To assess the diagnostic performance of high-sensitivity troponin T (hs-TnT) in patients with suspected non-ST elevation acute coronary syndrome (NSTE-ACS); confirm whether it shortens the time to diagnosis; and analyze the clinical consequences derived from its use.
MethodA prospective, longitudinal observational study was carried out in 5 emergency care departments. Patients seen for chest pain with suspected of NSTE-ACS were consecutively included. Patient care followed the internal protocols of the center, based on the consensus guidelines of the European Society of Cardiology. Serial conventional cardiac troponin (cTn) and hs-TnT determinations were made.
ResultsA total of 351 patients were included in the study. A final diagnosis of acute myocardial infarction (AMI) was established in 77 patients, with unstable angina in 102, and no acute coronary syndrome in 172 patients. The hs-TnT values were above percentile 99% in a large number of patients without AMI. In the initial determination, the diagnostic sensitivity of the hs-TnT was significantly greater than that of cTn (87.0% vs. 42.9%), which led to a negative predictive value of 95.1%.
ConclusionsHigh-sensitivity troponin T improves diagnostic performance compared with conventional troponin assay, shortens the time to diagnosis, and identifies a larger number of patients with smaller myocardial infarctions.
Los objetivos del estudio son evaluar el rendimiento diagnóstico de la troponina cardiaca T de alta sensibilidad (TnTc-hs) en pacientes con sospecha de síndrome coronario agudo sin elevación del segmento ST (SCASEST), confirmar si permite acortar el tiempo hasta el diagnóstico y analizar las consecuencias clínicas derivadas de su utilización.
MétodoSe trata de un estudio observacional, longitudinal y prospectivo, realizado en 5 servicios de urgencias hospitalarias. Se incluyó de forma consecutiva a los pacientes que acudían por dolor torácico sospechoso de SCASEST. El manejo del paciente y el tratamiento aplicado siguieron los protocolos internos basados en las guías de consenso de la Sociedad Europea de Cardiología. Se realizaron determinaciones seriadas de Tnc convencional (4ªG) y de TnTc-hs.
ResultadosSe incluyó en el estudio a 351 pacientes. El diagnóstico final de infarto agudo de miocardio (IAM) se estableció en 77 pacientes del total, angina inestable en 102 y 172 fueron pacientes diagnosticados como sin síndrome coronario agudo. Los valores de TnTc-hs estaban por encima del p99 en un alto número de pacientes sin IAM. En la determinación inicial del paciente, la sensibilidad diagnóstica de la TnTc-hs fue significativamente superior a la de la TnTc 4ªG (87,0 vs. 42,9%), lo que comportó un valor predictivo negativo del 95,1%.
ConclusionesLa TnTc-hs mejora el rendimiento diagnóstico al compararla con el ensayo de Tnc convencional, acorta el tiempo hasta el diagnóstico y reconoce mayor número de pacientes con IAM más pequeños.
Adequate interpretation of myocardial necrosis markers is essential for the diagnosis, stratification and treatment of patients with suspected acute coronary syndrome (ACS). The protocols on cardiac markers of acute myocardial infarction (AMI) are referred to the third universal definition of AMI.
This document establishes that cardiac troponin (cTn) elevation and lowering is fundamental for establishing the diagnosis in the context of ischemic symptoms, electrocardiographic changes or newly manifesting segmental alterations in an imaging study.1
Technological advances in the determination of cTn have improved the capacity to detect and quantify myocardial cell damage. The criteria referred to elevation of the cTn values depend on the test used and must be defined on the basis of the precision profile of each test, including that of high sensitivity testing.2 The existence of different commercial cTn immunoassays with different percentile 99 (p99) concentrations; the fact that some laboratories use different references to the myocardial damage test results (“normal”, “indeterminate” or “suggestive”); and the indiscriminate use of cTn testing in large populations with very different probabilities of presenting ACS, tend to generate confusion in the context of clinical management.
There are a significant number of patients who in the absence of AMI nevertheless exhibit cTn concentrations>p99. These patients usually have cardiovascular risk factors, and the likeliness of complications in such individuals is similar to that in patients with ACS.3
The latest European recommendations on the management of patients with non-ST elevation acute coronary syndrome (NSTE-ACS) advocate the use of high sensitivity cTn (hs-cTn) versus conventional or fourth generation (4G) troponins, and contemplate up to two algorithms (0h/3h, 0h/1h) for the confirmation (rule in) or exclusion (rule out) of AMI.4
Cardiac troponins are extremely specific of myocardial damage. However, in no way is myocardial damage exclusive of ACS. Consequently, the clinical guides underscore that cTn concentration must be interpreted in relation to the patient symptoms. In this respect, elevated cTn concentrations in patients with sepsis, hypertensive crises, pulmonary embolism, etc., are indicative of myocardial damage, but not of AMI, and should not be used to indicate specific treatments for ACS.5
The term “high sensitivity” should be reserved for those immunoassays capable of determining p99 with a coefficient of variation (CV) of <10%, and able to measure the cTn concentration in at least 50% of the healthy reference population. These are the two features that differentiate them from the previous tests.6
There are doubts in the scientific community as to whether the use of hs-cTn represents significant clinical improvement, and whether it implies more tests and interconsultations.7 The present study was carried out to evaluate the diagnostic performance of high sensitivity troponin T (hs-TnT), determine whether it shortens the time to diagnosis, and analyze the clinical consequences of its use.
MethodA prospective, longitudinal observational study was carried out in the hospital emergency services (HES) of Hospital Clínico (Valencia), Hospital Clínico (Málaga), Hospital Severo Ochoa (Leganés), Hospital Sant Pau (Barcelona) and Hospital Miguel Servet (Zaragoza)(Spain), involving the patients of the TUSCA (Troponina Ultrasensible en el SCA) trial8 between June 2009 and April 2010. The study was carried out in abidance with the principles of the Declaration of Helsinki, and was approved by the local Ethics Committees of the participating centers. Written informed consent was obtained from all patients. We consecutively included patients over 18 years of age seen in the HES due to chest pain suggestive of NSTE-ACS. Patient enrollment was made during initial care in the HES, after assessment of the inclusion and exclusion criteria. Patient management and the applied treatment followed internal protocols based on the consensus guides of the European Society of Cardiology, under the indications of the physicians in charge of the patients at all times, and with total independence of participation in the study. Serial 4G cTn determinations were made for the diagnosis of ACS, in accordance with routine clinical and immunoassay practice in each center.
