Noninvasive ventilation (NIV) with conventional therapy improves the outcome of patients with acute respiratory failure due to hypercapnic decompensation of chronic obstructive pulmonary disease (COPD) or acute cardiogenic pulmonary edema (ACPE). This review summarizes the main effects of NIV in these pathologies. In COPD, NIV improves gas exchange and symptoms, reducing the need for endotracheal intubation, hospital mortality and hospital stay compared with conventional oxygen therapy. NIV may also avoid reintubation and may decrease the length of invasive mechanical ventilation. In ACPE, NIV accelerates the remission of symptoms and the normalization of blood gas parameters, reduces the need for endotracheal intubation, and is associated with a trend toward lesser mortality, without increasing the incidence of myocardial infarction. The ventilation modality used in ACPE does not affect patient prognosis.
La ventilación no invasiva (VNI) junto con el tratamiento convencional mejora la evolución de los pacientes con insuficiencia respiratoria aguda por descompensación hipercápnica de la enfermedad pulmonar obstructiva crónica (EPOC) o por edema agudo de pulmón cardiogénico (EAPC). Esta revisión resume los principales efectos de la VNI en dichas enfermedades. En la EPOC la VNI mejora el intercambio de gases y la clínica, reduce la necesidad de intubación endotraqueal, la mortalidad hospitalaria y la estancia hospitalaria en comparación con la oxigenoterapia convencional. Además, puede evitar la reintubación y disminuir el tiempo de ventilación mecánica invasiva. En el EAPC el tratamiento con VNI acelera la remisión de los síntomas y la normalización gasométrica, reduce la necesidad de intubación endotraqueal y se asocia a una tendencia a menor mortalidad sin aumentar la incidencia de infarto de miocardio. La modalidad ventilatoria utilizada en el EAPC no afecta el pronóstico de los pacientes.
Since its introduction in the 1980s, the use of noninvasive ventilation (NIV) in patients with acute respiratory failure (ARF) has gradually increased as an alternative to endotracheal intubation in the Intensive Care Unit (ICU).1–3 According to some reports, the use of NIV has actually doubled over a period of 7 years.1,3 NIV is employed in 11–23% of all the ventilated patients,1,3,4 which means an 8–13% utilization rate among all patients admitted to the ICU.1,3,4
Noninvasive ventilation is regarded as the first choice management along with conventional treatment in hypercapnic ARF in the context of chronic obstructive pulmonary disease (COPD) and in ARF related to acute cardiogenic pulmonary edema (ACPE) or immune depression, in the absence of contraindications. It is important to underscore that NIV failure potentially may be associated with prolonged invasive mechanical ventilation5 and an increased mortality rate. In this context, mortality rates of over 45% have been reported in non-selected patient populations.1 Correct patient selection for the use of NIV is essential, since use of the technique is not intended to delay endotracheal intubation but to avoid it.
The present review focuses on the application of NIV in two very prevalent types of ARF in our setting, and in which NIV has shown clear benefits, i.e., in patients with COPD and patients with ACPE.
