To determine the degree of antiviral treatment recommendations adherence and its impact to critical ill patients affected by influenza A(H1N1)pdm09 mortality.
DesignSecondary analysis of prospective study.
SettingIntensive care (UCI).
PatientsPatients with influenza A(H1N1)pdm09 in the 2009 pandemic and 2010–11 post-pandemic periods.
VariablesAdherence to recommendations was classified as: total (AT); partial in doses (PD); partial in time (PT), and non-adherence (NA). Viral pneumonia, obesity and mechanical ventilation were considered severity criteria for the administration of high antiviral dose. The analysis was performed using t-test or “chi” square. Survival analysis was performed and adjusted by Cox regression analysis.
ResultsA total of 1058 patients, 661 (62.5%) included in the pandemic and 397 (37.5%) in post-pandemic period, respectively. Global adherence was achieved in 41.6% (43.9% and 38.0%; p=0.07, respectively). Severity criteria were similar in both periods (68.5% vs. 62.8%; p=0.06). The AT was 54.7% in pandemic and 36.4% in post-pandemic period, respectively (p<0.01). The NA (19.7% vs. 11.3%; p<0.05) and PT (20.8% vs. 9.9%; p<0.01) were more frequent in the post-pandemic period. The mortality rate was higher in the post-pandemic period (30% vs. 21.8%; p<0.001). APACHE II (HR=1.09) and hematologic disease (HR=2.2) were associated with a higher mortality and adherence (HR=0.47) was a protective factor.
ConclusionsA low degree of adherence to the antiviral treatment was observed in both periods. Adherence to antiviral treatment recommendations was associated with lower mortality rates and should be recommended in critically ill patients with suspected influenza A(H1N1)pdm09.
Evaluar el grado de adherencia a las recomendaciones sobre el tratamiento antivírico y su impacto en la mortalidad de pacientes críticos afectados por gripe A (H1N1)pdm09.
DiseñoAnálisis secundario de estudio prospectivo.
ÁmbitoMedicina intensiva (UCI).
PacientesPacientes con gripe A (H1N1)pdm09 en el periodo pandémico 2009 y pospandémico 2010–11.
VariablesLa adherencia a las recomendaciones se clasificó en: total (AT), parcial dosis (PD), parcial tiempo (PT) y no adherencia (NA). La neumonía vírica, obesidad y ventilación mecánica fueron considerados criterios de gravedad para el uso de dosificaciones elevadas de antivírico (CG). Análisis mediante «chi» cuadrado y t-test. Supervivencia mediante regresión de Cox.
ResultadosSe incluyeron 1.058 pacientes, 661 (62,5%) en pandemia y 397 (37,5%) en pospandemia. La AT global del estudio fue del 41,6% (el 43,9% y el 38%, respectivamente; p=0,07). Los pacientes con criterios de gravedad no fueron diferentes en ambos periodos (un 68,5% y un 62,8%; p=0,06). En estos pacientes la AT fue del 54,7% durante el 2009 y del 36,4% en pospandemia (p<0,01). La NA (19,7% vs. 11,3%; p<0,05) y la PT (20,8% vs. 9,9%; p<0,01) fueron más frecuentes durante la pospandemia. La mortalidad fue mayor en la pospandemia (30% vs. 21,8%; p<0,001). El APACHE II (HR=1,09) y la enfermedad hematológica (HR=2,2) se asociaron a mortalidad y la adherencia (HR=0,47) fue un factor protector.
ConclusionesSe evidencia un bajo grado de adherencia al tratamiento en ambos periodos. La adherencia al tratamiento antivírico se asocia con menor mortalidad y debería ser recomendada en pacientes críticos afectados por gripe A (H1N1)pdm09.
On facing the influenza A(H1N1) pandemic of early 2009, the health authorities planned communication strategies, ethical decision making protocols, the stockpiling of antivirals and improvement of the capacity of the health system to offer adequate care for a large number of patients. In this regard, guidelines were traced and strategies were defined, pooling the evidence available up to that time, with a view to optimizing antiviral therapy. In Spain, with the purpose of guaranteeing adequate and homogeneous care for adults with severe influenza A(H1N1)pdm09 disease,1,2 the Ministry of Health designed an intervention protocol3 based on the recommendations of the World Health Organization (WHO).4,5 However, the mentioned protocol was established on the basis of the efficacy data of the neuraminidase inhibitors obtained from studies in patients with only mild or moderate forms of the disease. As a result, the true impact of antiviral treatment in critically ill patients was difficult to calibrate, due to the lack of randomized studies in this special group of patients.
During the pandemic period, different health organizations4–7 agreed on the need to administer early antiviral treatment in the form of oseltamivir to all patients requiring hospital admission, with progressing serious disease, or who developed complications. This strategy was to be applied independently of the previous health condition or vaccination history of the patient. In this context, the recommended oseltamivir dose for adults was 75mg twice a day via the oral route. On the other hand, it was considered that certain patient groups might need a higher dose (300mg/day),6,8,9 due to a possible decrease in intestinal absorption of the drug and/or an increase in distribution volume when mechanical ventilation proved necessary and/or in obese individuals. The duration of the antiviral treatment in patients with mild or moderate disease was limited to 5 days (the viral elimination time),7–9 while in critical patients, where the viral elimination period could be significantly prolonged,6,8,9 a longer treatment course of 10 days was advised.
The degree of global adherence to the clinical guidelines is highly variable but generally low.10–12 The antiviral treatment guides during the pandemic period 2009 and in the post-pandemic period 2010–2011 were applied according to the criteria of each Spanish Autonomous Community, each health center, and each individual prescriber. We therefore postulate that there is a considerable difference between the treatment recommendations of the health authorities and the way in which they are actually implemented in clinical practice. Despite the many publications on influenza A(H1N1)pdm09, the degree of adherence to the antiviral treatment guides during the influenza A pandemic is not known. We therefore aimed to evaluate the degree of adherence to the antiviral treatment recommendations in critical patients with influenza A infection in two periods, pandemic and post-pandemic, and to explore the association among adherence, risk factors and mortality.
