Up to 60% of all children who receive bone marrow transplantation (BMT) develop respiratory distress. It constitutes a common complication in this kind of patients, due to the increasing number of therapeutic applications of BMT and to improvement in the therapeutic approach to the problems associated with this procedure. We describe the different causes of respiratory distress after BMT in relation to its initiation or the presence of infection in its origin. The diagnosis and treatment are also reviewed.
Hasta el 60% de los niños que reciben trasplante de médula ósea (TMO) presentan dificultad respiratoria. Supone una complicación cada vez más frecuente debido al aumento en el número de aplicaciones terapéuticas del TMO y al mejor abordaje terapéutico de los problemas asociados al mismo. Se describen las diferentes causas de dificultad respiratoria tras TMO en función de su instauración (precoz o tardía) o la presencia de infección en el origen de la misma. Se revisa a su vez el diagnóstico y tratamiento de cada una de ellas.
Up to 60% of all children subjected to bone marrow transplantation (BMT) suffer breathing difficulties.1–3 This is an increasingly common complication, due to the growing number of therapeutic applications of BMT and to improved management of the problems associated to the latter.4,5
In general, breathing difficulties following BMT are related to restrictive changes caused by lung fibrosis, alterations in alveolar-capillary membrane diffusion, and infections produced by pathogens which in the context of immune suppression can cause locoregional damage.2,3,6–11 The associated clinical manifestations can range from inexistent or larvated symptoms to catastrophic and rapidly evolving conditions.11
Although infections remain the most frequent cause of breathing difficulties in patients of this kind, the number of cases of dyspnea of non-infectious origin has increased. In this context, diseases such as bronchiolitis obliterans or idiopathic pneumonia are increasing in both incidence and importance.2,9,10,12–14
The mortality rate has decreased among patients subjected to BMT due to a number of reasons: improved donor screening (greater histocompatibility), fewer relapses before BMT (patients with better baseline conditions), and optimization of respiratory care in the Intensive Care Unit (ICU).5,15–18 In turn, improved understanding of the risk factors for developing lung damage in the context of BMT (Table 1) has made it possible to both adopt preventive strategies and to establish an early diagnosis.5,13,15,17,19,20
Considering the immune suppression found in patients subjected to BMT, three phases with their corresponding typical complications can be distinguished2,7,9,13,14:
- •
Neutropenic phase (one week before transplantation, and persisting until approximately 3 weeks after transplantation): increased frequency of bacterial and fungal infections (risk of Aspergillus infection in previously colonized patients). Consideration is required of the possibility of diffuse alveolar hemorrhage, non-cardiogenic acute lung edema and chemotherapy-related toxicity as frequent non-infectious conditions with high morbidity-mortality.7,13,21,22
- •
Neutropenia recovery phase (week 3–10): viral infections secondary to altered cellular immunity add to the risk of bacterial and fungal infections. The risk of cytomegalovirus (CMV) disease in carriers prior to BMT is particularly important.13,22–26
- •
Immune reconstitution and bone marrow graft phase (beyond day 100 after BMT): related to inflammatory phenomena caused by graft versus host disease (GVHD). Special mention should be made of bronchiolitis obliterans (rarely caused by viruses in the previous phases) or chronic GVHD itself.13,22,26
A description is provided below of the different presentations of breathing difficulty in patients with BMT, in relation to their timing with respect to the moment of transplantation (early14 or late; summary of non-infectious causes in Table 2). A summary is also provided of the causes of dyspnea of infectious origin in these patients.
Principal characteristics of the most prevalent conditions observed in children with dyspnea and bone marrow transplantation.