The present review uses the term NIV in reference to any type of noninvasive ventilatory assistance, whether inspiratory, expiratory or both, and including continuous positive airway pressure (CPAP) for the application of positive end-expiratory pressure (PEEP) in a noninvasive manner (using a mask with its own CPAP system or connected to MV) and noninvasive positive pressure ventilation (NIPPV) for the application of NIV with inspiratory support (pressure support, proportional assist ventilation (PAV), BiPAP, PS S/T, etc.) with or without PEEP.
Use of noninvasive ventilation in patients with chronic obstructive pulmonary diseaseEpidemiologyChronic obstructive pulmonary disease is associated with important morbidity–mortality and affects up to 10% of the Spanish population according to the EPI-SCAN study.6 The exacerbation of COPD is the cause of 20% of all emergency care consultations, and it is estimated that about 25% of the affected patients present acidosis upon arrival. Management of the exacerbations is based on drug treatment and ventilatory support. Evidence of the benefits of NIV in hypercapnic ARF among patients with COPD has been widely documented in the scientific literature, and the technique is warranted both by a high degree of recommendation in the different clinical guides7 and by the progressive increase in its use by specialists in recent years. A registry of hospitals in the United States during the period 1998–2008 referred to the use of MV in patients with COPD exacerbation revealed a progressive 462% increase in the use of NIV over the years (from 1% to 4.5% of all admissions), together with a 42% decrease in the use of invasive MV (from 6% to 3.5% of all admissions).8 In Europe, during the year 2008 a study on the use of NIV found the management of hypercapnic ARF to be the main indication of the technique (48% of the cases).9
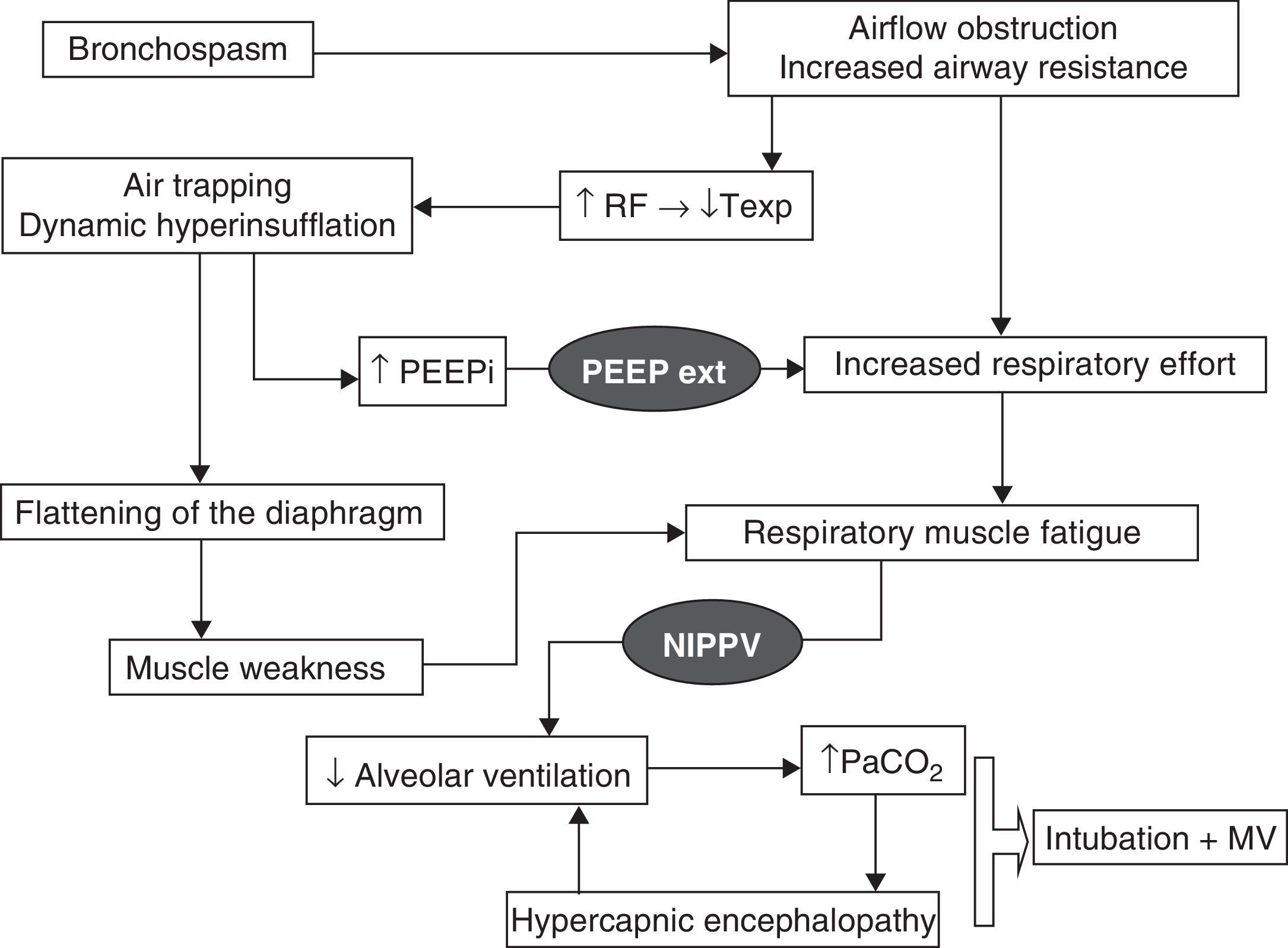
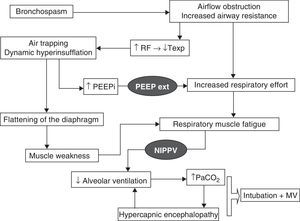
Physiopathological effectsChronic obstructive pulmonary disease exacerbation is characterized by an increase in mechanical loading due to the high airflow resistances and dynamic hyperinsufflation which initially give rise to an increase in respiratory effort and intrinsic PEEP. In later stages the condition evolves toward muscle fatigue and consequent clinical and blood gas deterioration. Dynamic hyperinsufflation occurs because of an expiratory time that is too short to reach the resting volume of the respiratory system, and increases with each successive respiration. Under these circumstances, NIPPV has beneficial effects, which will be summarized below and are represented in Fig. 1. On one hand, application of a positive inspiratory pressure reduces respiratory labor with an increase in alveolar ventilation and a lowering of the respiratory frequency–this in turn produces a prolongation of the expiratory time with lesser air trapping. In relation to gas exchange, and due to the increase in minute volume, we observe a lowering of PaCO2 and an increase in pH. On the other hand, the application of external PEEP counters the inspiratory effort needed to surpass the intrinsic PEEP due to the dynamic hyperinsufflation and which can originate up to 60% of the increase in respiratory effort–thereby reducing muscle work. Fig. 2 proposes an interventional algorithm for patients presenting COPD with hypercapnic ARF.
Physiopathology of hypercapnic acute respiratory failure in COPD and the processes that are blocked by the action of mechanical ventilation (represented in black). RF, respiratory frequency; PEEPext, positive end-expiratory pressure programmed by the ventilator; PEEPi, intrinsic PEEP; Texp, expiratory time; MV, mechanical ventilation; NIPPV, noninvasive positive pressure ventilation.