Materials and methodsA secondary analysis of a prospective, multicenter observational study was carried out.
Study populationThe study population consisted of two adult cohorts with confirmed influenza A(H1N1)pdm09 infection and admitted to 148 Spanish Intensive Care Units (ICUs). Other influenza viruses (H3N2 or B) were not included, since the circulation of virus A (H1N1)pdm09 was predominant during the study periods.
Inclusion and exclusion criteriaWe included all patients with clinical data compatible with influenza and who presented fever (>38°C), consistent respiratory symptoms characterized by cough, sore throat, flu-type disease or muscle pain, acute respiratory failure, and with microbiological confirmation of influenza A(H1N1)pdm09 infection. Patients under 15 years of age were excluded, as were those individuals with clinical manifestations compatible with flu syndrome but without microbiological confirmation of influenza A(H1N1)pdm09 infection.
Two periods were analyzed: (1) one patient cohort admitted between epidemiological weeks 23–52 of the year 2009, corresponding to the pandemic; and (2) another patient cohort during the post-pandemic period, which registered the cases diagnosed between epidemiological weeks 50–52 of the 2010 and weeks 1–9 of 2011.
Data collectionThe data were obtained from a voluntary registry created by the Spanish Society of Intensive and Critical Care Medicine and Coronary Units (Sociedad Española de Medicina Intensiva, Crítica y Unidades Coronarias, SEMICYUC). The study was approved by the Institutional Review Board of the hospital coordinating the project (IRB NEUMAGRIP/11809). The identity of the patients was kept anonymous, and the obtainment of informed consent was not considered necessary, due to the observational nature of the study and the sanitary urgency involved.
The data were reported by the supervising physician in each participating center using a voluntary registry form. The confirmation of influenza A(H1N1)pdm09 infection was made by real-time PCR using nasopharyngeal smears or tracheal secretion samples in each institution or on a centralized basis in a reference (core) laboratory. The criteria for admission to the ICU and the decisions referred to treatment for all the patients–including the need for intubation and the type of antibiotic or antiviral treatment provided–were established by the supervising physician.
Septic shock and organ failure (SOFA score) were established following the criteria of the American College of Chest Physicians and the Society of Critical Care Medicine.13,14 Primary viral pneumonia was defined by the presence of acute breathing difficulty and alveolar opacity in two or more lobes, with negative respiratory and blood cultures during the acute phase of influenza virus infection.12 Community-acquired respiratory coinfection was defined as a bacterial or fungal infection diagnosed within the first two days of hospital stay.14 Those infections occurring after the first 48h of stay were taken to be nosocomial processes.
Hematological disease was defined as that manifesting in patients with acute lymphoblastic leukemia, acute myeloblastic leukemia, chronic lymphocytic leukemia, chronic myeloid leukemia, Hodgkin lymphoma, non-Hodgkin lymphoma, myeloma, or graft- vs -host disease.12
Obese individuals were taken to be those with a body mass index (BMI) of >30kg/m2.15
Those patients previously vaccinated against influenza A(H1N1)pdm09 with the monovalent vaccine or the polyvalent vaccine for seasonal influenza 2010–2011 were defined as “vaccinated”. Acute renal failure and its grades were diagnosed according to the Acute Kidney Injury (AKI) criteria.16
As regards antiviral therapy, “early” treatment was taken to be therapy started within 48h or less after the onset of symptoms.3
Patients with “severity criteria” were defined as those presenting a combination of pneumonia, obesity (BMI>30kg/m2) and/or invasive mechanical ventilation following the recommendations on antiviral treatment of the SEMICYUC. According to these recommendations, this group of patients could benefit from the administration of oseltamivir at high doses (150mg/12h) and with treatment durations of over 10 days, on the grounds of the existence of an increased distribution volume. For the rest of the patients, i.e., “without severity criteria”, standard doses and durations were recommended (75mg/12h during less than 10 days).3
The variable “adherence” to the recommendations of antiviral treatment was established from the combination of the variables “severity criteria” and the duration and dosage of the antiviral treatment prescribed for each patient. In this way we established four degrees of adherence, as shown in Table 1. We also studied adherence as a dichotomic variable where “total adherence” (TA) corresponded to patients who had received the treatment as recommended in terms of both dose and duration, while the rest of the patients were considered to show “non-adherence” (NA).
Classification of the degrees of adherence to the antiviral treatment protocols.
| Adherence variable | Definition |
|---|---|
| Total adherence | Dose of 150mg/12h and duration ≥10 daysaDose of 75mg/12h and duration <10 daysb |
| Partial adherence in dose | Dose of 150mg/12h but duration <10 daysaDose of 75mg/12h but duration ≥10 daysb |
| Partial adherence in duration | Dose of 75mg/12h and duration ≥10 daysaDose of 150mg/12h and duration <10 daysb |
| Non-adherence | Dose of 75mg/12h and duration of <10 daysaDose of 150mg/12h and duration ≥10 daysb |
Categorical variables were reported as values (percentages), and continuous variables as the mean±standard deviation (SD) or median with the corresponding 25–75 interquartile ranges. With regard to the demographic and clinical characteristics of the patients, the differences between groups were evaluated using the chi-squared test or the Fisher exact test for categorical variables, and the Student t-test or Mann–Whitney U-test for continuous variables, as appropriate. The impact of the different variables upon mortality was analyzed using Cox regression analysis.17,18 In order to avoid false associations, the choice of variables entered in the analysis was decided by statistical criterion for those variables showing significance in the univariate analysis (p<0.1), or based on the criterion of potential clinical relevance.
In order to assess the possibility of a differential impact of the explanatory variables upon the results of each period, the interactions between the explanatory variables and periods were used in the models. The results are presented as hazard ratios (HRs), with 95% confidence intervals (95%CI).