| Early non-infectious disorders | |
| Idiopathic pneumonia | Diffuse pulmonary infiltrates and exclusion of infectious causes |
| Diffuse alveolar hemorrhage | Diffuse pulmonary infiltrates and bronchoalveolar lavage with progressive bleeding and ≥20% hemosiderin-loaded macrophages |
| Graft syndrome | Diffuse pulmonary infiltrates appearing 5 days after the rise in neutrophil counts. Cardiac or infectious disease must be discarded |
| Late non-infectious disorders | |
| Bronchiolitis obliterans | Fever, patchy consolidations in lungs, and typical histology |
| Delayed pulmonary toxicity syndrome | Ground glass opacities in autologous BMT or after high-dose conditioning chemotherapy; good response to treatment |
| Pulmonary cytolytic thrombosis | Fever and lung nodules in children with GVHD, and typical histology if biopsy is performed |
Idiopathic pneumonia is an important cause of mortality after successful bone marrow grafting, with rates as high as 74% in some series.2,10,13,14,23 The incidence is approximately 10%. Pneumonia of this kind has been related to bacterial or fungal infections that have not been diagnosed despite the conduction of complementary tests. At present, viruses are considered to be the likely explanation for such situations.2,10,13,14
Known risk factors are the use of methotrexate27 as chemotherapeutic drug, BMT performed in children beyond nursery school age, a prolonged interval between diagnosis of the background disease and transplantation, a poor clinical condition prior to BMT, and the administration of high irradiation doses (summative effect with methotrexate and prior cytomegalovirus seropositivity).2,13,28
Dyspnea, fever and hypoxemia are the three signs associated to this disorder, which is most often observed between days 40 and 75 after bone marrow grafting.2,13,29
An exclusion diagnosis should be established in all cases (Table 3). It is essential to discard any possible infectious origin of dyspnea and lung infiltrates in patients subjected to BMT.29,30 The chest X-rays often show diffuse interstitial infiltrates (in 90% of the cases)—thoracic computed tomography (CT) being a useful tool for establishing the differential diagnosis.7,11,13 Bronchoalveolar lavage (BAL) may prove necessary (revealing typical increments in tumor necrosis factor-alpha, TNF-alpha), together with transbronchial biopsy, with the purpose of completing the diagnostic workup.7,11,13,30
Diagnostic criteria of idiopathic pneumonia syndrome.
| Evidence of diffuse alveolar damage |
| Multilobular infiltrates |
| Increased alveolar-arterial oxygen gradient and pulmonary restriction |
| Absence of infectious origin of lung damage after extensive and adequate study repeated on at least two occasions |
| Bronchoalveolar lavage negative for pathogens (bacterial or otherwise) |
| No improvement with very broad spectrum antimicrobial treatment |
| Transbronchial biopsy, if feasible, showing inflammatory histology without signs of infection |
Ventilatory support is the basis of management (invasive mechanical ventilation may prove necessary).22,31 Medical treatment in turn is based on the administration of corticosteroids (methylprednisolone 2–5mg/kg/day, with dose escalation in the case of an unsatisfactory response) and etanercept (to be introduced early, administering 2 weekly doses, and generally observing a response after the second administration when such treatment proves effective). Broad spectrum antibiotic treatment should be associated in all cases. Even when the pneumonitis is resolved and radiological improvement is confirmed, the mortality rate is high (31%).32–34 Indicators of a poor prognosis are the absence of treatment response and the need for invasive mechanical ventilation for more than 7 days.35
Diffuse alveolar hemorrhageThe incidence of diffuse alveolar hemorrhage varies between 5 and 20% independently of the type of BMT. It tends to develop from 30 days after grafting, and the associated mortality is high in the absence of treatment (up to 90%). The mortality rate in turn decreases to 10–30% in the case of adequate treatment response. Diffuse alveolar hemorrhage is an unpredictable condition that has no defined risk factors other than the patient clinical condition before transplantation.25,30,36
The typical clinical manifestations comprise dyspnea, cough and progressive breathing difficulties. Hemoptysis is observed in 20% of the cases, and is not essential for establishing the diagnosis.30 Hypoxemia is very typical and constitutes a key clue for suspecting the condition.13,14
The chest X-ray findings tend to be suggestive of acute lung edema with normal cardiac function as evidenced by echocardiography.2–4,8 Thoracic CT is useful for evaluating the degree of involvement. Bronchoalveolar lavage in turn confirms the diagnosis, yielding hematic fluid with abundant neutrophils and hemosiderin.30,37,38
