Respiratory distress is a common phenomenon in children with cancer. It is the most frequent cause of admission to the pediatric intensive care unit (PICU) in this group of patients. Its etiology is varied, and early and appropriate treatment is required. This review describes the most prevalent forms of respiratory distress in children with cancer without bone marrow transplantation. The symptoms, diagnosis and treatment are commented.
La dificultad respiratoria es un fenómeno común en el niño con cáncer y supone la causa más frecuente de ingreso en la unidad de cuidados intensivos pediátricos (UCIP) en este grupo de pacientes. Su etiología es múltiple y requiere un tratamiento adecuado y precoz. En esta revisión se describen los cuadros más frecuentes de dificultad respiratoria en el niño con cáncer no sometido a trasplante de médula ósea comentando su clínica, diagnóstico y tratamiento.
Breathing difficulties are frequent in children with cancer, and represent an important cause of admission to the pediatric intensive care unit (PICU) in patients of this kind.1–5 Such breathing difficulties may appear both at the time of diagnosis of the neoplastic disease—in some cases constituting its only symptom—or as a consequence of the treatment provided, or of the course and complications of the illness.2,3,6
The underlying etiology is varied and requires adequate and early treatment.4 The present review describes the most common forms of breathing difficulties in children with cancer, offering a description of their clinical manifestations, diagnosis and initial treatment. The study is divided into three parts: oncological emergencies, infections and complications of treatment (iatrogenesis). We have not included breathing difficulties in bone marrow transplant patients in this article, since in view of their specific characteristics, these will be dealt with in a separate review.
Oncological emergenciesOncological emergencies are all those acute breathing difficulty conditions derived from the existence of cancer.2–4,7 Such problems may appear both at the onset of the disease and during its subsequent course. In all cases the clinical manifestations are the basis for suspecting the diagnosis, and the main aim of management is always to secure the airway of the patient.8,9
Superior vena cava syndrome and superior mediastinal syndromeBoth superior vena cava syndrome and superior mediastinal syndrome are caused by problems of space secondary to tumor growth within a cavity exhibiting limited distensibility (thorax, mediastinum and neck region). These problems are generally associated to Hodgkin lymphomas, non-Hodgkin lymphomas,10 acute lymphoblastic leukemia (ALL, particularly type T), Ewing sarcoma (bone tumor metastases), teratomas, thyroid gland tumors, rhabdomyosarcomas and peripheral neuroectodermal tumors (PNETs).2–4,11–13
The clinical manifestations comprise progressive dyspnea and orthopnea. Patients may suffer headache, blurry vision, neck discomfort and chest pain.12 The cardinal signs of conditions of this kind are facial edema, plethora and, in some cases, perioral cyanosis. Cases of important airway obstruction in turn are accompanied by stridor and diaphoresis.1,2,4,11–14
A first diagnostic evaluation requires an X-ray study of the chest and neck to assess pulmonary ventilation and soft parts distribution. The X-ray study can also provide clues to the underlying cause, as when mediastinal widening is identified in cases of T type lymphomas.2 Thoracic computed tomography (CT) should be used to obtain an adequate anatomical description. Consideration is required of the possible existence of pleural and pericardial effusion associated to deficient venous return, and of possible alterations in cardiac contractility due to the space restrictions imposed upon the heart muscle.11,15
Management should ensure airway patency.9 We then require a histological diagnosis of the mass causing the breathing difficulties (fine needle aspiration biopsy or tissue biopsy).11,16 When a T-cell tumor is suspected, both pleurocentesis (thoracocentesis) and pericardiocentesis should be avoided as far as possible, due to the risk of hemodynamic decompensation associated with these techniques.13 Once the patient has been stabilized, adequate chemotherapy should be started as soon as possible, associated where necessary to local radiotherapy.2