Lastly, a post hoc analysis was made, excluding those patients who died before the adherence objectives could be reached. In this way we specifically analyzed adherence to the recommendations only in those patients who had completed treatment, on a per protocol (PP) basis.
Statistical significance was considered for p<0.05. The SPSS version 16.0 statistical package for MS Windows (SPSS, Chicago, IL, USA) was used throughout.
ResultsThe present analysis included a total of 1058 patients admitted to ICUs with confirmed influenza A(H1N1)pdm09 infection: 661 patients (62.5%) were individuals infected during the pandemic of 2009, and 397 (37.5%) were patients corresponding to the period 2010–2011 (post-pandemic). The variables related to antiviral treatment evaluated for each period are shown in Table 2. The patients of the post-pandemic period were comparatively in more serious condition and had greater organ dysfunction and a higher frequency of shock, renal failure, and need for mechanical ventilation and dialysis. They also showed greater delays in diagnosis and admission to both hospital and the ICU. Regarding treatment, the post-pandemic period was characterized by a greater delay in the start of antiviral medication (one day) and a lesser indication of early empirical therapy, as well as a generally lesser use of high doses, and shorter durations of antiviral treatment.
Comparison of different variables defining antiviral treatment between the two periods.
| Variable | Pandemic 2009, n=661 | Pandemic 2010–2011, n=397 | p-Value |
|---|---|---|---|
| Severity, mean (SD) | |||
| APACHE II | 13.9 (7.2) | 16.2 (7.7) | <0.001 |
| SOFA | 5.7 (3.6) | 6.2 (4.0) | 0.03 |
| Clinical manifestations, n (%) | |||
| Primary viral pneumonia | 546 (69.5) | 267 (67.3) | 0.4 |
| Exacerbated COPD | 36 (5.5) | 24 (6.1) | 0.8 |
| Bacterial coinfection | 99 (15.2) | 76 (19.2) | 0.1 |
| Care variables, mean (SD) | |||
| Mean stay in ICU | 13.53 (14.34) | 13.88 (13.82) | 0.69 |
| Mean stay in hospital | 20.8 (17.83) | 20.01 (16.97) | 0.47 |
| Days from symptoms onset to hospital admission | 4.28 (2.7) | 4.92 (3.4) | 0.02 |
| Days of hospital stay to admission to ICU | 1.86 (2.1) | 2.37 (3.8) | 0.016 |
| Days of hospital stay to diagnosis | 2.32 (2.1) | 6.69 (3.9) | <0.001 |
| Evolutive indicators, n (%) | |||
| Septic shock | 295 (45.5) | 224 (56.4) | 0.001 |
| Mechanical ventilation | 471 (71.3) | 326 (82.1) | <0.001 |
| Invasive mechanical ventilation | 404 (61.2) | 269 (66.8) | 0.02 |
| Prone decubitus | 95 (14.7) | 82 (20.7) | 0.01 |
| Acute renal failure | 136 (21.7) | 112 (39.3) | 0.005 |
| Dialysis | 20 (3.1) | 29 (7.3) | 0.002 |
| Mortality in ICU | 144 (21.8) | 119 (30) | 0.003 |
| Treatment | |||
| Treatment with oseltamivir, n (%) | 643 (99.1) | 383 (96.7) | 0.008 |
| Treatment with zanamivir, n (%) | 4 (0.69) | 25 (6.3) | <0.001 |
| Empirical antiviral treatment, n (%) | 463 (72.9) | 274 (71) | 0.517 |
| Days from symptoms onset to antiviral administration, mean (SD) | 4.7 (2.9) | 5.7 (3.6) | <0.001 |
| Early antiviral treatment (<48h), n (%) | 153 (23.9) | 61 (15.8) | 0.002 |
| Use of high antiviral drug doses, n (%) | 453 (72.9) | 212 (55.4) | <0.001 |
| Duration of antiviral treatment, mean (SD) | 9.6 (4.26) | 8.88 (3.8) | 0.005 |
| Adherence to treatment protocol, n (%) | 267 (43.9) | 145 (38) | 0.075 |
Global total adherence in the study was 41.6% and was greater during the pandemic period (43.9%) vs the period 2010–2011 (38%), though the difference failed to reach statistical significance (p=0.07).
On analyzing the degree of adherence according to the two patient categories established in the protocol (presence or absence of severity criteria), the percentage of patients with “severity criteria” requiring high doses was seen to be greater during the post-pandemic period (68.5%) than during the pandemic period (62.8%), though statistical significance was not reached (p=0.06). Table 3 shows the different degrees of adherence according to the presence or absence of severity criteria in both study periods. “Total adherence” was 54.7% for 2009 and decreased significantly to 36.4% during 2010–2011. On the other hand, non-adherence and partial adherence in time (duration) were more frequent in the post-pandemic period. In contrast to the situation in patients with severity criteria, those without such criteria showed a greater degree of “total adherence” during the post-pandemic period.
Degrees of adherence in each period for patients with and without severity criteria.