Supportive treatment is required, correcting the thrombopenia and consumption coagulopathy usually seen in relation to the bleeding. A restrictive water balance is indicated, with ventilation support according to the needs of the patient.22,31,39 In the event mechanical ventilation is needed, difficulties may be found because of the restriction and alteration of the ventilation–perfusion ratio caused by the bleeding. We should administer high-dose methylprednisolone (1g/m2/day in 3 doses) during 5 days, followed by dose reduction if an adequate response is observed. Activated Factor VII is to be administered (90μg/kg in an initial attack dose, and then every 6hours, until completing 7 doses).35 In the event of a poor course, biological treatment can be started with etanercept, with the purpose of reducing the inflammatory component that may be present in the context of the bleeding.33–35
Graft syndromeThe incidence of graft syndrome is 10% in the context of autologous transplantation and 30% in allogenic grafts (typical in patients with hyperacute GVHD). The associated mortality rate is about 26%, and the syndrome generally develops three days after leukocyte elevation identified from the blood count.24,40,41 Graft syndrome is associated to a risk of diffuse alveolar hemorrhage that reaches 33% in some series.30
Known risk factors of graft syndrome are the infusion of CD34-positive cells in BMT, patients with BMT in an early stage of the disease, and the utilization of BMT in solid tumors with a poor response to treatment.2,13,14,30,35
The affected patients present fever, generalized skin rash and breathing difficulties. There also may be damage to other organs: liver alterations, renal failure, edemas with weight gain, and encephalopathy.2,13,14
Establishing the diagnosis is facilitated by the presence of a series of clinical signs and laboratory test findings such as fever (seen in 63% of the cases), hypoxia (in approximately 90% of the cases), and 5 consecutive days of neutrophil count elevation associated or not associated to acute lung edema as evidenced from the chest X-rays, without demonstrated alterations in heart function.2,30
Ventilatory support with a strict water balance is indicated. We should administer corticosteroids (methylprednisolone 500mg/m2/day in 2 doses during 2–3 days), antihistamines, antithermal agents and topical treatment for the skin lesions (tacrolimus or corticosteroids).30,34,35 A favorable treatment response is usually observed.
Prophylactic treatment is indicated, administering low-dose methylprednisolone (0.5–1mg/kg/day) during the first 5 days of leukocyte count elevation.30,35
Capillary leakage syndromeThe incidence of capillary leakage syndrome is approximately 20%, regardless of the type of transplant involved.2,9,11,14 The associated mortality rate is 90% in the absence of treatment, and the syndrome develops in the first 15 days after grafting, approximately.30,35
Generalized edema with the presence of pleural, pericardial and peritoneal fluid are the typical signs of this syndrome, and are crucial for establishing the diagnosis.2,9,11,14,30 The chest X-rays typically reflect acute lung edema without heart failure.7
High-dose corticosteroids are the basis of management. Such treatment can be associated to C1 inhibitor concentrate in the case of poor treatment response or serious initial clinical patient conditions (attack dose 60units/kg, followed by 2 doses of 30units/kg and 4 doses of 15units/kg every 12h).30,35
Late dyspnea of non-infectious origin in bone marrow transplantationBronchiolitis obliterans (BO)The incidence of bronchiolitis obliterans (BO) in autologous BMT is low, and reaches 2–20% in allogenic BMT. It typically manifests 6–12 months after cell infusion, though in some cases can develop early after only three months. Prevention through T lymphocyte depletion is useful.6,26,40,42 The long-term mortality rate is 20%, and the underlying risk factors are GVHD and hypogammaglobulinemia.5,17,20
BO is characterized by larvated clinical manifestations in the form of cough, wheezing and progressive dyspnea. The chest X-ray findings are usually normal or show hyperinsufflation. Thoracic CT is useful, since it is able to evidence both air trapping and—in some cases—bronchiectasis. Where possible, lung function tests should be performed, which will reveal an obstructive pattern (diminished FEV1) with a reduction in nitric oxide (NO) diffusion.6,26,40,42 The definitive diagnosis is established by transbronchial biopsy.37
Treatment is based on the use of bronchodilators and corticosteroids.22,31,39 More aggressive immune suppressive therapy is indicated in cases showing a poor clinical course (cyclosporine, azathioprine or mofetil mycophenolate).34 If associated hypogammaglobulinemia is observed, protocolized intravenous immunoglobulin administration is indicated. On an empirical basis, erythromycin (intravenous and nebulized) is advised in the case of worsened breathing, as coadjuvant therapy.22,31,39 In patients with severe breathing difficulties, broad spectrum antibiotic treatment should be started on the grounds of probable overinfection.35
Bronchiolitis obliterans with organized pneumoniaBronchiolitis obliterans with organized pneumonia generally represents a complication of BO overinfected with a bacterium or virus.5,6,17,20,26,40,42 It manifests as an exacerbation of the initial condition, with clinical features similar to those of severe bronchospasm. The chest X-rays may show consolidations with an alveolar distribution that must be confirmed by thoracic CT.