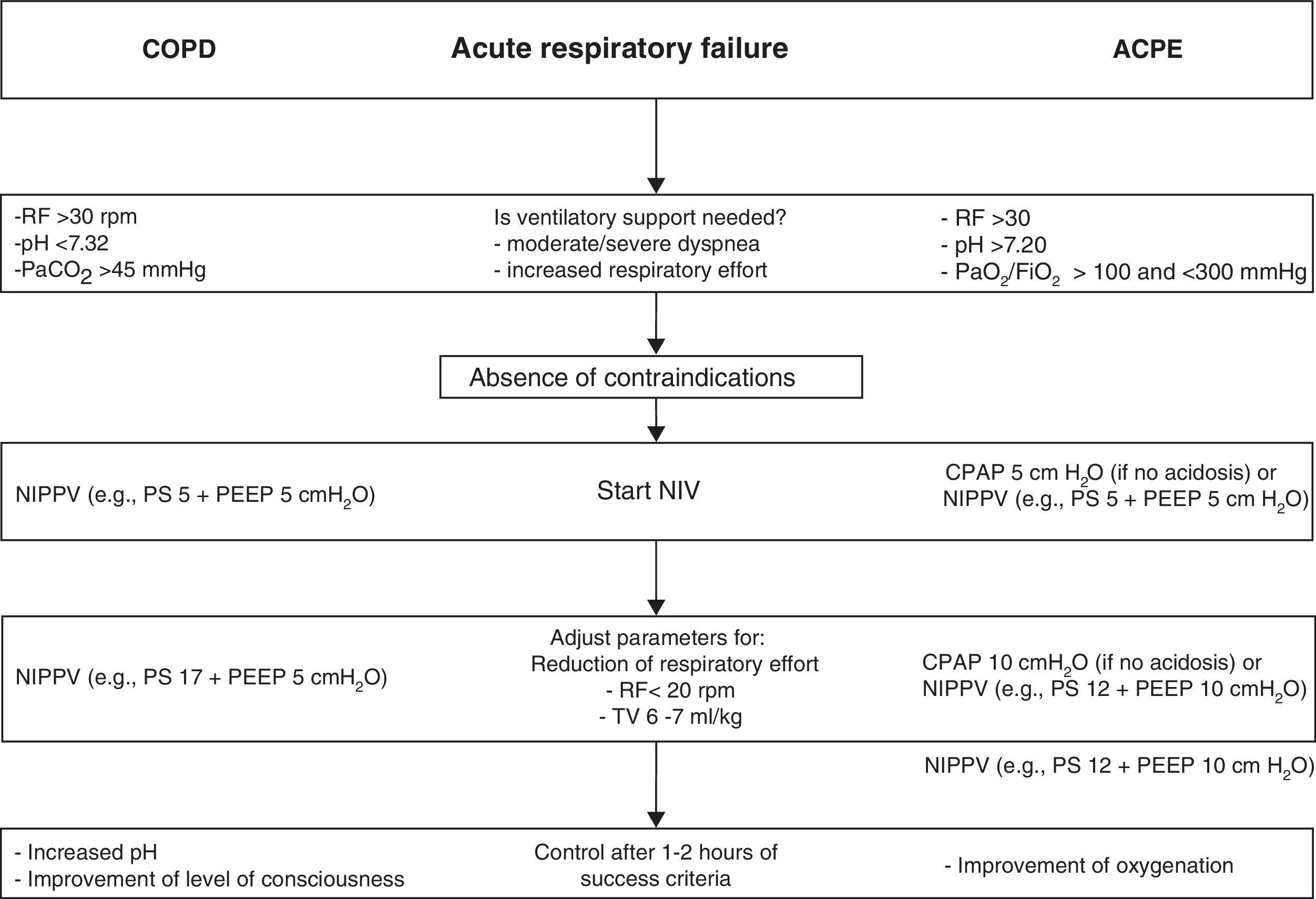
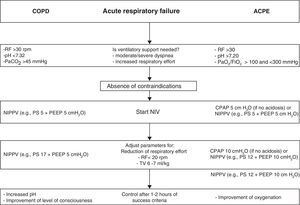
Application of noninvasive ventilation in acute respiratory failure in chronic obstructive pulmonary disease with hypercapnic decompensation, and in acute cardiogenic pulmonary edema. The ventilatory parameters used are orientative according to tolerance of the technique and the presence of leakage. CPAP, continuous positive airway pressure; ACPE, acute cardiogenic pulmonary edema; COPD, chronic obstructive pulmonary disease; RF, respiratory frequency; PEEP, positive end-expiratory pressure; NIPPV, noninvasive ventilation with inspiratory support (PS, pressure support; BiPAP, proportional assist ventilation, etc.); TV, tidal volume.
Hypercapnic ARF in patients with COPD is the main indication of NIPPV–the first studies having been published over two decades ago.10
Brochard et al.10 evaluated the physiological benefits of NIPPV in patients with exacerbated COPD and observed significant improvement of dyspnea, respiratory frequency and gas exchange, with favorable changes in pH, PaCO2 and PaO2. After comparing the results with an oxygen therapy control group, the use of NIPPV was seen to be associated with a lesser need for intubation (7% vs 77%, p<0.001) and a shorter stay in the ICU (7 vs 19days, p<0.01).
Posteriorly, the same group conducted a randomized study of 85 patients with exacerbated COPD,11 comparing the use of NIPPV with conventional oxygen therapy. The NIPPV group showed significant improvement in blood gases (pH, PaCO2 and PaO2) and respiratory frequency, together with a significant decrease in the need for intubation (26% vs 74%, p=0.001), the presence of complications, hospital stay (23 vs 35days, p=0.02) and hospital mortality (9% vs 29%, p=0.02).