| Patients with severity criteria: high dose | |||
|---|---|---|---|
| Degrees of adherence: n (%) | Pandemic 2009, n=380 | Period 2010–2011, n=264 | p-Value |
| Total adherence | 208 (54.7) | 96 (36.4) | <0.001 |
| Partial adherence in dose | 84 (22.1) | 61 (23.1) | 0.774 |
| Partial adherence in duration | 47 (11.3) | 52 (19.7) | 0.014 |
| Non-adherence | 41 (9.9) | 55 (20.8) | 0.007 |
| Patients without severity criteria: standard dose | |||
|---|---|---|---|
| Degrees of adherence: n (%) | Pandemic 2009, n=228 | Period 2010–2011, n=118 | p-Value |
| Total adherence | 59 (25.9) | 49 (41.5) | 0.003 |
| Partial adherence in dose | 18 (7.9) | 15 (12.7) | 0.177 |
| Partial adherence in duration | 89 (39) | 32 (27.1) | 0.032 |
| Non-adherence | 62 (27.2) | 22 (18.6) | 0.08 |
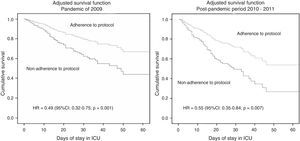
The mortality rate was significantly higher (p=0.001) in the patients of the post-pandemic period 2010–2011 (n=119; 30%) than in those of the pandemic period (n=144; 21.8%)–this being associated to increased patient severity as scored by the APACHE II (16.2 [7.7] vs 13.9 [7.2]; p<0.001) and increased organ dysfunction (SOFA score upon admission 6.2 [4.0] vs 5.7 [3.6]; p=0.03) for that period. Table 4 shows the variables associated to mortality in both study periods. On including the variables showing statistical significance in the multivariate analysis, we found the APACHE II score, hematological disease and adherence to treatment to be the variables independently associated to mortality (Table 5). On the other hand, early antiviral treatment was only found to be associated to survival in the pandemic period, while human immunodeficiency virus (HIV) infection was associated to survival in the seasonal outbreak (Table 5). The adjusted survival analysis showed adherence to the established antiviral treatment protocols to result in improved survival referred to both the global population (HR=0.57; 95%CI: 0.431–0.77; p<0.001) and the two periods considered separately (Figure 1).
Demographic and clinical characteristics of the survivors and deceased patients in the two study periods.
| Variables | Pandemic period (n=661) | Post-pandemic period (n=396) | ||||
|---|---|---|---|---|---|---|
| Survivors (n=517) | Deceased (n=144) | p-Value | Survivors (n=277) | Deceased (n=119) | p-Value | |
| Level of severity | ||||||
| SOFA | 4.9 (3) | 8.5 (4.3) | <0.001 | 5.2 (3.59) | 8.6 (4) | <0.001 |
| APACHE II | 12.5 (6.1) | 19.5 (8.4) | <0.001 | 14.1 (6.3) | 21 (8.4) | <0.001 |
| Demographic factors | ||||||
| Age, mean (SD) | 43.8 (14) | 48 (16.4) | 0.057 | 49.5 (14.4) | 51.9 (14.1) | 0.113 |
| Male sex | 287 (55.9) | 91 (63.3) | 0.209 | 184 (66.2) | 85 (71.4) | 0.590 |
| Comorbidities | ||||||
| >1 condition | 353 (69.6) | 154 (79.4) | 0.051 | 205 (73.7) | 94 (79) | 0.316 |
| COPD | 87 (16.8) | 21 (14.6) | 0.467 | 52 (21.3) | 26 (24.8) | 0.203 |
| Asthma | 69 (13.6) | 12 (8.5) | 0.829 | 27 (9.7) | 6 (5) | 0.4 |
| Heart failure | 31 (6.1) | 16 (11.3) | 0.034 | 26 (9.4) | 13 (11.8) | 0.189 |
| Chronic renal failure | 19 (3.7) | 14 (9.9) | 0.010 | 19 (6.8) | 13 (10.9) | 0.45 |
| Diabetes mellitus | 62 (12.2) | 20 (14.2) | 0.555 | 48 (17.3) | 16 (13.4) | 0.946 |
| Obesity | 180 (35.5) | 58 (41.1) | 0.713 | 107 (38.5) | 31 (26.1) | 0.017 |
| Autoimmune disease | 13 (2.5) | 8 (1.8) | 0.09 | 7 (2.9) | 7 (6.7) | 0.153 |
| Hematological disease | 19 (3.7) | 21 (14.9) | <0.001 | 15 (5.4) | 28 (23.5) | <0.001 |
| Neuromuscular disease | 18 (3.5) | 6 (4.2) | 0.783 | 2 (0.8) | 2 (1.9) | 0.632 |
| HIV infection | 9 (1.8) | 4 (2.8) | 0.165 | 2 (0.7) | 12 (10.1) | <0.001 |
| Pregnancy | 24 (4.6) | 7 (4.8) | 0.896 | 11 (4) | 2 (1.7) | 0.233 |
| Clinical presentation | ||||||
| Viral pneumonia | 359 (70.1) | 97 (67.4) | 0.178 | 186 (66.9) | 81 (68.1) | 0.844 |
| EPOC exacerbation | 31 (6.1) | 5 (3.5) | 0.427 | 22 (7.9) | 2 (1.7) | 0.217 |
| Respiratory bacterial coinfection | 70 (13.6) | 29 (20.9) | 0.056 | 45 (16.2) | 31.2 (6.1) | 0.119 |
| Complications | ||||||
| Septic shock | 190 (37.4) | 105 (74.5) | <0.001 | 127 (45.7) | 97 (91.5) | 0.001 |
| Multiorgan failure | 283 (55.5) | 125 (88) | <0.001 | 133 (47.89 | 100 (84) | 0.001 |
| Acute renal failure | 74 (15) | 38 (27.1) | <0.001 | 49 (17.6) | 64 (53.8) | <0.001 |
| Continuous renal replacement therapy | 74 (15) | 62 (45.9) | <0.001 | 9 (3.2) | 20 (16.8) | <0.001 |
| Noninvasive mechanical ventilation | 125 (24.2) | 38 (27) | 0.373 | 105 (38.5) | 37 (31.1) | 0.766 |
| Invasive mechanical ventilation | 266 (51.6) | 138 (95.8) | <0.001 | 156 (57.1) | 113 (95) | <0.001 |
| Renal failure | 74 (15) | 62 (45.9) | <0.001 | 51 (18.3) | 61 (51.2) | <0.001 |
| Prone decubitus | 52 (10.2) | 43 (30.7) | 0.002 | 33 (11.9) | 49 (41.2) | <0.001 |
| 49 | ||||||
| Treatment | ||||||
| Treatment with oseltamivir | 506 (99.6) | 137 (97.2) | <0.001 | 68 (96.8) | 115 (96.6) | 0.322 |
| Treatment with zanamivir | 2 (0.4) | 2 (1.4) | 0.475 | 16 (5.8) | 9 (7.6) | 0.973 |
| Empirical antiviral treatment | 371 (74.3) | 92 (67.6) | 0.207 | 196 (72.6) | 78 (67.2) | 0.07 |
| Days to start of antiviral treatment | 4.6 (2.7) | 5.3 (3.6) | 0.085 | 5.4 (3.1) | 6.5 (4.4) | 0.033 |
| Early antiviral treatment (≤48h) | 129 (25.6) | 24 (17.8) | 0.361 | 49 (18.1) | 12 (10.3) | 0.263 |
| Patients with severity criteria | 284 (54.9) | 131 (91) | <0.001 | 160 (57.6) | 112 (94.1) | <0.001 |
| Use of high doses | 221 (45.4) | 49 (40.5) | 0.113 | 88 (33) | 30 (26.1) | 0.001 |