Treatment is based on ventilatory support, antimicrobial use2,12 (against bacteria, fungi and viruses), and low-dose corticosteroids in the presence of clear wheezing with a poor response to therapy.22,31,39
Interstitial pneumoniaThe incidence of interstitial pneumonia is approximately 30%, and the underlying physiopathology is similar to that of idiopathic pneumonia. An established risk factor is the presence of chronic GVHD, in view of the inflammation and progressive fibrosis of the lung tissues that appear in graft disease.2,9,13,30,35
Fever associated to dry cough and tachypnea, with interstitial infiltrates on the chest X-rays are typical clinical–radiological findings in this disease. Any possible infectious origin must be discarded in all cases.29,38
Patient respiratory wellbeing must be ensured,43 with associated immune suppressor therapy (low-dose methylprednisolone, with escalation of the latter if no response is obtained when added to the rest of the immune suppressors used to treat GVHD).22,34
Delayed pulmonary toxicity syndromeDelayed pulmonary toxicity syndrome has an incidence of up to 72% in patients subjected to autologous BMT, with a very low mortality rate. The syndrome develops 2–4 months after grafting, and its good response to treatment and high incidence distinguish it from idiopathic pneumonia described above.2,30,35,40 The underlying risk factors comprise the administration of high doses of cyclophosphamide and cisplatin.2
The clinical picture generally consists of cough, dyspnea and fever. The diagnosis is suspected from the clinical manifestations of the patient. A characteristic of this syndrome is its good response to treatment.30,35 Infection must be discarded in all cases. Bilateral interstitial infiltrates are frequently observed on the chest X-rays, with a ground-glass image in the thoracic CT scan. When performed, the lung function tests show a decrease in carbon monoxide diffusion.2,7,35 No transbronchial biopsy is required if the clinical picture is clearly suggestive and the patient course proves favorable.37,44
Low-dose corticosteroid therapy (methylprednisolone) is advised in the acute phase, associated to broad spectrum antibiotic treatment. Inhalatory corticosteroid prophylaxis is recommended to avoid further episodes.22 Treatment with interferon-gamma is recommended in patients showing a poor clinical course.30
Pulmonary cytolytic thrombosisThis condition has been described mainly in a single hospital center in the United States. Pulmonary cytolytic thrombosis has a very low prevalence and is associated to Allogenic BMT. In the first stages of the disease the clinical picture is consistent with GVHD. It appears to be caused by minor hemorrhagic infarctions secondary to inflammatory involvement of the pulmonary endothelium. The disorder manifests by day 72 after transplantation, and has a good prognosis with practically no mortality.2,11,30
In addition to the manifestations of GVHD, the affected patients show signs such as fever and cough. There are usually no major breathing difficulties, and the chest X-ray findings are normal in 25% of the cases (nodules or atelectasis may be observed in some cases).2,11,30 The thoracic CT scan can reveal the presence of generally peripheral lung nodules. The possibility of infection must be discarded in all cases, with the obtainment of biopsy samples for the exclusion diagnosis.30
Both respiratory support and management of the complications (bleeding) are required. There is no clear etiological treatment. Low-dose methylprednisolone is indicated despite the fact that immunosuppressive therapy has not been clearly shown to be useful in this group of patients.22,39
Infectious complications in bone marrow transplant patientsAs has been commented above, in all BMT patients with breathing difficulties, we must consider the possibility of infection as the underlying cause.2,4,8–10,12,13,21,35 The immune condition of the patient exerts a crucial influence upon both the course and etiology of the disorder.10,22 We must establish the drug treatments (prophylaxis) and measures of asepsis and isolation required in order to avoid the development of such problems.10,13,35 Treatment in all cases should be based on ventilation support adapted to the clinical condition of the patient, management of the possible complications, and the administration of broad spectrum antimicrobial therapy targeted to bacteria, fungi and viruses.10,21,45
A description is provided below of the respiratory infections in patients with BMT.