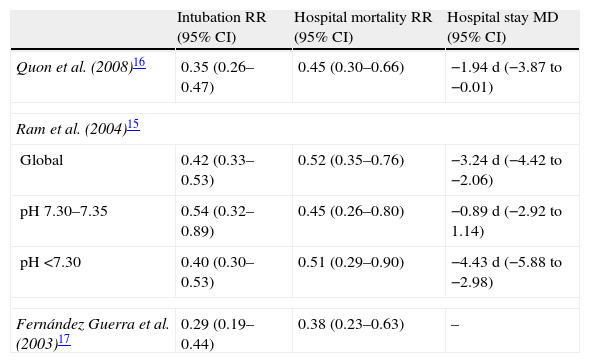
Since then, many studies have confirmed the benefits of NIPPV in patients with COPD and hypercapnic ARF in terms of a lesser need for intubation, diminished hospital mortality and a shortened hospital stay.12 Different metaanalyses have examined these results, revealing significant decreases in the need for intubation (18–28%) and in hospital mortality (10–13%),13,14 together with a significant shortening of hospital stay (2–5days)14–17 (Table 1).
Results of different metaanalyses on the effects of the application of noninvasive ventilation on intubation, hospital mortality and hospital stay in patients with COPD and hypercapnic acute respiratory failure.
| Intubation RR (95% CI) | Hospital mortality RR (95% CI) | Hospital stay MD (95% CI) | |
| Quon et al. (2008)16 | 0.35 (0.26–0.47) | 0.45 (0.30–0.66) | −1.94 d (−3.87 to −0.01) |
| Ram et al. (2004)15 | |||
| Global | 0.42 (0.33–0.53) | 0.52 (0.35–0.76) | −3.24 d (−4.42 to −2.06) |
| pH 7.30–7.35 | 0.54 (0.32–0.89) | 0.45 (0.26–0.80) | −0.89 d (−2.92 to 1.14) |
| pH <7.30 | 0.40 (0.30–0.53) | 0.51 (0.29–0.90) | −4.43 d (−5.88 to −2.98) |
| Fernández Guerra et al. (2003)17 | 0.29 (0.19–0.44) | 0.38 (0.23–0.63) | – |
MD, mean difference; CI, confidence interval; RR, relative risk.
Most of the patients included in the abovementioned studies had respiratory acidosis. On considering the studies that included patients with less severe exacerbations (initial pH>7.30), NIPPV did not show advantages with respect to conventional oxygen therapy,18,19 as has been confirmed in a metaanalysis13 on evaluating the subgroup of patients with pH>7.30. In contrast, another metaanalysis that did not include the study of Keenan et al.18 detected a significant reduction in the percentage of intubations and in mortality in this subgroup of patients15 (Table 1).
In sum, the application of NIPPV together with regular medical treatment in patients with COPD and hypercapnic ARF reduces the incidence of orotracheal intubation, hospital mortality and hospital stay.
Postextubation noninvasive ventilationInvasive mechanical ventilation for over three days in patients with COPD is associated with complications such as muscle atrophy and an increased risk of nosocomial pneumonia. It therefore seems prudent to design strategies to shorten the duration of MV. On the contrary, nosocomial pneumonia is associated with increased patient mortality. NIPPV could be used as ventilatory support after invasive MV in three contexts: to facilitate weaning from invasive MV, to prevent postextubation ARF, or for the management of postextubation ARF.
Noninvasive ventilation to facilitate weaning from invasive mechanical ventilationNava et al.20 evaluated the impact of NIPPV in patients with COPD exacerbation requiring invasive MV for at least 36–48h and who failed the spontaneous breathing test. The patients were randomized to two groups: the usual weaning process was continued in one group, while in the other group the patients were extubated and NIPPV was immediately applied. The group that received NIPPV showed a lesser incidence of pneumonia, a shorter stay in the ICU (15 vs 24days, p=0.02), and increased survival after 90 days (92% vs 72%, p<0.01).
Another study involving a more heterogeneous patient population (58% with COPD) with MV weaning test failure during three consecutive days compared extubation and the application of NIPPV vs conventional weaning.21 The group treated with NIPPV showed a lesser incidence of nosocomial pneumonia and a lesser need for tracheostomy, as well as a shorter stay both in the ICU and in hospital, and superior survival in the ICU and after 90 days.
The recently published VENISE study22 included 208 patients with hypercapnic ARF (69% with COPD) in which a first weaning test had failed. The patients were randomized to three treatment groups: a first group underwent conventional weaning (based on daily weaning tests or progressive reduction of pressure support); a second group was extubated followed by conventional oxygen therapy; and a third group was extubated and connected to NIPPV. No significant inter-group differences were observed in the percentage of reintubations after 7 days (30%, 37% and 32%, respectively). It should be noted that the use of NIPPV as rescue technique was allowed, with rescue therapy success rates of 45% in the conventional weaning group and 58% in the oxygen therapy group. However, a significant decrease was recorded in the risk of postextubation ARF in the NIPPV group (54%, 71% and 33%, respectively, p<0.001). The fact that a group was subjected to extubation after MV weaning test failure, followed by oxygen therapy without adherence to any weaning protocol, has been criticized from the ethical perspective.
Noninvasive ventilation in the prevention of postextubation acute respiratory failureNoninvasive positive pressure ventilation applied immediately after protocolized extubation could reduce the risk of postextubation ARF and avoid the need for reintubation–the latter being an independent risk factor for nosocomial pneumonia and mortality.