The patient characteristics were entered in a case report form and comprised demographic data, time referred to symptoms onset, the presence of risk factors (arterial hypertension, diabetes mellitus, chronic obstructive pulmonary disease [COPD], heart failure and renal failure). We also recorded the ECG characteristics with and without pain, as well as information referred to management after initial emergency care (catheterization, angioplasties, bypass surgery, etc.), the final diagnosis, and patient destination.
Patients with ST-segment elevation ACS were excluded, as were those with newly manifesting left bundle block, defibrillation or cardioversion before the first laboratory determinations, heart surgery in the three weeks before admission, pregnant women, patients with severe cognitive impairment, and subjects with chronic renal failure subjected to hemodialysis.
During the first 12h of stay in the HES, Intensive Care Unit (ICU) or hospital ward, serial blood samples were obtained for the measurement of hs-TnT, based on the following protocol:
- -
Sample T0. At the time of first assistance in the HES.
- -
Sample T1. Two hours after the sampling timepoint 0 or immediately before admission to some hospital unit (ICU or ward).
- -
Sample T2. Between 4 and 6h after sampling timepoint 0 or immediately before admission to some hospital unit.
- -
Sample T3. Between 8 and 12h after sampling timepoint 0 if the patient was still in the service, or immediately before admission to some hospital unit, or at the time of patient discharge from the HES.
Sufficient blood was collected to obtain 5 aliquots of 0.5ml of heparinized plasma from each of the samples of the patient, in addition to the sample required for routine testing. The aliquots were frozen at −80°C, and were identified with the code of each center: three digits corresponding to the patient number and the collection timepoint (T0, T1, T2 or T3), according to the sample involved. An average of 3.7 samples per patient were obtained. All the samples were transferred to the core laboratory (Hospital Sant Pau, Barcelona) for the analysis of 4G cTn and hs-TnT, with no more than one month being allowed to elapse from each collection. The hospitals all performed the routine 4G cTn test being used at the time (2 centers with cTnT and 3 with cTnI). The final patient diagnosis was established by an expert, based on all the clinical information provided by the case report form completed by the principal investigator in each center, reflecting the clinical and ECG data, and the centralized 4G cTn determination made in the core laboratory. The expert was blinded to the hs-cTn determinations.
Infarction was diagnosed in the presence of an elevation or reduction in cTnT concentration of >20% (Δ criterion) among several samples, with at least one value being greater than p99 for the reference population (≥ 0.01μg/l). The diagnosis of unstable angina (UA) was established in patients with clinical evidence of myocardial ischemia without positive 4G cTn. The patients with UA presented a history of resting angina, worsening of previous chronic angina, ST-segment changes suggestive of ischemia without biomarker variations (kinetics), positive ischemia detection tests, or stenosis>70% of a coronary artery as evidenced by coronary angiography. The cTnT levels in turn were measured using a high sensitivity technique on a Cobas e601 analyzer (hs-TnT, Roche Diagnostics, Basel, Switzerland). The technique has a limit of detection of 5.0ng/l, and the reference p99, used as the clinical decision limit, was 13.0ng/l. The analytical imprecision of these cTn tests was evaluated in 20 repetitions of 8 groups of samples with end concentrations adjusted to cover the range between 7 and 928ng/l, diluting a high cTnT concentration sample with a serum pool presenting undetectable hs-TnT levels (< 3ng/l). The p99 of the cTnT test (10ng/l=0.01μg/l) was measured with a coefficient of variation (CV) of 15.2%. The value with CV 10% was 35ng/l=0.035μg/l. The p99 of the hs-TnT test (13ng/l) was measured with a CV of 6.9%.
Frequencies and percentages were calculated for the descriptive analysis of the qualitative variables, with central tendency measures in the case of the quantitative variables. The data were expressed as the mean and standard deviation (SD) in the presence of a normal distribution, and as the median and interquartile range (IQR) in the case of a non-normal distribution as evidenced by the Kolmogorov–Smirnov test.
The comparison of quantitative variables was based on the Student's t-test or Mann–Whitney U-test in the presence or absence of a normal data distribution, respectively. The comparison of qualitative variables in turn was based on the chi-squared test or the Fisher exact test, as applicable.
In all cases the level of statistical significance was defined as 5% (α=0.05%) in two-tailed testing.
The 4G cTn and hs-TnT discrimination study was carried out calculating the area (AUC) under the receiver operating characteristic (ROC) curve at the different sampling timepoints, with the corresponding 95% confidence interval (CI). The significance of these curves was evaluated from calculation of the CI of the AUC, defining as significant those values in area with a lower limit of over 0.5%. The comparison of areas under the ROC curve was made by studying the overlap of CI values.
Diagnostic performance was assessed by calculating sensitivity, specificity and the positive and negative predictive values. The 95% confidence interval was calculated for clinical assessment of the magnitude of these measures – the interval being regarded as significant in the absence of overlap of the values.