Pulmonary infection caused by bacteria (bacterial pneumonia)The most prevalent causal bacteria are those that form part of the usual flora colonizing the gastrointestinal tract (gramnegative organisms, typically in the early neutropenic phase, and anaerobic bacteria) and the skin (grampositive organisms that invade the bloodstream associated to the use of central venous and/or epicutaneous catheters; in the presence of GVHD, the involvement of these bacteria must be discarded in all cases).2,46 Thorough clinical evaluation is required in order to locate possible infectious foci. The chest X-rays typically reveal consolidation images that should be confirmed by thoracic CT.2,7,9,12
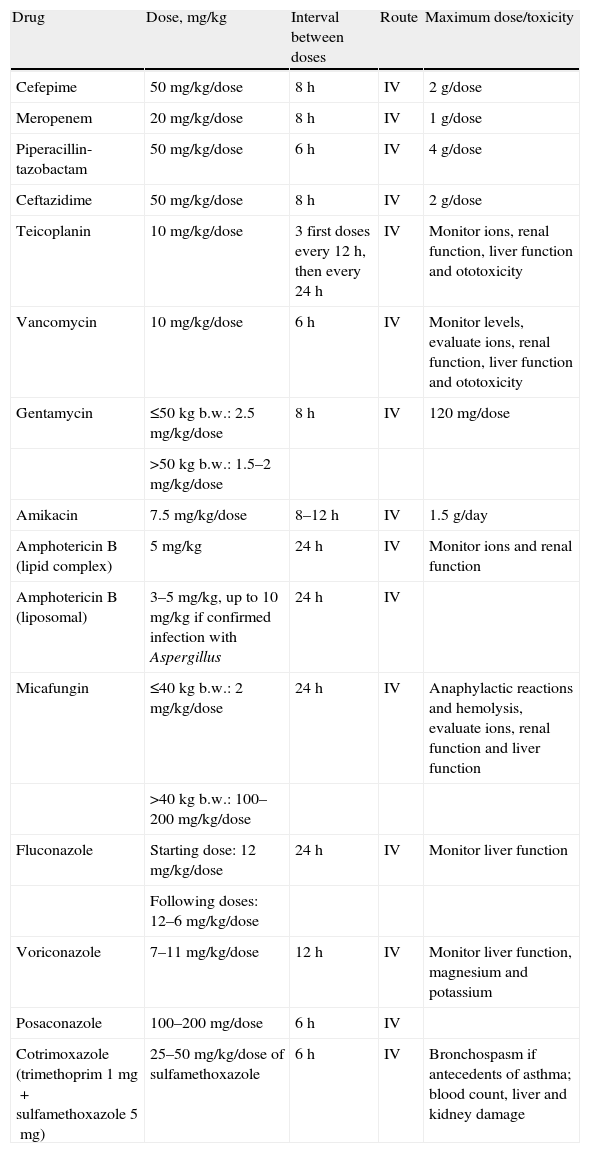
Broad spectrum antibiotics are indicated, using different drug combinations according to the suspected microorganisms involved.22 The antibiotic treatment used for preventive purposes should be replaced by some other treatment with a similar or expanded spectrum of action, due to possible resistance on the part of the causal organism (Table 4)2,7,9,12:
- -
Meropenem or cefepime associated to vancomycin in the case of probable infection caused by gramnegative or grampositive bacteria.
- -
Meropenem or cefepime associated to vancomycin and amikacin in the case of a probable gramnegative organism of abdominal origin.
- -
Addition of metronidazole in the event of possible infection caused by anaerobes (previous abdominal abscesses or cavitary pneumonia).
- -
Consideration of possible infection due to multiresistant Pseudomona spp. (use of cephalosporins with antipseudomonal activity).46–50
Empirical antifungal and antibacterial treatment.