The different studies made to assess the efficacy of NIPPV in preventing postextubation ARF have obtained disparate results,23–25 with the observation of possible benefits in highly selected patients with a high risk of developing postextubation ARF (Table 2). In this sense, Nava et al.,24 in patients with reintubation risk, of which 32% had COPD, reported that the use of NIPPV immediately after extubation compared with conventional management is associated with a lesser percentage of reintubations, and results in a 10% reduction of the risk of death in the ICU. In this same line, Ferrer et al.,25 in patients at a high risk of developing postextubation ARF, of which 51% had COPD, reported a significant decrease in postextubation ARF in the group treated with NIPPV (16% vs 33%, p=0.029), together with a decrease in mortality in the ICU (2% vs 12%, p=0.015) that did not persist after 90 days. In this same study, a sub-analysis comparing patients with and without hypercapnia during the spontaneous breathing test found the application of NIPPV to reduce mortality both in the ICU and after 90 days in the patients with hypercapnia. For this reason, these investigators, in a more recent study involving 106 intubated patients with chronic respiratory diseases (62% with COPD) and hypercapnic ARF in the MV weaning phase and a well-tolerated weaning test, compared treatment with postextubation NIPPV vs conventional oxygen therapy.26 Here again, the group receiving NIPPV showed a lesser incidence of postextubation ARF (15% vs 48%, p<0.0001) and a decrease in mortality after 90 days (p=0.015).
Risk factors for postextubation acute respiratory failure.
| Nava et al. (2005)24 |
| More than one consecutive MV weaning test failure |
| Antecedents of heart failure |
| PaCO2 >45mmHg postextubation |
| More than one comorbid condition |
| Upper airway stridor not requiring immediate reintubation |
| Non-productive cough or bronchorrhea |
| Ferrer et al. (2006)25 |
| Age over 65 years |
| Heart failure as cause of intubation |
| APACHE II >12 points on day of extubation |
The studies made on the use of NIPPV in the management of postextubation ARF offer disappointing results,27,28 observing even increased mortality in relation to the use of NIPPV.27 Nevertheless, mention should be made of the low percentage of patients with COPD included in both studies (about 10%)–a fact that could warrant the conduction of new studies in this subgroup of patients.
Ventilation modesThe pressure support mode has commonly been used in NIV applied to hypercapnic ARF in COPD patients. Since a constant level of inspiratory support is provided, a decrease is observed in the variability of the respiratory pattern, and this in turn may be associated with asynchronies and overdistension. More recent modes have been introduced that use a more physiological inspiratory support system in order to improve synchronization between the patient and the ventilator. One of these modes is proportional assist ventilation (PAV), in which inspiratory support proportional to the inspiratory flow is provided. The degree of support is programmed according to the resistance of the airway and compliance. However, although this mode is interesting, it has not yet been able to demonstrate advantages with respect to pressure support.29 Another recently introduced ventilation mode is the neurally adjusted ventilatory assist mode (NAVA), which is based on the detection of diaphragm contraction through esophageal electrodes. NIV with this mode appears to reduce the severe asynchronies observed with pressure support, while affording similar results in terms of gas exchange.30
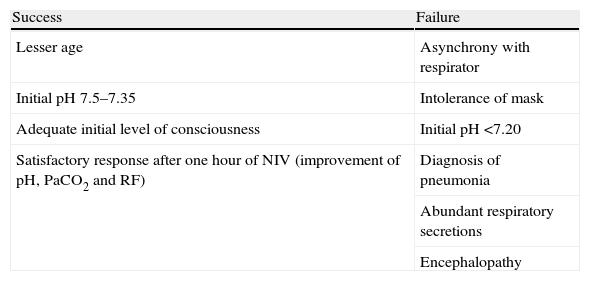
Predictors of noninvasive ventilation success and failureThe percentage of failure of NIPPV in patients with COPD and hypercapnic ARF that finally need endotracheal intubation is less than 25%.11,12,31,32 The parameters associated with success and failure of the technique are summarized in Table 3. An adequate level of patient consciousness at the start of NIPPV, and the improvement of pH, PaCO2 and level of consciousness after one hour of NIPPV are related to a good response to NIPPV in hypercapnic ARF in patients with COPD.32 In patients in whom NIV fails, a recent observational study indicates that the range of time on NIV prior to intubation is very wide and that an increase in the duration of NIV prior to intubation is associated with a decrease in hospital survival–though only in the group of patients without previous cardiac or respiratory disease33–hence the importance of the time window for evaluating the success or failure of the technique, with a view to avoiding delays in intubation that could worsen the prognosis.
Predictors of noninvasive ventilation success and failure.
| Success | Failure |
| Lesser age | Asynchrony with respirator |
| Initial pH 7.5–7.35 | Intolerance of mask |
| Adequate initial level of consciousness | Initial pH <7.20 |
| Satisfactory response after one hour of NIV (improvement of pH, PaCO2 and RF) | Diagnosis of pneumonia |
| Abundant respiratory secretions | |
| Encephalopathy |
RF, respiratory frequency.