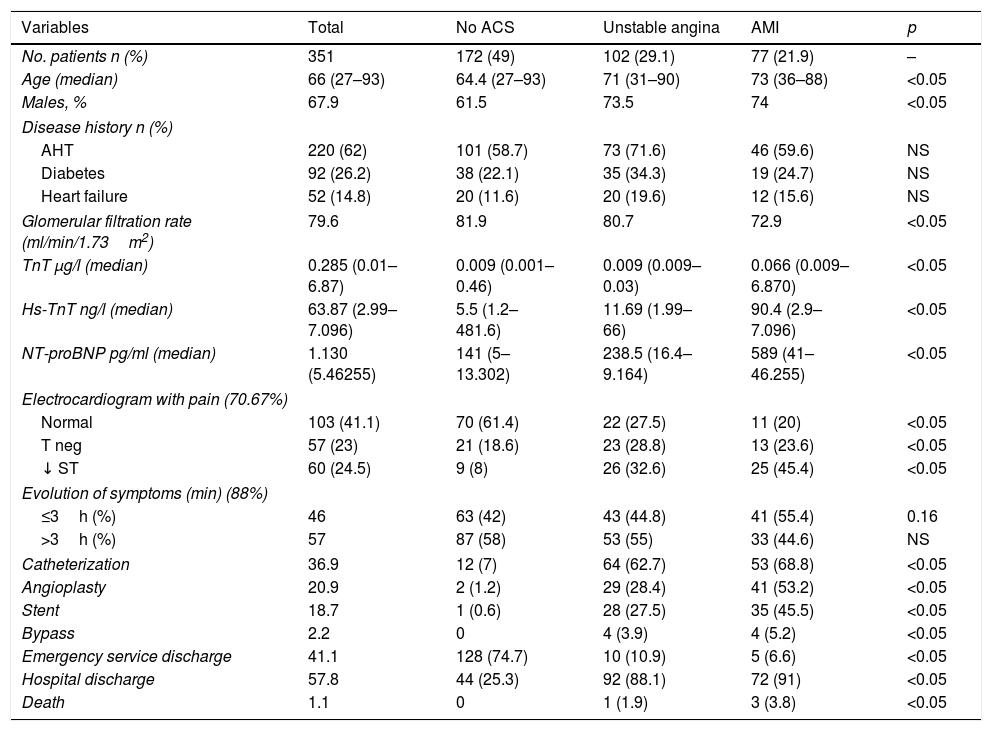
ResultsThe study comprised a total of 351 patients; 234 were males (67.9%), and the mean age was 65.7 years. The main demographic data and cardiovascular risk factors are summarized in Table 1. A final diagnosis of AMI was established in 77 patients, with UA in 102 patients. A total of 172 patients were diagnosed as having no ACS.
Characteristics and cardiovascular risk factors of the study sample.
| Variables | Total | No ACS | Unstable angina | AMI | p |
|---|---|---|---|---|---|
| No. patients n (%) | 351 | 172 (49) | 102 (29.1) | 77 (21.9) | – |
| Age (median) | 66 (27–93) | 64.4 (27–93) | 71 (31–90) | 73 (36–88) | <0.05 |
| Males, % | 67.9 | 61.5 | 73.5 | 74 | <0.05 |
| Disease history n (%) | |||||
| AHT | 220 (62) | 101 (58.7) | 73 (71.6) | 46 (59.6) | NS |
| Diabetes | 92 (26.2) | 38 (22.1) | 35 (34.3) | 19 (24.7) | NS |
| Heart failure | 52 (14.8) | 20 (11.6) | 20 (19.6) | 12 (15.6) | NS |
| Glomerular filtration rate (ml/min/1.73m2) | 79.6 | 81.9 | 80.7 | 72.9 | <0.05 |
| TnT μg/l (median) | 0.285 (0.01–6.87) | 0.009 (0.001–0.46) | 0.009 (0.009–0.03) | 0.066 (0.009–6.870) | <0.05 |
| Hs-TnT ng/l (median) | 63.87 (2.99–7.096) | 5.5 (1.2–481.6) | 11.69 (1.99–66) | 90.4 (2.9–7.096) | <0.05 |
| NT-proBNP pg/ml (median) | 1.130 (5.46255) | 141 (5–13.302) | 238.5 (16.4–9.164) | 589 (41–46.255) | <0.05 |
| Electrocardiogram with pain (70.67%) | |||||
| Normal | 103 (41.1) | 70 (61.4) | 22 (27.5) | 11 (20) | <0.05 |
| T neg | 57 (23) | 21 (18.6) | 23 (28.8) | 13 (23.6) | <0.05 |
| ↓ ST | 60 (24.5) | 9 (8) | 26 (32.6) | 25 (45.4) | <0.05 |
| Evolution of symptoms (min) (88%) | |||||
| ≤3h (%) | 46 | 63 (42) | 43 (44.8) | 41 (55.4) | 0.16 |
| >3h (%) | 57 | 87 (58) | 53 (55) | 33 (44.6) | NS |
| Catheterization | 36.9 | 12 (7) | 64 (62.7) | 53 (68.8) | <0.05 |
| Angioplasty | 20.9 | 2 (1.2) | 29 (28.4) | 41 (53.2) | <0.05 |
| Stent | 18.7 | 1 (0.6) | 28 (27.5) | 35 (45.5) | <0.05 |
| Bypass | 2.2 | 0 | 4 (3.9) | 4 (5.2) | <0.05 |
| Emergency service discharge | 41.1 | 128 (74.7) | 10 (10.9) | 5 (6.6) | <0.05 |
| Hospital discharge | 57.8 | 44 (25.3) | 92 (88.1) | 72 (91) | <0.05 |
| Death | 1.1 | 0 | 1 (1.9) | 3 (3.8) | <0.05 |
AHT: arterial hypertension; AMI: acute myocardial infarction; NS: nonsignificant; NT-proBNP: brain natriuretic peptide; ACS: acute coronary syndrome; TnT: troponin T; hs-TnT: high sensitivity troponin T.