| Drug | Dose, mg/kg | Interval between doses | Route | Maximum dose/toxicity |
| Cefepime | 50mg/kg/dose | 8h | IV | 2g/dose |
| Meropenem | 20mg/kg/dose | 8h | IV | 1g/dose |
| Piperacillin-tazobactam | 50mg/kg/dose | 6h | IV | 4g/dose |
| Ceftazidime | 50mg/kg/dose | 8h | IV | 2g/dose |
| Teicoplanin | 10mg/kg/dose | 3 first doses every 12h, then every 24h | IV | Monitor ions, renal function, liver function and ototoxicity |
| Vancomycin | 10mg/kg/dose | 6h | IV | Monitor levels, evaluate ions, renal function, liver function and ototoxicity |
| Gentamycin | ≤50kg b.w.: 2.5mg/kg/dose | 8h | IV | 120mg/dose |
| >50kg b.w.: 1.5–2mg/kg/dose | ||||
| Amikacin | 7.5mg/kg/dose | 8–12h | IV | 1.5g/day |
| Amphotericin B (lipid complex) | 5mg/kg | 24h | IV | Monitor ions and renal function |
| Amphotericin B (liposomal) | 3–5mg/kg, up to 10mg/kg if confirmed infection with Aspergillus | 24h | IV | |
| Micafungin | ≤40kg b.w.: 2mg/kg/dose | 24h | IV | Anaphylactic reactions and hemolysis, evaluate ions, renal function and liver function |
| >40kg b.w.: 100–200mg/kg/dose | ||||
| Fluconazole | Starting dose: 12mg/kg/dose | 24h | IV | Monitor liver function |
| Following doses: 12–6mg/kg/dose | ||||
| Voriconazole | 7–11mg/kg/dose | 12h | IV | Monitor liver function, magnesium and potassium |
| Posaconazole | 100–200mg/dose | 6h | IV | |
| Cotrimoxazole (trimethoprim 1mg+sulfamethoxazole 5mg) | 25–50mg/kg/dose of sulfamethoxazole | 6h | IV | Bronchospasm if antecedents of asthma; blood count, liver and kidney damage |
The duration of treatment should be individualized according to the course, microbiological findings, and background disease condition of the patient.
Candida spp. and Aspergillus spp. are the most prevalent fungal causes of infection in patients subjected to BMT,21,35 causing great morbidity and high mortality in the case of a poor response to treatment (approximately 90% in infections due to Aspergillus, according to some series).2 Prophylactic antifungal treatment with fluconazole has reduced the global number of episodes, though at the cost of selecting fungi with resistance to this drug.45,51,52
Infection produced by Aspergillus is always nosocomial and is related to the spore count in the air. This count must be monitored, and the opportune respiratory isolation measures should be adopted (positive flow in the rooms).45,51,52
A risk factor for fungal infection is a neutrophil count of under 100cells/μl during a period of more than 7 days. The affected patients tend to suffer cough, fever and progressive dyspnea. In the case of Aspergillus infection, these manifestations in turn are accompanied by pleuritic pain, hemoptysis and wheezing with a poor response to bronchodilator treatment.35
The chest X-ray findings initially may be normal—a thoracic CT scan being needed for both early diagnosis and for follow-up of the disease. In this context, the presence of lung nodules, cavitations with the sequestration of pulmonary parenchyma or the so-called halo sign are strongly suggestive of fungal infection.7 Biological samples (bronchoalveolar lavage, biopsy of the lesions) should be obtained for culture, with DNA amplification using polymerase chain reaction (PCR) techniques, or serological positivity suggesting the underlying etiology (e.g., galactomannan in the case of Aspergillus).2,45,53
The prevention of infection should be a priority concern. In this context, and as has been commented, the prophylactic use of fluconazole is selecting more virulent forms of non-albicans Candida with greater resistance to treatment.52,54,55 In general, the recommended first choice is empirical treatment with amphotericin B (liposomal or lipidic) or echinocandins. Drugs such as itraconazole, voriconazole and posaconazole are indicated in the case of a poor response to resistance.
- -
Liposomal amphotericin B: rapid fungicidal effects with good activity against many emerging fungi of clinical interest (particularly dematiaceous fungi).45,52,56,57
- -
Echinocandins (caspofungin, micafungin and anidulafungin): very broad spectrum antifungals that prove effective when used on an empirical basis to treat fungal infections. These drugs presently have a limited role in the treatment of emerging fungal species.45,52,56,57
- -
Broad spectrum triazolic antifungals: voriconazole and posaconazole are usually prescribed in combination with amphotericin B. The minimum inhibitory concentration (MIC) for resistant or emerging organisms is ≤0.125μg/ml, and is slightly higher for voriconazole, with no clear clinical significance. The references to MIC in the case of posaconazole are more limited. Due to the lack of clinical data in children, the choice of triazole drug should be established on an individualized basis.45,52,56,57
- -
Terbinafine: the in vitro activity of this drug covers many fungi capable of causing serious infections (hyaline fungi, dematiaceous fungi and dimorphic fungi). In turn, in vitro studies have reported good synergy between terbinafine and itraconazole or voriconazole in application to certain fungal species. Combination therapy is indicated in the case of resistant, disseminated or poorly evolving infections.45,52,56,57
In BMT, pulmonary disease of viral origin occurs as a result of the reactivation of a latent virus, treatment-induced immune suppression, or primary infection.2,10,13,35 In general terms, the pathogen most often responsible for lung damage in BMT is cytomegalovirus (CMV).28 The most common viral infections are summarized below.