| Use of high doses (150mg/12h) | 350 (70.9) | 103 (81.1) | 0.204 | 148 (55.2) | 64 (55.7) | 0.147 |
| More than 10 days of treatment | 276 (56.4) | 59 (48) | <0.001 | 138 (51.7) | 47 (40.9) | <0.001 |
| Duration of antiviral treatment | 9.79 (3.98 | 8.88 (5.17) | <0.001 | 9.34 (3.36) | 7.8 (4.5) | <0.001 |
| Total adherence to treatment | 218 (44.8) | 49 (40.5) | 0.002 | 113 (42.3) | 32 (27.8) | <0.001 |
| Vaccination | – | – | – | 16 (5.8) | 6 (5) | 0.883 |
COPD: chronic obstructive pulmonary disease; ICU: Intensive Care Unit; HIV: human immunodeficiency virus.
Variables showing statistical significance associated to mortality (Cox regression analysis) for both of the study periods.
| Variables | Pandemic period | Post-pandemic period | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | p-Value | HR | 95%CI | p-Value | |
| APACHE II score (per point) | 1.09 | 1.06–1.12 | 0.001 | 1.07 | 1.05–1.09 | 0.001 |
| Hematological disease | 2.20 | 1.26–3.85 | 0.005 | 2.27 | 1.39–3.70 | 0.001 |
| Adherence to treatment | 0.47 | 0.32–0.75 | 0.001 | 0.55 | 0.35–0.80 | 0.007 |
| Early treatment (<48h) | 0.56 | 0.32–0.97 | 0.03 | 0.83 | 0.35–1.83 | 0.07 |
| Positive HIV serological test | 2.73 | 0.96–7.72 | 0.058 | 3.20 | 1.63–6.30 | 0.001 |
The post hoc analysis identified only 23 patients (2.2%) who died before completing the treatment prescribed by the supervising physician; these subjects were therefore excluded. The final population for this analysis consisted of 1035 individuals. Twelve of the deceased patients (1.2%) corresponded to the pandemic period and 11 (1.0%) to the post-pandemic period. Global adherence was similar (42.6%) to that recorded for the total population, without statistically significant differences between the periods. In this population, adherence to the recommendations (HR=0.62; 95%CI: 0.43–0.96) and early antiviral treatment (HR=0.64; 95%CI: 0.43–0.95) were the only variables independently associated to lesser mortality in the multivariate analysis.
DiscussionThe main finding of the present study was the low global adherence to the treatment recommendations referred to influenza A during the period 2009–2011, and which proved more evident for the post-pandemic period. Adherence to the treatment recommendations was low even among those patients considered to be at high risk. In this particular subgroup of patients, the management protocol was only followed in one out of every two individuals during the pandemic period, and this situation was even worse in the post-pandemic period, when only four out of every 10 patients adhered to the mentioned recommendations. Another observation of great interest was that adherence to the treatment recommendations was associated to a 50% decrease in mortality on considering the global population, on distinguishing between the two periods, and on excluding the early mortality cases.
Our findings are consistent with those obtained by other studies on treatment compliance in other populations.11,12 Seale et al.19 published the degree of adherence to the antiviral drug treatment protocol developed by the Health Protection Agency (HPA) of the United Kingdom for the pediatric population in ICUs. The recommendations of the HPA were to start antiviral treatment early (within the first 48h after symptoms onset) in this particular subgroup of patients. The authors evaluated a total of 36 children, of which nine had been treated before publication of the guide and the rest after its implementation. The prescription rate for oseltamivir upon admission to the ICU increased significantly from 11% (1/9) to 50% (9/18) (OR=8). As can be seen, despite this improvement in compliance, in the end only one out of every two children were treated in line with the recommendations. On the other hand, Fietje et al.,20 on analyzing oseltamivir prescription practice in The Netherlands during the pandemic, recorded an increase of 100% with respect to 2008. The aim of the study was to determine whether the dispensation of oseltamivir complied with the prescription recommendations of the national guides for the pandemic. In addition, an analysis was made of the use of this drug by the patients, based on a questionnaire. A total of 630 patients were contacted, and 361 (57.3%) completed the questionnaire. Thirty-seven percent of the subjects (n=111) received oseltamivir in the absence of any criterion justifying such prescription. Regarding patient adherence, the results reflected strong compliance, since 97.4% of the patients started the prescribed treatment and 90.8% adhered to the established duration of therapy. On the other hand, Hersh et al.21 evaluated the degree of adherence to the guidelines of the United States Centers for Disease Control and Prevention (CDC) regarding the prescription of antiviral drugs during the influenza A(H1N1) pandemic of 2009 in the United States. In contrast to the abovementioned study, the frequency of prescription was found to be no different during the influenza A(H1N1)pdm09 period (58%) with respect to earlier years (59%). During the pandemic, antivirals were prescribed for only 47% of the patients under two years of age and for 68% of the patients over 65 years of age. These results indicate infrautilization of antiviral treatment in these risk groups with possible complications that can be prevented, and suggest opportunities for improving implementation of the public health guides in clinical practice.