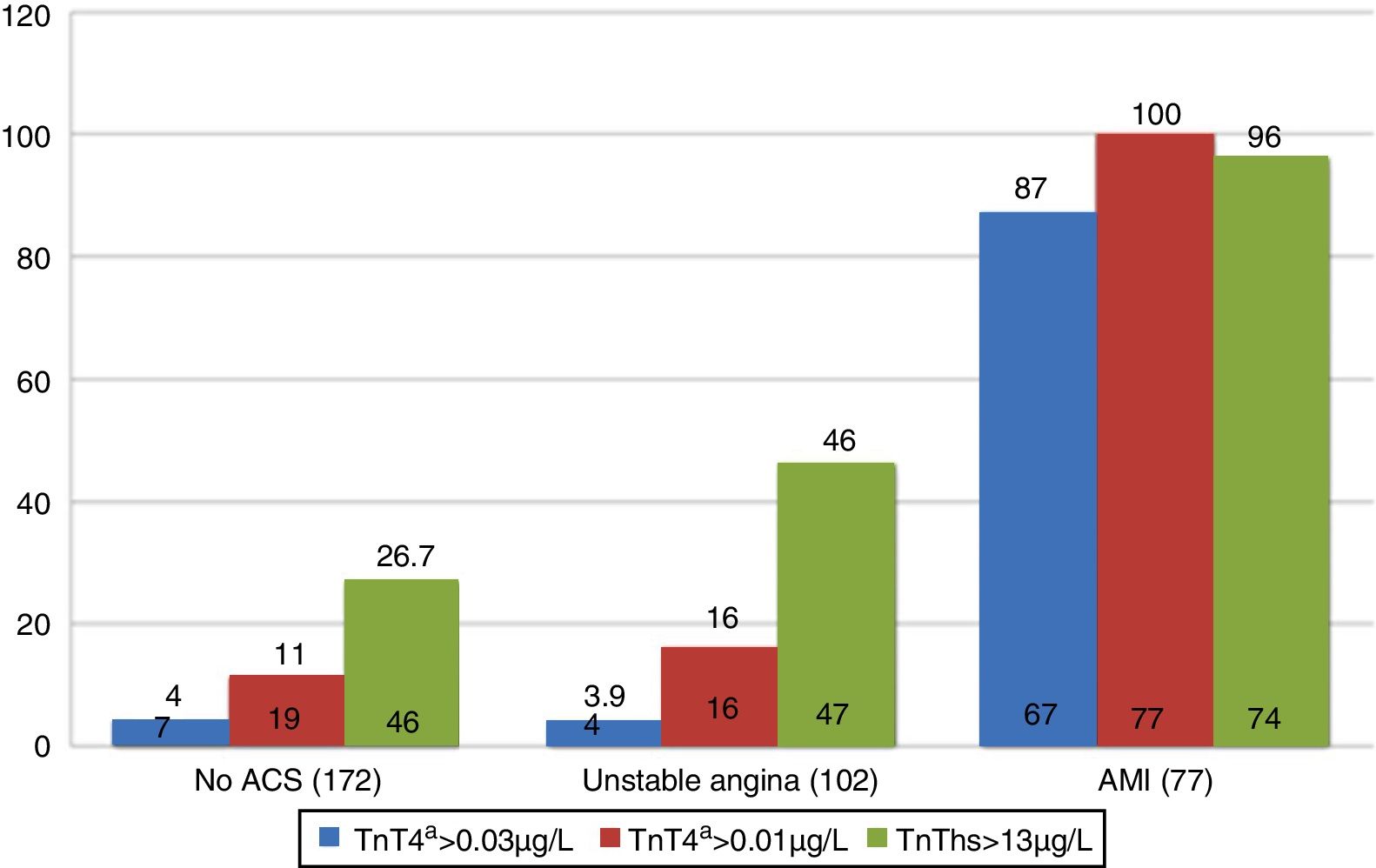
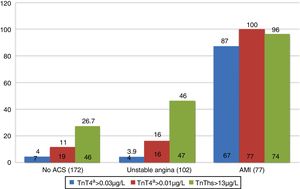
A total of 55.4% of the patients with AMI reached the HES within the first three hours, while a greater percentage of both those with UA and those without ACS arrived after three hours. With regard to positivity of the markers according to the final diagnosis, 87% of the patients with a final diagnosis of AMI presented 4G cTn>0.035μg/l during the first 12h of the study. In the case of non-ACS patients and those with UA, positivity was recorded in 4% of the cases. The hs-TnT concentration was >13ng/l (p99) in 26.7% and 46% of the patients diagnosed with non-ACS and UA, respectively (Fig. 1). On the other hand, 4G cTn using p99 with a cut-off point of 0.01μg/l (the gold standard of the study) proved positive in 16% of the patients diagnosed with UA and in 11% of those without ACS.
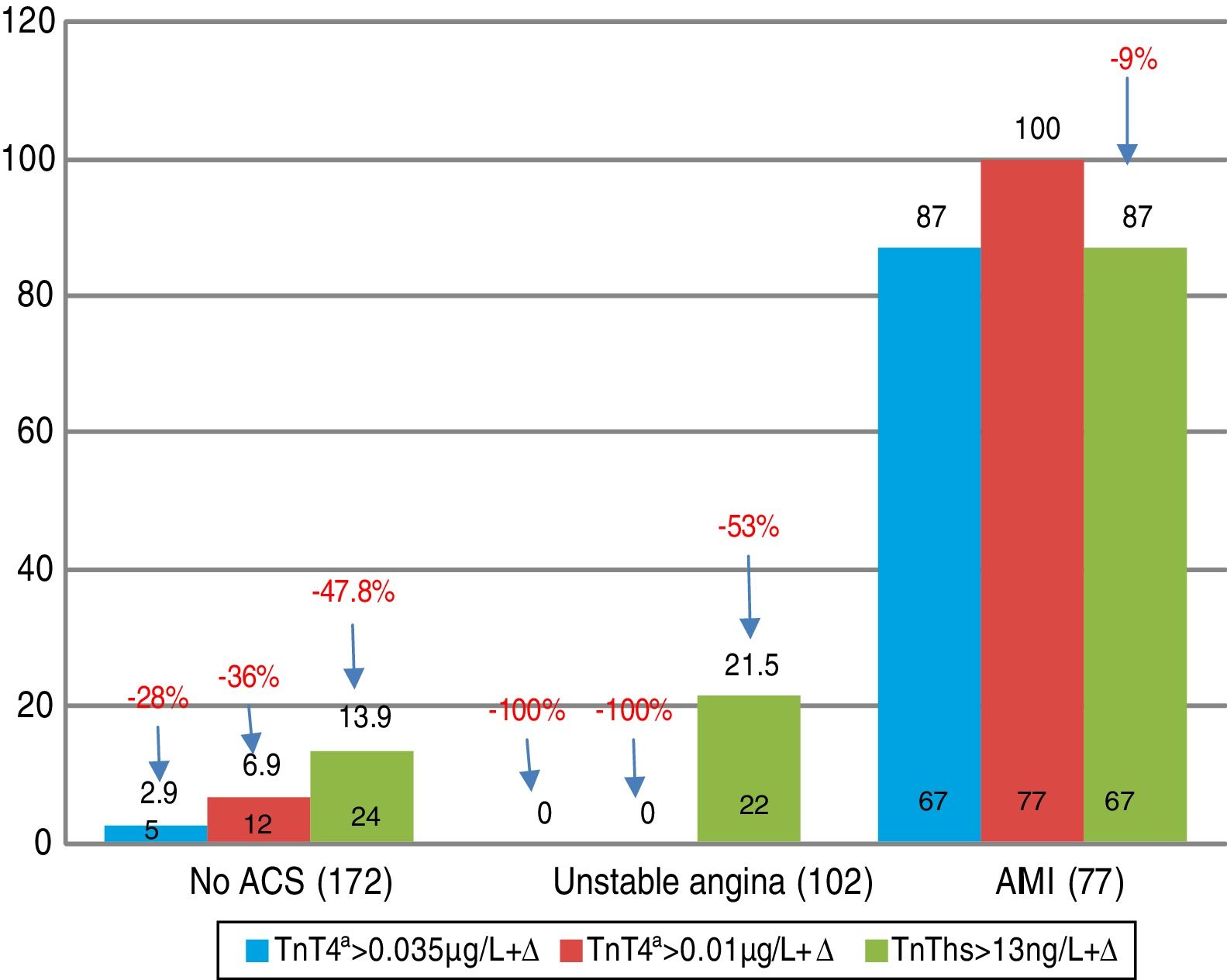
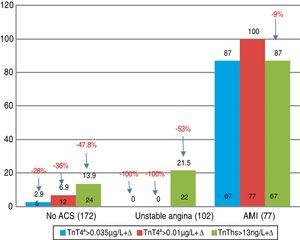
On applying kinetics among some of the four determinations (Δ criterion), 21.5% of the patients with UA showed elevation of hs-TnT+Δ (Fig. 2).