HyperleukocytosisHyperleukocytosis is defined as the presence of over 100,000leukocytes/μl of peripheral blood.17 It is observed in 9–13% of all patients with ALL, and is also common in the chronic phase of myeloid leukemia2 (in leukemias of this kind, characterized by an increased blast cell depositing and aggregation tendency, the risk of bleeding complications is much higher18). As a result of the increased cell presence in the bloodstream, blood viscosity is seen to increase—this in turn facilitating leukostasis in zones with small-diameter capillaries.19 If this occurs within the lung tissues, microcirculatory damage results, with the formation of leukocyte aggregates and bleeding secondary to endovascular damage.19
Dyspnea and hypoxemia appear as a result of the alterations in gas exchange caused by the vascular damage. Right-side heart failure may develop even in the absence of severe lung damage, due to the increase in pulmonary capillary pressure caused by the generated blood stasis. Evaluation is also required of the possible involvement of other organs and systems through mechanisms similar to those underlying the lung damage (thrombosis of the lower extremities or cerebrovascular events).19
The diagnosis of hyperleukocytosis is confirmed from the complete blood count, though initial suspicion is based on the clinical picture. Chest X-rays are usually of little help, except in cases of important lung edema secondary to heart failure. Doppler ultrasound evaluation should be considered when suspecting alterations in blood flow in other body regions. Likewise, when cerebrovascular events or stroke are suspected, imaging techniques are indicated (cranial CT in the acute phase, or magnetic resonance imaging during the course of the disorder).
The main objective of treatment should be to reduce blood viscosity. To this effect, hyperhydration is essential, through the intravenous perfusion of low-saline glucose solutions (3l per square meter of body surface in 24h).9,20,21 Attention should center on the concentrations of the different ions, due to the risk of tumor lysis syndrome associated to cell rupture (monitoring potassium and renal function).22 Both leukapheresis and replacement transfusion have been successfully used in these situations.2,21 At present there is no clear protocol for the application of these two techniques—the latter being recommended in the presence of a compatible clinical picture and poor patient response to initial supportive therapy provided.20
Lung or airway dysfunction due to tumor presenceLung or airway dysfunction appears in the presence of space-occupying masses within the thoracic cavity (typical of teratoma and ganglioneuroma), or of partial or complete obstruction of the upper airway (nasal fibroma, hemangioma, fibromatosis or adenopathies in the case of ALL or lymphoma).2–4 The oncological diseases that produce these problems are usually slow growing—thus allowing good tolerance even up to the time of diagnosis. They do not give rise to alterations in venous return such as those seen in patients with superior vena cava syndrome or superior mediastinal syndrome (commented above); rather, air passage is obstructed by direct tumor compression.15
The initial clinical manifestations consist of mild and nonspecific symptoms and signs (dry cough, dry throat, nocturnal snoring23) that progress towards breathing difficulties with manifest dyspnea and even stridor in the case of severe airway compression.2,7 Restrictive pulmonary insufficiency may develop secondary to the diminished space within the chest cavity (some abdominal tumors, as in the case of large hepatomegalia associated to stage IVs neuroblastoma, can also give rise to restrictive phenomena similar to those produced by masses located in the abdominal cavity2).
The diagnosis is based on imaging techniques that allow us to estimate the size of the tumors and the degree of compression caused by them (chest X-rays, ultrasound and pulmonary CT).2,3,24 A precise diagnosis is required, since primary lung tumors are very infrequent in children, and their detection may modify both the initial approach to the case and its posterior treatment.15 In the presence of breathing difficulties without thoracic involvement, it must be remembered that patients with tumors of the central nervous system can suffer hypoventilation as a result of damage to the respiratory nuclei located at bulbar level.2
Management should afford respiratory wellbeing.2,9 Chemotherapy is to be started early, and in some cases emergency surgery may prove necessary.7,15 Orotracheal intubation may be required to secure the airway; due to the restrictive pattern found in these cases, both intubation and posterior ventilation may prove difficult.2
InfectionsOncological treatment gives rise to partial or complete patient immune suppression.25 This in turn facilitates and increases the development of infections, particularly in neutropenic children (<500 total neutrophils/μl of peripheral blood2).