CytomegalovirusCMV infection generally develops by day 50–60 post-transplantation. The associated mortality is high and greater in BMT than in solid organ transplantation. CMV disease is rare in autologous transplant patients, despite the fact that the overall infection rate in these cases is also high.28
Risk factors are patient seropositivity prior to BMT, patients close to adult age (increased risk of infection), previous radiotherapy, and severe GVHD.2
It is essential to determine the seropositivity status of both the donor and recipient prior to BMT. Gancyclovir prophylaxis is indicated up until day 100 post-transplantation. In the case of probable or established infection, it is advisable to administer gancyclovir associated to specific anti-CMV globulin.2,28,35
Herpes virusThese infections are generally the result of viral reactivation –the prior presence of mucositis being typical in such cases. In the case of primary infection, the patient shows the skin lesions characteristic of this virus. Risk factors are prolonged neutropenia and seropositivity prior to transplantation.2
The physical examination should evaluate the presence of skin lesions. Herpes virus produces two types of pneumonia, depending on the viral dissemination route involved: single or multiple focal pneumonia in the case of spread from the oropharynx, and diffuse pneumonia in the case of hematogenous dissemination.
High-dose acyclovir is indicated, and adequate prophylaxis is crucial in patients found to be seropositive before transplantation (Table 5).
Respiratory syncytial virus (RSV)Infection caused by RSV is associated with important morbidity–mortality, and represents a possible cause of idiopathic pneumonia. RSV infection develops in both autologous and allogenic BMT, and the associated risk factors are transplantation performed during a period in which the disease is endemic in the general population, the infection of other hospitalized individuals, and spread from hospital visits.2
The clinical picture is reminiscent of that seen in immunocompetent individuals (fever, cough, rhinorrhea and wheezing upon auscultation), with progressive breathing difficulties. The flu symptoms tend to appear before the manifestation of consistent radiological or other imaging alterations.10
The clinical manifestations are fundamental for establishing the diagnosis, and in this context the rapid RSV test in nasopharyngeal secretions is very useful.
The administration of palivizumab (monoclonal antibody) is indicated for the prevention of infection in BMT. Nebulized ribavirin can be used in patients with established infection and a poor response to treatment.22,35,58
Epstein–Barr virus (EBV) (post-transplantation lymphoproliferative disease)This infection is mainly related to allogenic BMT, with a lesser reported prevalence in autologous or peripheral stem cell transplantation. It is also frequently seen in the context of GVHD (due to the immune suppression needed to treat the latter) or after the infusion of T cell precursors depleted in the transplant.22,35 EBV produces pulmonary manifestations with nonspecific radiological alterations. The presence of disseminated adenopathies is common, together with enlargement of the solid organs that have a hematopoietic capacity (liver and spleen).2
Treatment is of a supportive nature, with the addition in all cases of rituximab (anti-CD20 monoclonal antibody), which causes destruction of the lymphocytes infected with the virus.22,35
Mycobacterial lung infectionsMycobacterial lung infections are very infrequent in BMT, and are more often found in solid organ transplants (in direct relation to diminished cellular immune function). Tuberculosis must be discarded in all patients programmed for BMT.22,35 The Mantoux test should be interpreted with caution, due to the immune suppression found in these patients, which increases the number of false-negative readings.2
Pneumocystis jirovecci lung infectionsThe incidence of these infections has decreased since the introduction of prophylactic trimethoprim-sulfamethoxazole therapy (lesser doses, with administration 2–3 days a week). The disease tends to manifest in the two months after transplantation, though cases within the first 14 days have also been reported. A typical finding is rapid progression of the breathing difficulties, with refractory hypoxemia and LDH elevation in the biochemical tests.22,35 The chest X-rays in turn reveal bilateral diffuse infiltrates that are conformed by the thoracic CT scan.
Treatment is based on the administration of cotrimoxazole, with adoption of the respiratory supportive measures needed to ensure patient wellbeing.2,22,35
Conflicts of interestThe authors have no conflicts of interest to declare.
Thanks are due to the physicians, nurses and auxiliary personnel of the Intensive Care Unit and Oncohematology Unit of our hospital.
Please cite this article as: García Salido A, et al. Dificultad respiratoria en el niño sometido a trasplante de médula ósea. Med Intensiva. 2011;35:569–77.