On the other hand, earliness in the introduction of NIPPV is fundamental for the success of the technique. Patient extenuation, which is associated with intense respiratory acidosis and possible encephalopathy, is to be avoided since these conditions are characterized by a high failure risk. Likewise, the presence of pneumonia or acute respiratory distress syndrome (ARDS) is also associated with failure of the technique.34
Poor tolerance of the technique is associated with NIPPV failure–asynchrony being one of the most relevant factors in this sense. Asynchrony is defined by the presence of an ineffective trigger, double trigger, auto-trigger, premature cycling or late cycling, and is present in up to 43% of all patients–the level of pressure support and the magnitude of leakage being the main factors responsible for severe asynchronies.35
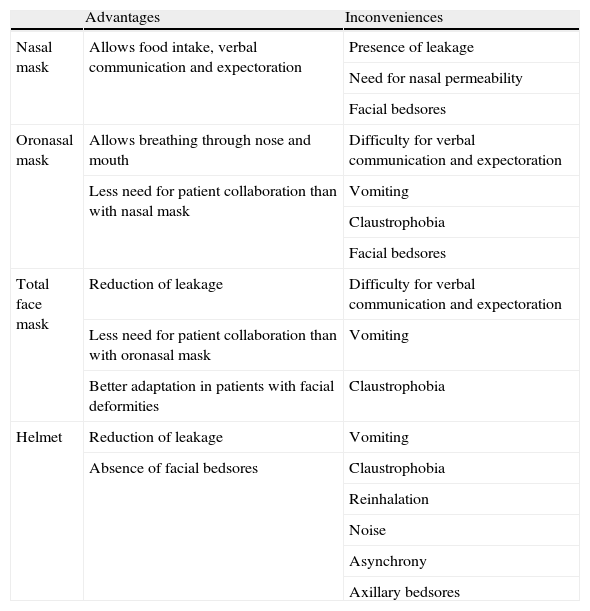
The type of mask used also plays an important role in defining patient tolerance of the technique. The current main interfaces comprise nasal, oronasal, facial or helmet masks. In this context, oronasal masks are the preferred option in the presence of ARF for both patients and specialists.9Table 4 describes the main advantages and inconveniences of the different interfaces.
Principal advantages and inconveniences of the main interfaces for the application of noninvasive ventilation.
| Advantages | Inconveniences | |
| Nasal mask | Allows food intake, verbal communication and expectoration | Presence of leakage |
| Need for nasal permeability | ||
| Facial bedsores | ||
| Oronasal mask | Allows breathing through nose and mouth | Difficulty for verbal communication and expectoration |
| Less need for patient collaboration than with nasal mask | Vomiting | |
| Claustrophobia | ||
| Facial bedsores | ||
| Total face mask | Reduction of leakage | Difficulty for verbal communication and expectoration |
| Less need for patient collaboration than with oronasal mask | Vomiting | |
| Better adaptation in patients with facial deformities | Claustrophobia | |
| Helmet | Reduction of leakage | Vomiting |
| Absence of facial bedsores | Claustrophobia | |
| Reinhalation | ||
| Noise | ||
| Asynchrony | ||
| Axillary bedsores |
Although most studies on NIPPV have been carried out in ICUs, the use of the technique has spread in recent years to Emergency Care Departments and Pneumology wards, with equally satisfactory results.15 In a European registry published in 2008,9 the use of NIPPV was found to be more widespread among pneumologists than among intensivists or anesthetists. In patients with NIV failure risk (Table 3), the technique should be used in units that have expert professionals and are able to react quickly in the event of failure of the technique.31,36 These requirements are met by Intensive or Semicritical Care Units. When NIPPV is started outside this setting and gas exchange, respiratory frequency or dyspnea fails to improve; the degree of encephalopathy worsens; or hemodynamic deterioration is moreover observed, then transfer of these patients to such units should be considered.
Use of noninvasive ventilation in patients with acute cardiogenic pulmonary edemaEpidemiologyHeart failure affects 2–3% of the population, and its prevalence progressively increases with age–reaching 10–20% among patients between 70 and 80 years of age. The mean age of patients with heart failure in the industrialized world is 75 years. The disease is the cause of 5% of all emergency hospital admissions and occupies 10% of the global hospital beds.37 On the other hand, NIV is used in 80% of the episodes of acute cardiogenic pulmonary edema (ACPE).31
Physiological effects of noninvasive ventilationAcute cardiogenic pulmonary edema is characterized by an increase in left ventricle filling pressures that retrogradely generate a rise in pulmonary capillary pressure, followed by fluid extravasation toward the pulmonary interstitial compartment and alveolar spaces. All these in turn produce an increase in airway resistances, a decrease in lung diffusion capacity, a drop in functional residual capacity, and an increased intrapulmonary shunt effect. The result is the development of hypoxemia with an increase in respiratory effort.
The application of intrathoracic positive pressure in patients with lung edema, particularly with PEEP, whether with or without inspiratory support, on one hand produces a decrease in venous return and in right ventricle preload, and on the other reduces left ventricle postload secondary to a decrease in transmural pressure (transmural pressure=intraventricular pressure−intrathoracic pressure), thereby improving cardiac output and myocardial contractility. Furthermore, in these patients, the application of positive pressure favors alveolar recruitment and increases functional residual capacity, lung compliance and alveolar ventilation, with a reduction of the intrapulmonary shunt and respiratory effort, and improved oxygenation (Table 5).
Physiological effects of positive pressure ventilation in patients with acute cardiogenic pulmonary edema.
| Cardiovascular effects |
| Decreased right ventricle preload |
| Decreased left ventricle postload |
| Increased myocardial contractility |
| Increased cardiac output |
| Pulmonary effects |
| Alveolar recruitment |
| Increased functional residual capacity |
| Increased lung compliance |
| Decreased intrapulmonary shunt |
| Decreased respiratory effort |
Separate mention is required of situations of cardiogenic shock or right ventricular failure, in which the application of NIV may prove deleterious.37
Clinical effectsFaster clinical (reduction of respiratory frequency and of dyspneic sensation) and blood gas improvements (increased PaO2, reduction of PaCO2 and of acidosis) have been observed with the application of NIV compared with conventional oxygen therapy,38 with both CPAP39,40 and NIPPV41,42–the differences being manifest from 30min of application of the technique.