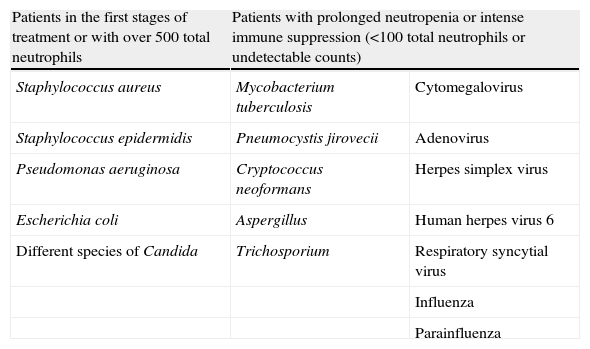
Respiratory infections in children with cancer are of a varied and complex etiology,26 and give rise to important morbidity–mortality27,28 (up to 80% of the cases with associated clinical manifestations of sepsis2,29). Due to the prolonged hospital stays and poor background immune condition of these patients, nosocomial pathogens are frequent causal agents.27,28 In practical terms, we can distinguish two groups of patients according to their degree of immune suppression (Table 1):
- •
Patients in the early phases of treatment or with over 500 total neutrophils: suspicion should focus on gramnegative30 (colonization of the airway by bacteria present in the digestive tract) and grampositive pathogens (generally staphylococci or streptococci), present in the skin, and which cause infection through central venous catheters.9,31
- •
Patients with prolonged neutropenia or severe immune suppression (under 100 total neutrophils, or undetectable counts): we add fundamentally opportunistic agents,30 such as fungal (Candida as the most common species, and Aspergillus as an example of fungal infection with important mortality and complications32–34) and viral infections (herpes simplex virus or adenovirus). In all these cases, early and adequate empirical treatment should be started.9,28
Frequent causal agents of respiratory infection in children with cancer.
| Patients in the first stages of treatment or with over 500 total neutrophils | Patients with prolonged neutropenia or intense immune suppression (<100 total neutrophils or undetectable counts) | |
| Staphylococcus aureus | Mycobacterium tuberculosis | Cytomegalovirus |
| Staphylococcus epidermidis | Pneumocystis jirovecii | Adenovirus |
| Pseudomonas aeruginosa | Cryptococcus neoformans | Herpes simplex virus |
| Escherichia coli | Aspergillus | Human herpes virus 6 |
| Different species of Candida | Trichosporium | Respiratory syncytial virus |
| Influenza | ||
| Parainfluenza | ||
As a result of the existing immune suppression, few manifestations of inflammation are observed (with no important leukocytosis or increases in acute phase reactants)—the only signs and symptoms being discrete dyspnea, cough or mild chest discomfort.2,35,36