Our results suggest that adherence to the treatment protocols is a variable independently associated to mortality. A decrease of close to 50% was observed both in general and on considering the different periods–in concordance with the existing literatures.22,23 Failure to comply with the treatment guides in severe community-acquired pneumonia is related to increased morbidity-mortality, as well as to increased hospital stay and healthcare costs.24–26 However, few studies have evaluated the impact of adherence to the antiviral treatment recommendations upon mortality in critical patients in ICUs during the pandemic of 2009 and in the post-pandemic period 2010–2011. These recommendations were based on the cumulative body of experience gained with seasonal influenza (1970–2008), where early treatment with neuraminidase inhibitors reduced the severity and duration of the disorder, as well as the risk of complications.27,28 However, in comparison with seasonal influenza (non-pandemic H1N1 or H3N2), influenza A(H1N1)pdm09 behaves differently–not only because it affects younger individuals, but also because of its capacity to produce rapid and severe lung damage, with the development of acute respiratory distress syndrome and death secondary to refractory respiratory failure. In this special context, it is difficult to extrapolate the impact of antiviral treatment from studies in which the included patients show only mild or moderate disease caused by some other type of virus A. Controversy therefore exists regarding the impact of antiviral treatment upon infection caused by influenza A(H1N1)pdm09, since there are no randomized and controlled studies in critical patients that have firmly demonstrated its usefulness. A review29 of 11 published studies showed (albeit with important methodological limitations) that the complications of influenza A(H1N1)pdm09 are reduced by the use of antivirals in patients at both low and high risk. On the other hand, Jain et al.30 found the only variable independently associated to mortality to be the administration of oseltamivir within the first 48h, while Dominguez-Cherit et al.31 reported greater survival (OR=7.4) in patients who received antivirals. Three retrospective studies were published in 2011,32–34 with results in this same line. However, and despite the fact that different publications of the Cochrane Library27,28,35 reveal no benefit with oseltamivir in terms of the prevention and/or treatment of seasonal influenza, the studies considered in those reviews did not include critical patients. On the other hand, these data are in contrast to those obtained by the Spanish multicenter GETGAG study,22 which evidenced a decrease in mortality among patients receiving early antiviral treatment. In this same sense, a recent international study on the effectiveness of antiviral treatment during the pandemic period, including over 29,000 patients, documented a decrease in mortality risk among the patients that received antiviral therapy.23
The present study has a number of limitations that must be commented, though its findings are relevant and of great interest. The main limitation is referred to the analysis of adherence, which only considers adhesion to the treatment recommendations in relation to the known risk factors. Although our results show a decrease in mortality risk when following the treatment recommendations for influenza A(H1N1)pdm09, the type of analysis performed does not allow us to establish a cause-effect relationship, since other uncontrolled variables (confounding factors) could influence the results. In this regard, it is possible that those patients who followed the treatment recommendations also received more adequate medical care, and hence the impact upon mortality may not have been determined only by administration of the antiviral drug. However, the objective of the study was to evaluate adherence to the recommendations of treatment in general, considering the known severity factors. Furthermore, after adjusting the model for risk by means of the multivariate analysis and excluding those patients with early mortality in a post hoc analysis, adherence to the treatment recommendations remained as a factor independently associated to mortality.
On the other hand, the body of data included in this study is representative of prescription practice and the use of antivirals in Spanish ICUs, but such data cannot be extrapolated to other ICUs or to other types of patients. Lastly, it must be mentioned that there may have been uncontrolled social, economical or healthcare factors that could have contributed to the lack of adherence to the treatment recommendations, as has been recognized in a recent editorial.36 However, because of the characteristics of the study, it was not possible to analyze these variables. Consequently, our findings must be interpreted with caution, and within the limitations of its design.
In conclusion, the results of the present study reveal a low level of adherence to antiviral treatment in critical patients and a close association between this situation and mortality. Adherence was found to be even lower in the post-pandemic period, in which the recorded mortality was higher than during the pandemic period. The introduction of educational and diffusion programs designed to increase adherence to the treatment recommendations should be considered before each winter season.
Conflicts of interestThe authors declare that they have no conflicts of interest.
This study was endorsed by the Spanish Society of Intensive and Critical Care Medicine and Coronary Units (Sociedad Española de Medicina Intensiva, Crítica y Unidades Coronarias, SEMICYUC). The SEMICYUC has not influenced the study design; data collection, analysis or interpretation; drafting of the manuscript; or the decision to submit the study for publication. The content of the manuscript is the responsibility of the authors, and does not necessarily reflect the official view of the SEMICYUC.
The authors thank the GTEIS/SEMICYUC for its support of this project.
Andalusia: Pedro Cobo (Hospital Punta de Europa, Algeciras); Javier Martins (Hospital Santa Ana Motril, Granada); Cecilia Carbayo (Hospital Torrecárdenas, Almería); Emilio Robles-Musso, Antonio Cárdenas, Javier Fierro (Hospital del Poniente, Almería); Ocaña Fernández (Hospital Huercal–Overa, Almería); Rafael Sierra (Hospital Puerta del Mar, Cádiz); M. Jesús Huertos (Hospital Puerto Real, Cádiz); Juan Carlos Pozo, R. Guerrero (Hospital Reina Sofía, Córdoba); Enrique Márquez (Hospital Infanta Elena, Huelva); Manuel Rodríguez-Carvajal (Hospital Juan Ramón Jiménez, Huelva); Antonio Jareño (Hospital del SAS de Jerez, Jerez de la Frontera); José Pomares, José Luis Ballesteros (Hospital Universitario San Cecilio, Granada); Yolanda Fernández, Francisco Lobato, José F. Prieto, José Albofedo-Sánchez (Hospital Costa del Sol, Marbella); Pilar Martínez (Hospital Virgen de la Victoria, Málaga); Miguel Ángel Díaz Castellanos (Hospital Santa Ana de Motril, Granada); Guillermo Sevilla (Clínica Sagrado Corazón, Sevilla); José Garnacho-Montero, Rafael Hinojosa, Esteban Fernández (Hospital Virgen del Rocío, Sevilla); Ana Loza, Cristóbal León (Hospital Universitario Nuestra Señora de Valme, Sevilla); Ángel Arenzana (Hospital Virgen de la Macarena, Sevilla), Dolores Ocaña (Hospital de la Inmaculada, Sevilla); Inés Navarrete (Hospital Virgen de las Nieves, Granada).
