., haemostatic agent administration, medical support and increased MBP awareness).18,34,35
The FFP:RBC 1:1 ratio target in our MBP was based on several reports suggesting early used of balanced transfusions as a central aspect of haemostatic resuscitation.8,14,34,35 Despite the 1:1 target, our best performance was only 1:1.5, with the majority of patients between 1:1.5 and 1:3 ratio (a FFP:RBC ratio ≥1:2 was attained in 40% of patients after MBP implementation). This observation likely reflects late activation of the MBP, with unbalanced RBC transfusion starting with moderate but continuous bleeding until the MBP alarm is triggered (typically after 8 RBC units). Several authors have reported on MBP use in trauma and non-trauma cohorts where only 50–60% of patients effectively received a FFP:RBC ratio of 1:2 and only 8–12% of patients achieved a 1:1 ratio.27,30,36 Additionally, a large randomized trial has recently showed no benefit of 1:1:1 RBC:FFP:PLT ratio over 2:1:1 ratio in terms of mortality.36 Based on the available evidence, many centers have modified local protocols accordingly.27,34,36–39
From the implementation perspective, the low percentage of proactive MBP triggering is probably a consequence of a slow gain of compliance with the protocol, which has been considered sub-optimal after 6 years of clinical use. This finding may also help to explain the lack of association between the improvement in transfusion support and mortality. We believe that appropriate training, education and awareness among the medical staff and nurses may be useful for improving compliance with the MBP and patient outcome.
The application of a computer-generated alarm system for the triggering of the MBP and the leading role of the hematologist on the clinical decision making during the transfusion act should be emphasized as part of a successful MBP implementation. Although these items may represent an unmeasured variable that may have contributed to the clinical benefit observed, we believe that it is an uttermost important part of the care of patients with massive bleeding.
In our series, global use of RBC units was lower than that typical reported for MBP use in trauma, thus underlining the differences in the characteristics of massive bleeding between trauma and medical populations.30,40 Additionally, the numbers of transfused blood components remained stable after MBP implementation (Table 1); this was an unexpected finding, as the MBP implementation was expected to increase overall blood product usage as the transfusion ratios improve.36
As for recombinant factor VII (rFVII), it was typically used as a last resort haemostatic agent and, therefore, the higher mortality rate observed among patients receiving rFVII was not surprising. However, the evidence for the use of rFVII is scarce and, in our center, it has become anecdotal since 2010.
The use of viscoelastic tests (e.g., TEG®/ROTEM®) to guide transfusion practice has been preconized in recent studies.41,42 However, a Cochrane systematic review found that while transfusion guided by TEG or ROTEM may reduce bleeding, it does not improve morbidity or mortality.43 In this regard, it is worth emphasizing that only standard coagulation tests were used for decision-making within our MBP (APTT, PT and fibrinogen). The results seem to support the use of laboratory coagulation parameters within a goal-directed transfusion strategy, as recommended by recent UK guidelines,26 particularly in centers where there is rapid turnaround time for laboratory tests.
Our results have the limitations proper of a single-center retrospective analysis, such as the possibility of unmeasured variables, selection bias and the lack of statistical power to draw definitive conclusions about mortality. Although an independent association has been found between MBP implementation and mortality, the study design provide results of associative nature and therefore no cause-effect relationship can be firmly established.
In conclusion, our results show that implementation of an institutional MBP for prompt and aggressive management of non-trauma, massive bleeding patients was associated with a lower rate of mid-term mortality. This change in mortality rates could be driven by the improvement in patient support together with improved FFP availability, better FFP:RBC transfusion ratios and increased use of pro-haemostatic agents. Further studies are required to prospectively confirm the effect of MBP in non-trauma patients.
FundingNo funding was required for the development of this work.
Authors’ contributionsNM performed data review and wrote the manuscript, FH designed the MTP and wrote the manuscript, AA performed data review and reviewed the manuscript, MH designed MTP and managed blood bank requests, MM helped with statistical analysis and reviewed the manuscript, JAP and RL designed the MTP and reviewed the manuscript.
Conflicts of interestThe authors declare no conflicts of interests.
The authors gratefully acknowledge the support from laboratory technicians and nurses from the Haematology Department and Blood Bank in implementing MBP. We would also like to thank Ana Ezponda and Maria Marcos for their valuable help in data entry.