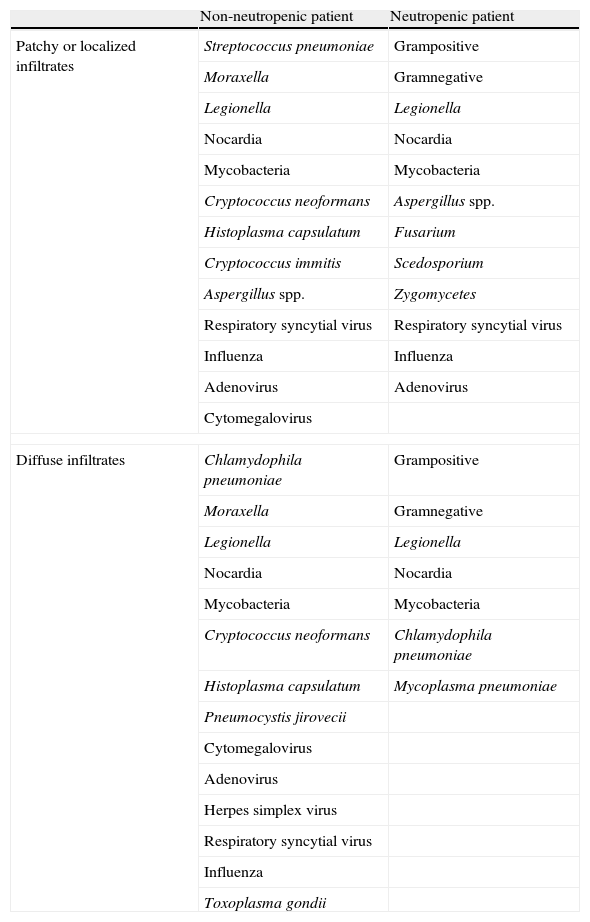
Chest X-rays are useful only for evolutive monitorization of the disease, since there may be no initial changes because of the incapacity to generate consolidations and leukocyte infiltrates.28,32 If such changes do appear, their distribution may be useful for establishing a tentative diagnosis (Table 2).2,37 Thoracic CT is the instrument of choice for establishing an early diagnosis, since it is able to identify alterations even in the presence of normal plain X-ray findings.2,28,32
Immune condition, type of infiltrate and probable causal agent of respiratory infection.
| Non-neutropenic patient | Neutropenic patient | |
| Patchy or localized infiltrates | Streptococcus pneumoniae | Grampositive |
| Moraxella | Gramnegative | |
| Legionella | Legionella | |
| Nocardia | Nocardia | |
| Mycobacteria | Mycobacteria | |
| Cryptococcus neoformans | Aspergillus spp. | |
| Histoplasma capsulatum | Fusarium | |
| Cryptococcus immitis | Scedosporium | |
| Aspergillus spp. | Zygomycetes | |
| Respiratory syncytial virus | Respiratory syncytial virus | |
| Influenza | Influenza | |
| Adenovirus | Adenovirus | |
| Cytomegalovirus | ||
| Diffuse infiltrates | Chlamydophila pneumoniae | Grampositive |
| Moraxella | Gramnegative | |
| Legionella | Legionella | |
| Nocardia | Nocardia | |
| Mycobacteria | Mycobacteria | |
| Cryptococcus neoformans | Chlamydophila pneumoniae | |
| Histoplasma capsulatum | Mycoplasma pneumoniae | |
| Pneumocystis jirovecii | ||
| Cytomegalovirus | ||
| Adenovirus | ||
| Herpes simplex virus | ||
| Respiratory syncytial virus | ||
| Influenza | ||
| Toxoplasma gondii | ||
Adequate support of the clinical condition of the patient is required, with the optimization of oxygenation. Respiratory wellbeing is to be sought, reducing the existing dyspnea as far as possible (mask with a reservoir or noninvasive positive pressure ventilation).2,3,35,38 When mechanical ventilation is required, specific ventilation maneuvers are usually needed, with high oxygen output, high pressure peaks, and peak end expiratory pressure (PEEP) values higher than those usually employed (between 6 and 10cm H2O).