Aragón: Manuel Luis Avellanas, Arantxa Lander, S. Garrido Ramírez de Arellano, M.I. Marquina Lacueva (Hospital San Jorge, Huesca); Pilar Luque (Hospital Lozano Blesa, Zaragoza); Ignacio González (Hospital Miquel Servet, Zaragoza); Jose M. Montón (Hospital Obispo Polanco, Teruel); Jose M. Díaz, Pilar López-Reina, Sergio Sáez (Hospital Virgen de la Salud, Teruel).
Asturias: Lisardo Iglesias, Carmen Pascual González (Hospital Universitario Central de Asturias – HUCA, Oviedo); Quiroga (Hospital de Cabueñes, Gijón); Águeda García-Rodríguez (Hospital Valle del Nalón, Langreo).
Balearic Islands: Lorenzo Socias, Pedro Ibánez, Marcío Borges-Sa, A. Socias, del Castillo A (Hospital Son LLatzer, Palma de Mallorca); Ricard Jordà Marcos (Clínica Rotger, Palma de Mallorca); José M. Bonell (USP. Clínica Palmaplanas, Palma de Mallorca); Ignacio Amestarán (Hospital Son Dureta, Palma de Mallorca).
Canary Islands: Sergio Ruiz-Santana, Juan José Díaz (Hospital Dr Negrín, Las Palmas de Gran Canaria); Montserrat Sisón (Hospital Doctor José Molina, Lanzarote); David Hernández, Ana Trujillo, Luis Regalado (Hospital General la Palma, La Palma); Leonardo Lorente (Hospital Universitario de Canarias, Tenerife); Mar Martín (Hospital de la Candelaria, Tenerife), Sergio Martínez, J.J. Cáceres (Hospital Insular de Gran Canaria).
Cantabria: Borja Suberviola, P. Ugarte (Hospital Universitario Marqués de Valdecilla, Santander).
Castilla La Mancha: Fernando García-López (Hospital General, Albacete); Ángel Álvaro Alonso, Antonio Pasilla (Hospital General La Mancha Centro, Alcázar de San Juan); M. Luisa Gómez Grande (Hospital General de Ciudad Real, Ciudad Real); Antonio Albaya (Hospital Universitario de Guadalajara, Guadalajara); Alfonso Canabal, Luis Marina (Hospital Virgen de la Salud, Toledo).
Castilla y León: Juan B. López Messa (Complejo Asistencial de Palencia, Palencia), M. Jesús López Pueyo (Hospital General Yagüe, Burgos); Zulema Ferreras (Hospital Universitario de Salamanca, Salamanca); Santiago Macias (Hospital General de Segovia, Segovia); José Ángel Berezo, Jesús Blanco Varela (Hospital Universitario Río Hortega, Valladolid), A. Andaluz Ojeda (Hospital Universitario, Valladolid); Antonio Álvarez Terrero (Hospital Virgen de la Concha, Zamora); Fabiola Tena Ezpeleta (Hospital Santa Bárbara, Soria).
Catalonia: Rosa M. Catalán (Hospital General de Vic, Vic); Miquel Ferrer, Antoni Torres (Hospital Clínic, Barcelona); Sandra Barbadillo (Hospital General de Catalunya – CAPIO, Barcelona); Lluís Cabré (Hospital de Barcelona, Barcelona); Assumpta Rovira (Hospital General de l’Hospitalet, L’Hospitalet); Francisco Álvarez-Lerma, Antonia Vázquez, Joan Nolla (Hospital Del Mar, Barcelona); Francisco Fernández, Joaquim Ramón Cervelló (Centro Médico Delfos, Barcelona); Rafael Mañéz, J. Ballús, Rosa M. Granada (Hospital de Bellvitge, Barcelona); Jordi Vallés, Marta Ortíz, C. Guía (Hospital de Sabadell, Sabadell); Fernando Arméstar, Joaquim Páez (Hospital Dos De Mayo, Barcelona); Jordi Almirall, Xavier Balanzo (Hospital de Mataró, Mataró); Jordi Rello, Elena Arnau, Lluis Llopart, Mercedes Palomar (Hospital Vall d’Hebron, Barcelona); Iñaki Catalán (Hospital Sant Joan de Déu, Manresa); Josep M. Sirvent, Cristina Ferri, Nerea López de Arbina (Hospital Josep Trueta, Girona); Mariona Badía, Montserrat Valverdú-Vidal, Fernando Barcenilla (Hospital Arnau de Vilanova, Lleida); Mònica Magret (Hospital Sant Joan de Reus, Reus); M.F. Esteban, José Luna (Hospital Verge de la Cinta, Tortosa); Juan M. Nava, J González de Molina (Hospital Universitario Mutua de Terrassa, Terrassa); Zoran Josic (Hospital de Igualada, Igualada); Francisco Gurri (Hospital Quirón, Barcelona); Alejandro Rodríguez, Thiago Lisboa, Diego de Mendoza, Sandra Trefler (Hospital Universitario Joan XXIII, Tarragona), Rosa María Díaz (Hospital San Camil, Sant Pere de Ribes, Barcelona).