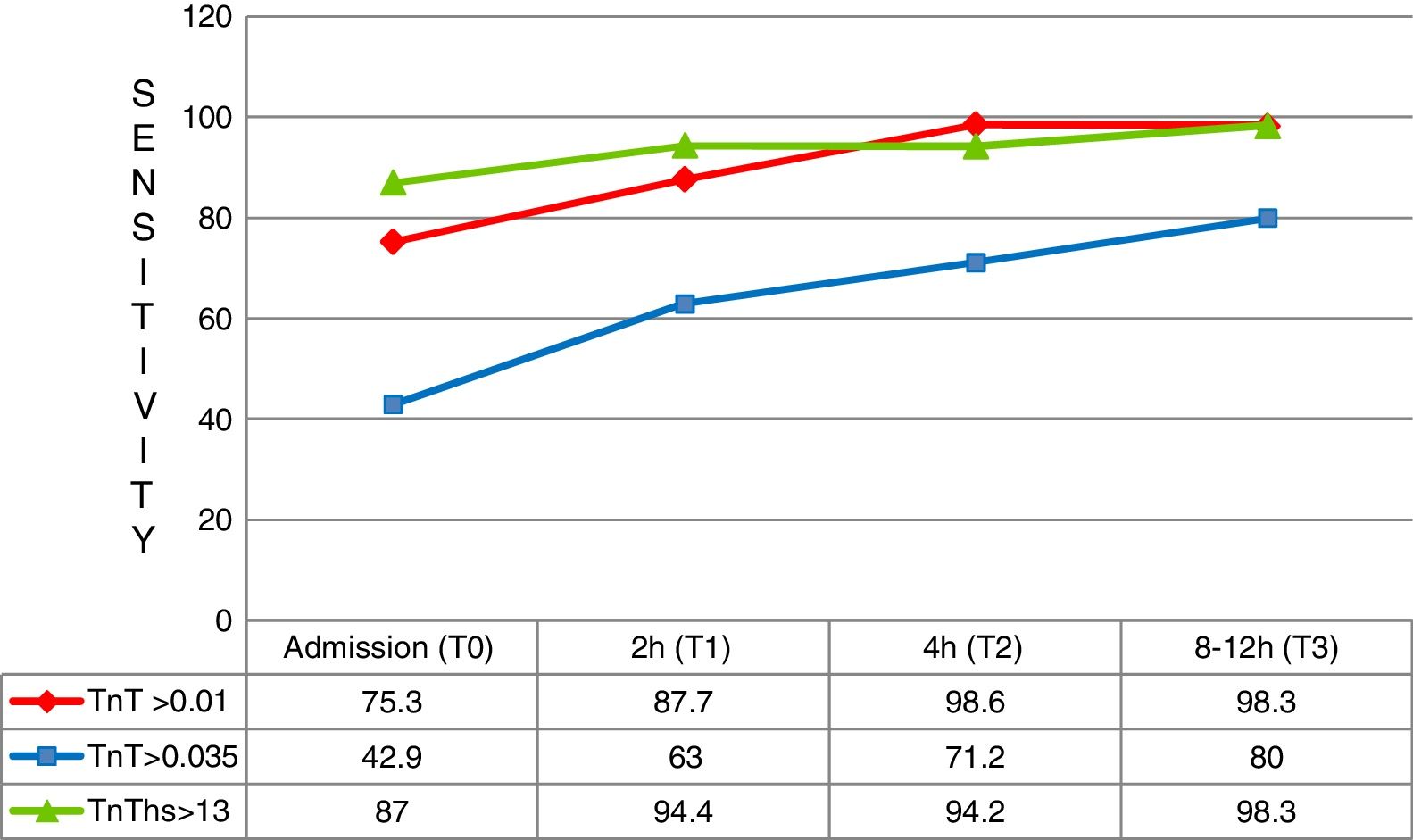
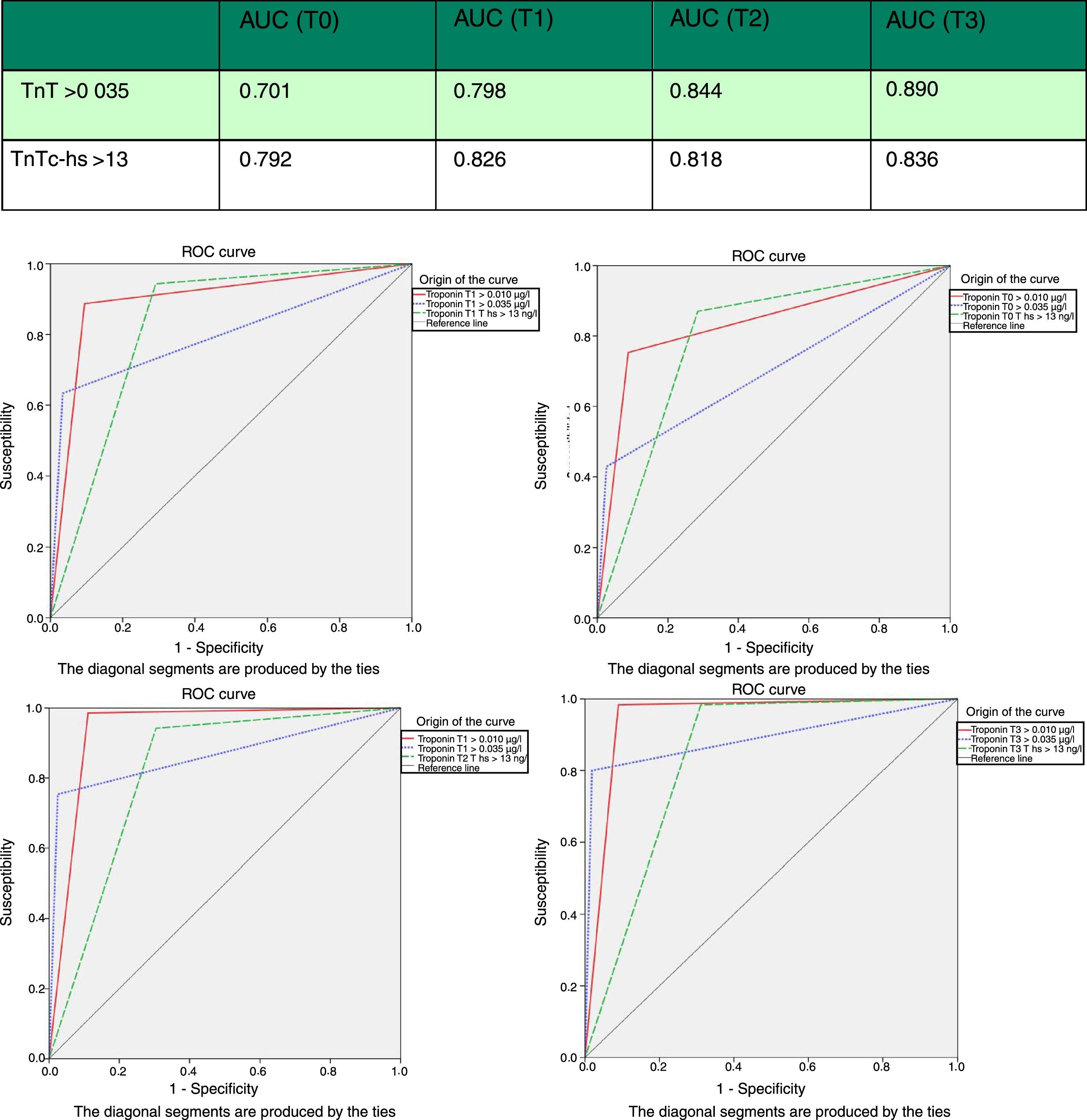
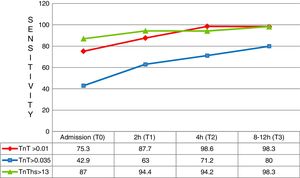
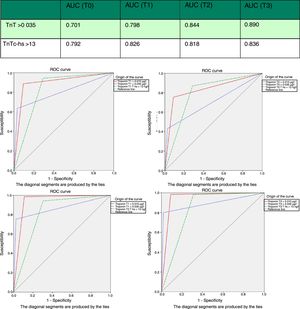
At first measurement (T0), the diagnostic sensitivity of hs-TnT was significantly greater than that of 4G cTn (CV) (87.0%; 83.0–90.3% vs. 42.9%; 37.6–48.2%; p<0.05), resulting in a negative predictive value of 95.1%. However, its specificity (71.5%; 66.5–76.2% vs. 97.4%; 95.2–98.8%; p<0.05) and positive predictive value (46.2% vs. 82.5; p<0.05) were clearly lower than those of 4G cTn (CV). On analyzing the area under the ROC curve, hs-TnT was seen to be superior to 4G cTn (CV) (0.792; 0.746–0.833 vs. 0.701; 0.650–0.748) (Figs. 3 and 4).
Two hours after patient arrival in the HES (T1), hs-TnT continued to show far higher diagnostic sensitivity than 4G cTn (94.4%; 91.3–96.6 vs. 63.0%; 57.6–68.2; p<0.05). The negative predictive value of hs-TnT was practically absolute (97.9%), and its diagnostic accuracy tended to be superior to that of 4G cTn (ROC 0.826 vs. 0.798). The low specificity of hs-TnT resulted in a low positive predictive value (46.5%). The rest of the sampling results proved similar, though reflecting lesser diagnostic performance.