Associated early empirical treatment is essential, combining antibiotics, antifungal agents and antiviral drugs.39 The most commonly used drugs are specified below (Table 3):
- •
Antibiotics: broad spectrum betalactams are indicated, such as cefepime, meropenem or imipenem39,40 combined in the case of suspected grampositive infection with a glycopeptide (vancomycin).27,31,35,41 In the case of important hypoxemia without major radiological alterations, treatment with trimethoprim sulfamethoxazole should be considered, due to possible infection with Pneumocystis jirovecii.42
- •
Antifungals: a common first choice is fluconazole (generally used as prevention against fungal disease, and not useful in the case of Aspergillus infection), which should be replaced by amphotericin B (liposomal or lipidic), micafungin or voriconazole when suspecting infection by a fungus other than Candida, or in the case of a poor patient response to the initial empirical treatment provided.27,32,36
- •
Antivirals: these drugs are indicated in the case of great immune suppression or when viral infection is suspected. Ribavirin is indicated in the case of respiratory syncytial virus (RSV) infection,43 and empirical treatment with oseltamivir is advised in the presence of influenza or pseudoinfluenza symptoms. It is also common to use ganciclovir or aciclovir in the case of possible pneumonia due to herpes simplex virus.27,32,36
Empirical treatment of respiratory infections in children with cancer.47–49
| Clinical picture | Recommended antimicrobials (associate one of each group) |
| Patient with breathing difficulties | Cefepime, meropenem, imipenem, piperacillin-tazobactam |
| Vancomycin, linezolid | |
| Amikacin | |
| Patient with breathing difficulties and allergy to betalactams | Aztreonam |
| Amikacin | |
| Vancomycin, teicoplanin, linezolid | |
| Patient with breathing difficulties unresponsive to empirical treatment or with suspected fungal infection | Add to empirical antibiotic treatment: amphotericin B in lipid formulation, micafungin (echinocandin) or voriconazole |
| Patient with breathing difficulties and clinical manifestations consistent with viral infection | Add to empirical antibiotic treatment: ribavirin and palivizumab (RSV), oseltamivir (influenza virus), cidofovir or ganciclovir (adenovirus), ganciclovir or foscarnet (herpes virus type 6), aciclovir (herpes simplex virus) |
Consider the administration of hematopoietic growth factors in patients with intense neutropenia.
Oncological treatment generally implies important aggression for the body. The drugs, radiotherapy and surgical procedures to which these patients are exposed12 can directly give rise to breathing difficulties.
Radiotherapy, as a result of the associated inflammation and posterior cicatrization, produces restrictive symptoms through direct action upon the lung parenchyma and surrounding structures, and alterations in gas diffusion across the alveolar-capillary membrane or barrier (the latter being destroyed and replaced by connective tissue).44,45 The clinical manifestations are rarely of an acute nature—the typical symptoms being dry cough with episodes that can be confused with bronchospasm.2 A fundamental element of care is the use of anti-inflammatory agents to improve the respiratory conditions of the affected children (inhalatory corticosteroids such as budesonide, for chronic prophylactic treatment, or systemic corticosteroids such as methylprednisolone, for the acute conditions).46