Extremadura: Juliá-Narváez José (Hospital Infanta Cristina, Badajoz); Alberto Fernández-Zapata, Teresa Recio, Abilio Arrascaeta, M. José García-Ramos, Elena Gallego (Hospital San Pedro de Alcántara, Cáceres); F. Bueno (Hospital Virgen del Puerto, Plasencia).
Galicia: M. Lourdes Cordero, José A. Pastor, Luis Álvarez-Rocha (CHUAC, A Coruña); Dolores Vila (Hospital Do Meixoeiro, Vigo); Ana Díaz Lamas (Hospital Arquitecto Marcide, Ferrol); Javier Blanco Pérez, M. Ortiz Piquer (Hospital Xeral-Calde, Lugo); Eleuterio Merayo, Victor Jose López-Ciudad, Juan Cortez, Eva Vilaboy (Complejo Hospitalario de Ourense, Ourense); Eva Maria Saborido (Hospital Montecelo, Pontevedra); Raul José González (H. Miguel Domínguez, Pontevedra); Santiago Freita (Complejo Hospitalario de Pontevedra, Pontevedra).
La Rioja: José Luis Monzón, Félix Goñi (Hospital San Pedro, Logroño).
Madrid: Frutos del Nogal Sáez, M. Blasco Navalpotro (Hospital Severo Ochoa, Madrid); M. Carmen García-Torrejón (Hospital Infanta Elena, Madrid);César Pérez-Calvo, Diego López (Fundación Jiménez Díaz, Madrid); Luis Arnaiz, S. Sánchez-Alonso, Carlos Velayos (Hospital Fuenlabrada, Madrid); Francisco del Río, Miguel Ángel González (Hospital Clínico San Carlos, Madrid); María Cruz Martín, José M. Molina (Hospital Nuestra Señora de América, Madrid); Juan Carlos Montejo, Mercedes Catalán (Hospital Universitario 12 de Octubre, Madrid); Patricia Albert, Ana de Pablo (Hospital del Sureste, Arganda del rey);José Eugenio Guerrero, Jaime Benitez Peyrat (Hospital Gregorio Marañón, Madrid); Enrique Cerdá, Manuel Alvarez, Carlos Pey (Hospital Infanta Cristina, Madrid);Montse Rodríguez, Eduardo Palencia (Hospital Infanta Leonor, Madrid); Rafael Caballero (Hospital de San Rafael, Madrid); Rafael Guerrero (Hospital Reina Sofía, Madrid); Concepción Vaquero, Francisco Mariscal, S. García (Hospital Infanta Sofía, Madrid); Almudena Simón (Hospital Nuestra Señora del Prado, Madrid); Nieves Carrasco (Hospital Universitario La Princesa, Madrid); Isidro Prieto, A Liétor, R. Ramos (Hospital Ramón y Cajal, Madrid); Beatriz Galván, Juan C. Figueira, M. Cruz Soriano (Hospital La Paz, Madrid); P Galdós, Bárbara Balandin Moreno (Hospital Puerta de Hierro, Madrid); Fernández del Cabo (Hospital Monte Príncipe, Madrid); Cecilia Hermosa, Federico Gordo (Hospital de Henares, Madrid); Alejandro Algora (Hospital Universitario Fundación Alcorcón, Madrid); Amparo Paredes (Hospital Sur de Alcorcón, Madrid); J.A. Cambronero (Hospital Universitario Príncipe de Asturias, Madrid); Sonia Gómez-Rosado (Hospital de Móstoles, Madrid).
Murcia: Sofía Martínez (Hospital Santa María del Rosell, Murcia); F. Felices Abad (Hospital Universitario Reina Sofía, Murcia); Mariano Martínez (Hospital Universitario Virgen de la Arrixaca, Murcia); Sergio Manuel Butí, Gil Rueda, Francisco García (Hospital Morales Messeguer, Murcia).
Navarre: Laura Macaya, Enrique Maraví-Poma, I. Jimenez Urra, L. Macaya Redin, A Tellería (Hospital Virgen del Camino, Pamplona); Josu Insansti (Hospital de Navarra, Pamplona).
Basque Country: Nagore González, Pilar Marco, Loreto Vidaur (Hospital de Donostia, San Sebastián); B. Santamaría (Hospital de Basurto, Bilbao); Juan Carlos Vergara, Jose Ramon Iruretagoyena Amiano (Hospital de Cruces, Bilbao); Alberto Manzano (Hospital Santiago Apóstol, Vitoria);Carlos Castillo Arenal (Hospital Txagorritxu, Vitoria).
Valencia: José Blanquer (Hospital Clinic Universitari, Valencia); Roberto Reig Valero, A. Belenger, Susana Altaba (Hospital General de Castellón, Castellón); Bernabé Álvarez-Sánchez (Hospital General de Alicante, Alicante); Santiago Alberto Picos (Hospital Torrevieja Salud, Alicante); Ángel Sánchez-Miralles (Hospital San Juan, Alicante); Juan Bonastre, M. Palamo, Javier Cebrian, José Cuñat (Hospital La Fe, Valencia); Belén Romero (Hospital de Manises, Valencia); Rafael Zaragoza (Hospital Dr. Peset, Valencia); Virgilio Paricio (Hospital de Requena, Valencia); Asunción Marques, S. Sánchez-Morcillo, S. Tormo (Hospital de la Ribera, Valencia); J. Latour (H. G. Universitario de Elche, Valencia), M. Ángel García (Hospital de Sagunto, Castellón).
Please cite this article as: Canadell L, Martín-Loeches I, Díaz E, Trefler S, Grau S, Yebenes JC, et al. Grado de adherencia al tratamiento antivírico recomendado durante la pandemia y periodo pospandémico de gripe A (H1N1)pdm09 en 148 unidades de cuidados intensivos españolas. Med Intensiva. 2015;39:222–233.