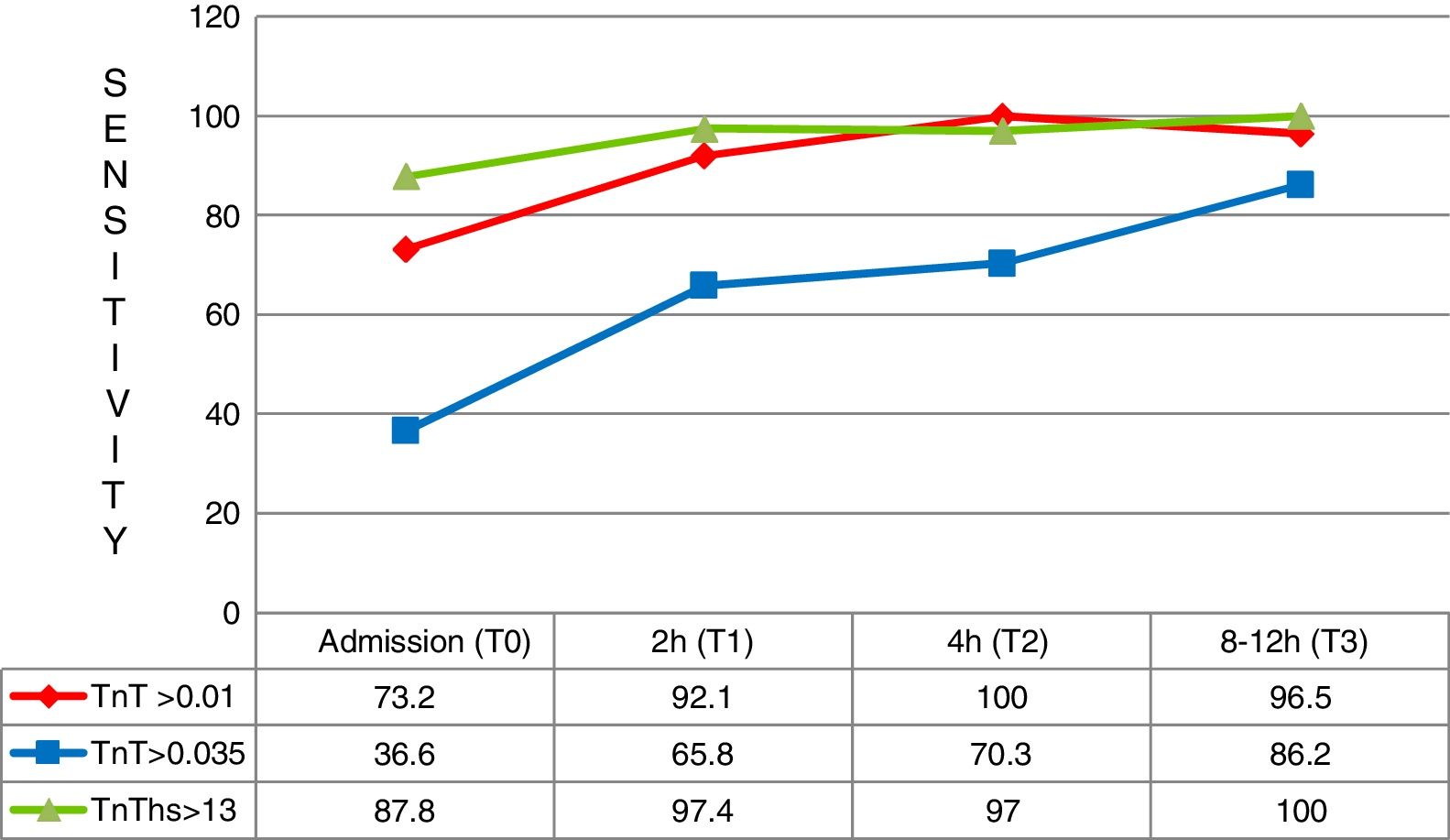
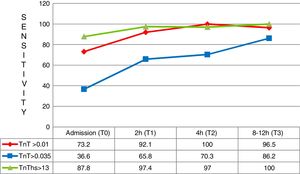
In the first three hours after symptoms onset, a total of 41 patients arrived with a final diagnosis of AMI. In these cases the superior diagnostic sensitivity of hs-TnT with respect to 4G cTn (CV) and also 4G cTn (p99) was particularly significant, with a sensitivity of 87.8% versus 36.6% and 73.2%, respectively (Fig. 5).
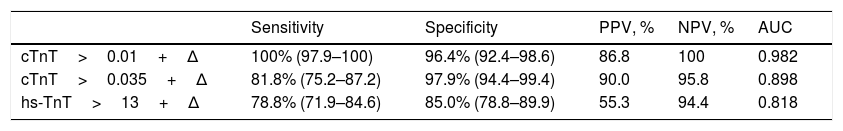
The patients diagnosed with non-ST elevation myocardial infarction (n=36) and presenting long evolutive times (> 3h) showed lesser diagnostic accuracy with hs-TnT (due to the lower specificity) than the patients with a shorter time from symptoms onset. On applying kinetics, gaining in specificity was again observed, with an increase in AUC. Accordingly, in the case of hs-TnT, we started with a specificity of 69% and, on applying Δ, the specificity reached 85%, with a positive predictive value of 55.3%, that improved the overall diagnostic performance (Table 2).
Kinetics (Δ) of the different cardiac troponins in patients with symptoms onset >180min.
| Sensitivity | Specificity | PPV, % | NPV, % | AUC | |
|---|---|---|---|---|---|
| cTnT>0.01+Δ | 100% (97.9–100) | 96.4% (92.4–98.6) | 86.8 | 100 | 0.982 |
| cTnT>0.035+Δ | 81.8% (75.2–87.2) | 97.9% (94.4–99.4) | 90.0 | 95.8 | 0.898 |
| hs-TnT>13+Δ | 78.8% (71.9–84.6) | 85.0% (78.8–89.9) | 55.3 | 94.4 | 0.818 |
AUC: area under the curve; cTnT: cardiac troponin T; hs-TnT: high sensitivity troponin T; PPV: positive predictive value; NPV: negative predictive value.
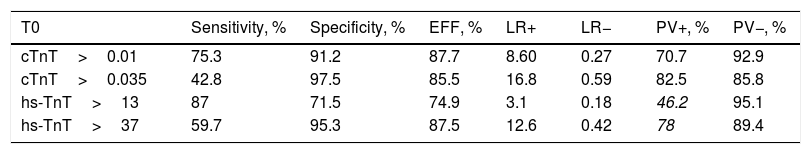
In addition, we evaluated the optimum cut-off points derived from the ROC analysis. The optimum point was established as 37ng/l (hs-TnT-ROC) (Table 3). The value for hs-TnT-ROC presented a sensitivity of 59.7% – far below that of hs-TnT, but greater than that of 4G TnT (CV) – and a specificity of 95.3% in the first measurement. This greater sensitivity with respect to 4G TnT (CV) persisted in the rest of the samplings.
Comparison of the diagnostic performance of hs-TnT ROC (> 37).
| T0 | Sensitivity, % | Specificity, % | EFF, % | LR+ | LR− | PV+, % | PV−, % |
|---|---|---|---|---|---|---|---|
| cTnT>0.01 | 75.3 | 91.2 | 87.7 | 8.60 | 0.27 | 70.7 | 92.9 |
| cTnT>0.035 | 42.8 | 97.5 | 85.5 | 16.8 | 0.59 | 82.5 | 85.8 |
| hs-TnT>13 | 87 | 71.5 | 74.9 | 3.1 | 0.18 | 46.2 | 95.1 |
| hs-TnT>37 | 59.7 | 95.3 | 87.5 | 12.6 | 0.42 | 78 | 89.4 |
cTnT: cardiac troponin T; hs-TnT: high sensitivity troponin T.