Both NIPPV and CPAP show rapid response times (from 30min)43–clinical resolution generally being faster with the application of NIPPV,44,45 reaching differences of almost one hour between the two techniques.44 Likewise, studies with a physiological basis have shown that although the use of CPAP is associated with a decrease in respiratory effort among patients with ACPE, this effect is greater when NIPPV is applied.46
Other studies report that NIV in ACPE may be especially beneficial for patients with hypercapnia, by reducing the percentage of intubations with respect to non-hypercapnic individuals.41,47 However, in the study published by Nava et al.,41 72% of the subgroup of hypercapnic patients consisted of individuals who moreover had COPD, while in the non-hypercapnic subgroup these individuals represented only 11%. This suggests that it is probably the underlying disease, not hypercapnia alone, that constitutes the marker of success. Nouira et al.44 conducted a sub-analysis of the patients with hypercapnia. In these subjects the authors compared CPAP vs NIPPV, and no differences in patient course were seen between the two techniques.
Effects on hospital stayThe mean duration of NIV in ACPE is between 2 and 4h.48 Hospital stay is not modified by the use of NIV vs conventional treatment, and is reported to be 8–14 days.38,39,41,42,48
Effects on the need for endotracheal intubationThe percentage of endotracheal intubation in patients with ACPE subjected to conventional oxygen therapy is 10–28%, depending on the series. However, when using NIV, this percentage drops to 3–12%, with both CPAP and NIPPV.49
The largest study to date on the effects of NIV in ACPE was published by Gray et al. in 2008.38 This study randomized 1069 patients to three treatment groups: CPAP, NIPPV and a control group administered conventional oxygen therapy. The authors observed faster improvement of dyspneic sensation and of pH with the use of CPAP or NIPPV in comparison with conventional oxygen therapy, with no significant differences in mortality or in the need for intubation between the groups. Likewise, no differences were recorded in hospital stay or in the incidence of myocardial infarction. It must be noted that this study excluded the more seriously ill patients (which are the subjects who possibly could derive greater benefit from the technique), with a global percentage of intubations of under 3%, and that moreover 15% of the patients in the conventional oxygen therapy group were rescued with NIV.
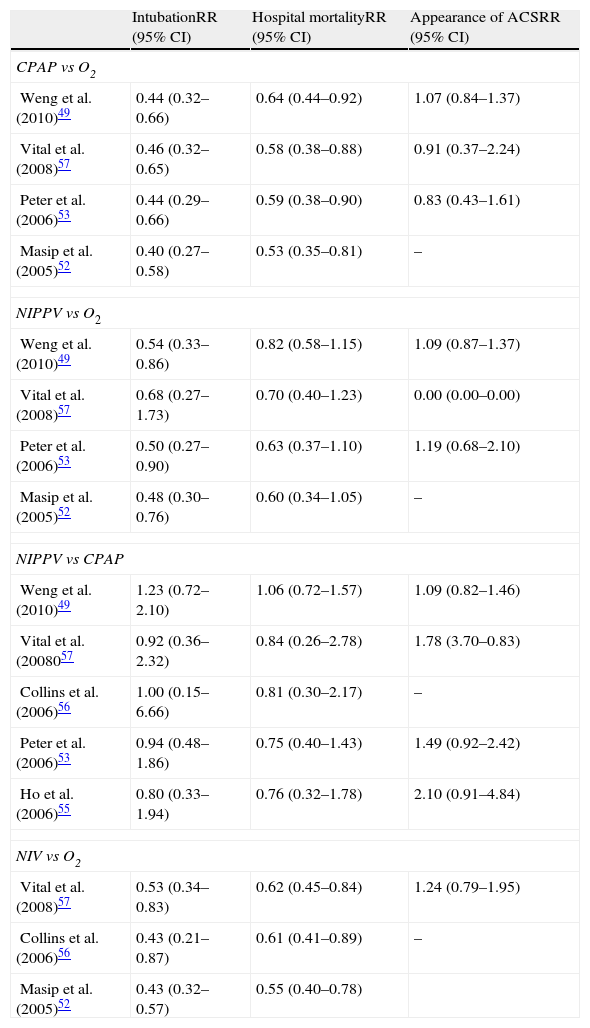
Most randomized studies report a clear tendency toward a lesser need for intubation with NIV.40,48,50,51 The different metaanalyses49,52–57 coincide in reporting a significant decrease in the percentage of intubations with the application of NIV, in comparison with conventional oxygen therapy (as can be seen in Table 6), on considering both CPAP and NIPPV–with calculation of an absolute reduction of the need for intubation of 22% and 18%, respectively.54
Results of different metaanalyses on the application of noninvasive ventilation in patients with acute cardiogenic pulmonary edema. Effects upon intubation, hospital mortality and the appearance of acute coronary syndrome.