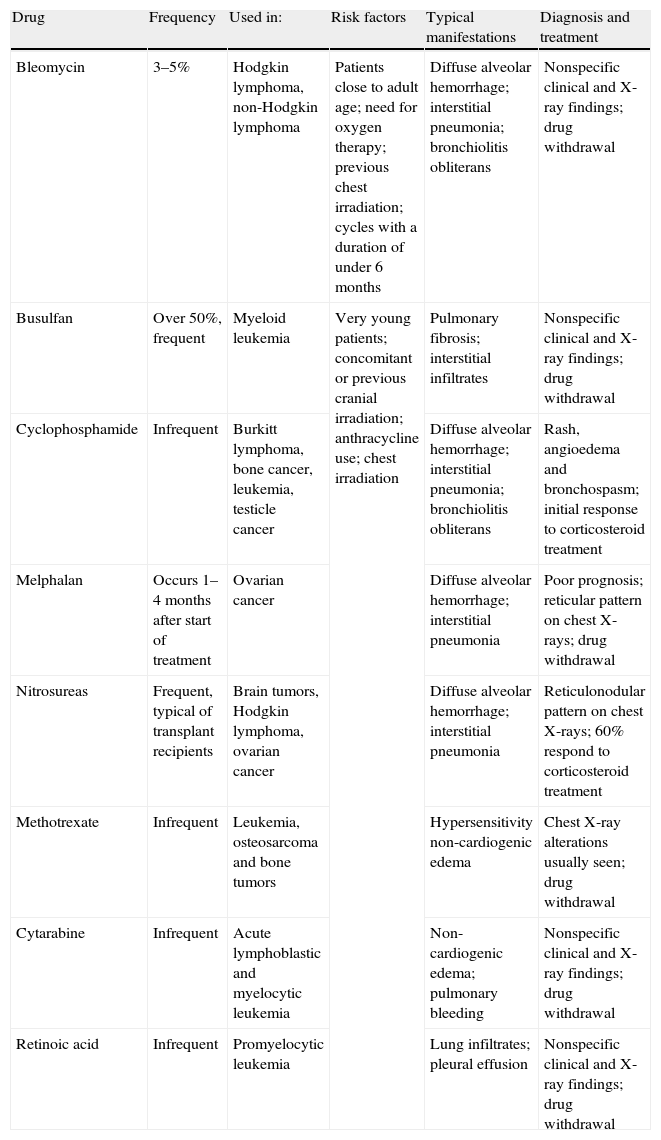
Chemotherapy is able to produce respiratory symptoms from the start of administration.44,45 A high degree of suspicion must be observed, since dyspnea is rarely attributed to cytostatic drug use, due to the fact that the previously described complications are comparatively much more frequent. It is therefore useful to be familiarized both with the typical presentations of this condition (Table 4) and with the factors that facilitate its development (very young patients, the use of concomitant or previous cranial irradiation, anthracycline use, and the application of chest irradiation).2 Management generally involves suspension of the drug treatment and provision of the supportive measures needed in each individual case (Table 4).2,44,45,50
Breathing difficulties due to chemotherapy.
| Drug | Frequency | Used in: | Risk factors | Typical manifestations | Diagnosis and treatment |
| Bleomycin | 3–5% | Hodgkin lymphoma, non-Hodgkin lymphoma | Patients close to adult age; need for oxygen therapy; previous chest irradiation; cycles with a duration of under 6 months | Diffuse alveolar hemorrhage; interstitial pneumonia; bronchiolitis obliterans | Nonspecific clinical and X-ray findings; drug withdrawal |
| Busulfan | Over 50%, frequent | Myeloid leukemia | Very young patients; concomitant or previous cranial irradiation; anthracycline use; chest irradiation | Pulmonary fibrosis; interstitial infiltrates | Nonspecific clinical and X-ray findings; drug withdrawal |
| Cyclophosphamide | Infrequent | Burkitt lymphoma, bone cancer, leukemia, testicle cancer | Diffuse alveolar hemorrhage; interstitial pneumonia; bronchiolitis obliterans | Rash, angioedema and bronchospasm; initial response to corticosteroid treatment | |
| Melphalan | Occurs 1–4 months after start of treatment | Ovarian cancer | Diffuse alveolar hemorrhage; interstitial pneumonia | Poor prognosis; reticular pattern on chest X-rays; drug withdrawal | |
| Nitrosureas | Frequent, typical of transplant recipients | Brain tumors, Hodgkin lymphoma, ovarian cancer | Diffuse alveolar hemorrhage; interstitial pneumonia | Reticulonodular pattern on chest X-rays; 60% respond to corticosteroid treatment | |
| Methotrexate | Infrequent | Leukemia, osteosarcoma and bone tumors | Hypersensitivity non-cardiogenic edema | Chest X-ray alterations usually seen; drug withdrawal | |
| Cytarabine | Infrequent | Acute lymphoblastic and myelocytic leukemia | Non-cardiogenic edema; pulmonary bleeding | Nonspecific clinical and X-ray findings; drug withdrawal | |
| Retinoic acid | Infrequent | Promyelocytic leukemia | Lung infiltrates; pleural effusion | Nonspecific clinical and X-ray findings; drug withdrawal |
The authors have no conflict of interest to declare.
Thanks are due to the physicians, nurses and auxiliary personnel of the Intensive Care Unit and Oncohematology Unit of our hospital.
Please cite this article as: García Salido A, et al. Dificultad respiratoria en el niño con cáncer. Med Intensiva. 2011;35:562–8.