The time saving in reaching a diagnosis when using hs-TnT versus 4G cTn (CV) was 113min. The saving in turn would be 147min (p<0.001) in the case of patients admitted in under 3h from symptoms onset, and 63min (p=0.024) in the case of patients admitted over three hours from symptoms onset.
DiscussionThe data obtained in the present study are able to clarify certain issues regarding the use of hs-TnT in decision making, in view of the strong evidence of its usefulness as a diagnostic and prognostic tool in ACS. The diagnostic performance of our hs-TnT was ultimately seen to be greater than that of 4G cTn at each of the different sampling timepoints. Performance in turn was seen to decrease over the hours following symptoms onset. In our study we used relative changes between two samples (specifically 20%) that discretely improved the positive predictive value, though without becoming of practical use. In this regard, a Δ of 20% possibly may be too small and, as proposed by some authors, it could have been advisable to increase Δ to a higher percentage, since many cases of AMI were found to be at values discretely higher than the upper reference limit (URL).9,10 Other investigators propose relative changes of Δ (between 30 and 250%) to increase diagnostic specificity and improve the diagnosis of AMI.11–14 Mueller et al.15 published results that explain why relative changes of Δ fail for ruling in non-ST elevation myocardial infarction. With the use of absolute changes of Δ, an increase or decrease of at least 9.2ng/l for the population of patients with ACS and non-ACS, or of 6.9ng/l for a population with ACS, appears to be more adequate than relative changes of Δ for ruling out AMI. In addition to these problems with the decrease in specificity, there are differences in URL according to the test used. This highlights the importance of race, gender, age or the number of participants in the study,16 and adds increased difficulty in reaching consensus in the making of decisions.
In our study, the superiority of the sensitivity of hs-TnT in diagnosing non-ST elevation myocardial infarction was found to be greater in patients with recent symptoms onset, in concordance with the findings of other authors.17,18 For a patient reporting with chest pain for less than three hours, and with a second negative determination two hours after arrival, we can rule out AMI in almost 100% of the cases. This, combined with the clinical and ECG findings, can identify candidates for early discharge and outpatient management. On the other hand, patients diagnosed with non-ST elevation myocardial infarction and presenting long evolutive times (over 3h) show lesser diagnostic accuracy with hs-TnT, even when performing two serial determinations. In these cases we should evaluate the optimum cut-off points derived from the ROC analysis. In our study the cut-off point was 37ng/l, which would allow us to “rule in”, i.e., patients with scores above this cut-off point would have a very high probability of presenting AMI. It also has been argued that this decrease in specificity has been underestimated in the literature, and that it does not reflect the patients seen in the HES of our hospitals, where the mean age, comorbidities and cTn elevations tend to be high.
The time to diagnosis has been greatly reduced (from 247 to 71.5min).19,20 In our study the time saving for the diagnosis of non-ST elevation myocardial infarction was 147min in the case of patients reporting under three hours after symptoms onset and 63min when reporting after 3h.
Twenty-two of the patients (21.5%) with a diagnosis of UA at discharge would be non-ST elevation myocardial infarction cases after two determinations of hs-TnT with kinetics of over 20% between them. Up to 13.9% of the non-ACS patients (n=24) would exhibit positivity with hs-TnT on applying such kinetics, and would correspond to other disease conditions causing progressive cardiac damage, such as myocarditis, pulmonary embolism or takotsubo syndrome. Different hs-TnT analyses detect up to 27% more cases of AMI than the current method in patients with chest pain and a low or intermediate probability of ACS.20,21 It even could be speculated that the diagnosis of UA would disappear, as stressed by Sabatine et al.22 in patients with positive exercise testing evaluated with ultrasensitive techniques. In this study, hs-TnI was detectable in all the patients before the tests; remained without change in the patients without ischemia; and increased 24% in those with mild ischemia and 40% in those with moderate to severe ischemia. No changes were observed with 4G cTn. Consequently, the results referred to the diagnosis of non-ST elevation myocardial infarction using conventional cTn are a matter of concern, since myocardial damage has a limited measure and the cTn values do not always rise early, or their increments are not sufficient to allow early identification.
Rapid solution protocols should be introduced, whereby our patients with low ischemic risk and a normal ECG tracing would only spend 3–4h in hospital.23,24 It has been pondered whether a single hs-TnT determination suffices to rule out AMI,25 and other studies have assessed the possibility of a single determination if the hs-TnT value is <3ng/l (i.e., undetectable).24 All these situations need to be considered in future.
ConclusionsHigh sensitivity troponin T testing improves diagnostic performance, shortens the time to diagnosis of AMI, and is able to identify a larger number of patients with smaller infarctions, which previously were diagnosed as UA and which have a high mortality risk, when compared with conventional cTn. Furthermore, it shortens the time for ruling out cases, since AMI could be discarded within 2h. All this entails earlier treatment for patients with myocardial necrosis and a shortening of patient stay in the HES. However, in those individuals that report to the HES after three hours of symptoms onset, hs-TnT would not be able to improve upon the diagnostic performance of 4G cTn, and decision making therefore would need to be more careful. The hs-cTn tests are changing the management and concept of patients with chest pain suggestive of ACS. This makes it necessary for emergency and critical care physicians to become familiarized with their routine use.
Conflict of interestThe authors declare that they have no conflicts of interest.
Thanks are due to the authors of the TUSCA (Troponina Ultrasensible en los Síndromes Coronarios Agudos) trial: Aitor Alquézar, Miguel Santaló, Miguel Rizzi, Ignacio Gich, Margarita Grau, Alessandro Sionis, Jordi Ordóñez-Llanos, Javier Mercé (Hospital de la Santa Creu i Sant Pau, Barcelona), Alfonso Martín, Mar Muñoz (Hospital Severo Ochoa, Leganés, Madrid), Javier Povar, Joaquín Velilla, Pilar Calmarza (Hospital Miguel Servet, Zaragoza), Francisco Temboury, Carmen Ortiz (Hospital Clínico, Málaga) and José Balaguer and Arturo Carratalá (Hospital Clínico, Valencia).
Please cite this article as: Velilla Moliner J, Gros Bañeres B, Povar Marco J, Santaló Bel M, Ordoñez Llanos J, Martín Martín A, et al. Rendimiento diagnóstico de la troponina de alta sensibilidad en el síndrome coronario agudo sin elevación del segmento ST. Med Intensiva. 2020;44:88–95.