| IntubationRR (95% CI) | Hospital mortalityRR (95% CI) | Appearance of ACSRR (95% CI) | |
| CPAP vs O2 | |||
| Weng et al. (2010)49 | 0.44 (0.32–0.66) | 0.64 (0.44–0.92) | 1.07 (0.84–1.37) |
| Vital et al. (2008)57 | 0.46 (0.32–0.65) | 0.58 (0.38–0.88) | 0.91 (0.37–2.24) |
| Peter et al. (2006)53 | 0.44 (0.29–0.66) | 0.59 (0.38–0.90) | 0.83 (0.43–1.61) |
| Masip et al. (2005)52 | 0.40 (0.27–0.58) | 0.53 (0.35–0.81) | – |
| NIPPV vs O2 | |||
| Weng et al. (2010)49 | 0.54 (0.33–0.86) | 0.82 (0.58–1.15) | 1.09 (0.87–1.37) |
| Vital et al. (2008)57 | 0.68 (0.27–1.73) | 0.70 (0.40–1.23) | 0.00 (0.00–0.00) |
| Peter et al. (2006)53 | 0.50 (0.27–0.90) | 0.63 (0.37–1.10) | 1.19 (0.68–2.10) |
| Masip et al. (2005)52 | 0.48 (0.30–0.76) | 0.60 (0.34–1.05) | – |
| NIPPV vs CPAP | |||
| Weng et al. (2010)49 | 1.23 (0.72–2.10) | 1.06 (0.72–1.57) | 1.09 (0.82–1.46) |
| Vital et al. (2008057 | 0.92 (0.36–2.32) | 0.84 (0.26–2.78) | 1.78 (3.70–0.83) |
| Collins et al. (2006)56 | 1.00 (0.15–6.66) | 0.81 (0.30–2.17) | – |
| Peter et al. (2006)53 | 0.94 (0.48–1.86) | 0.75 (0.40–1.43) | 1.49 (0.92–2.42) |
| Ho et al. (2006)55 | 0.80 (0.33–1.94) | 0.76 (0.32–1.78) | 2.10 (0.91–4.84) |
| NIV vs O2 | |||
| Vital et al. (2008)57 | 0.53 (0.34–0.83) | 0.62 (0.45–0.84) | 1.24 (0.79–1.95) |
| Collins et al. (2006)56 | 0.43 (0.21–0.87) | 0.61 (0.41–0.89) | – |
| Masip et al. (2005)52 | 0.43 (0.32–0.57) | 0.55 (0.40–0.78) | |
CPAP, continuous positive airway pressure; CI, confidence interval; O2, conventional oxygen therapy; RR, relative risk; ACS, acute coronary syndrome; NIV, noninvasive ventilation; NIPPV, noninvasive positive pressure ventilation.
On separately analyzing the effect of the application of NIPPV or CPAP vs conventional oxygen therapy, both techniques have demonstrated their efficacy in reducing the need for intubation, particularly when CPAP is used. The global studies published to date have been unable to demonstrate the superiority of one technique over the other (Table 6), and in this sense similar intubation percentages are seen with the use of both CPAP and NIPPV in application to ACPE.
Effects on hospital mortalityThere is controversy regarding the impact of NIV upon hospital mortality in patients with ACPE. Although most randomized studies report a tendency toward lesser mortality in the groups treated with NIV, they have been unable to demonstrate significant reductions in patient mortality,38,48,51,58 probably because of the limited statistical power involved. The mortality rate among the patients receiving conventional oxygen therapy is about 16%, vs 10% in those who receive NIV.49
The effect of NIV upon hospital mortality has been reflected in different metaanalyses comparing the technique with conventional oxygen therapy.49,52–57 Although these analyses are based on studies with a limited number of patients and with variable methodological designs, they agree that the use of NIV is associated with a significant decrease in hospital mortality (Table 6).
On separately analyzing the two ventilation modes, the application of CPAP is seen to maintain a significant reduction in mortality compared with conventional treatment, with an absolute reduction of 13%,54 while the application of NIPPV only reveals a tendency toward decreased mortality (absolute reduction 7%).54 Nevertheless, no significant differences are observed on comparing the two techniques (Table 6).
Use of noninvasive ventilation in acute coronary syndromeThe concern about acute myocardial infarction (AMI) during the application of NIV is mainly attributable to a randomized study involving patients with ACPE in which a comparison was made of NIPPV vs CPAP–a tendency toward more frequent AMI being observed in the group of patients treated with NIPPV. However, this study only included a total of 27 patients, and the NIPPV treatment group had a greater proportion of patients with chest pain and left bundle block at the start of the study.45 Posteriorly, numerous randomized studies have been unable to confirm these results,41,43,44,48,58,59 though it is true that most of them excluded patients with acute coronary syndrome (ACS) as a cause of ACPE43,44,59–particularly those patients amenable to immediate revascularization.41,48,58
The different metaanalyses likewise have not detected a relationship between AMI and the use of NIV in patients with ACPE49,53–55,57 either on comparing NIV with conventional oxygen therapy or on comparing the two NIV techniques with each other, both during treatment with NIV49,53,54,57 and after its application57 (Table 6).
On the basis of the existing results, the recommendation is to use NIV in ACPE in the absence of shock or ACS amenable to urgent revascularization, and making no distinction between CPAP and NIPPV.7
Use of noninvasive ventilation in the pre-hospital settingTreatment in ACPE must be provided on an early basis. This therefore also applies to NIV, which has been shown to be cost-effective provided it is introduced early. In the pre-hospital setting, a randomized study of 71 patients with ARF (67% of whom had ACPE) compared the use of conventional oxygen therapy vs CPAP (10cmH2O). A significant reduction was observed in the percentage of intubations and in patient mortality in the group treated with CPAP.60 In another study in the pre-hospital setting involving 124 patients with ACPE, a comparison was made of the effects of the immediate application of CPAP during the first 15min, followed by the addition of conventional treatment (oxygen and medication), vs the immediate provision of conventional treatment followed 15min later by the addition of CPAP.50 The early CPAP group showed faster improvement of the physiological parameters (dyspnea, respiratory frequency, PaCO2 and PaO2), a lesser need for inotropic drugs and intubation, and a tendency toward lesser mortality.
Ventilation modesIn applying CPAP, we usually employ CPAP masks without the need for a respirator–a fact that simplifies treatment. This is particularly interesting in the pre-hospital setting or in Departments with limited resources. The PEEP values used in the treatment of ACPE vary from 5 to 12.5cmH2O, depending on the study.
In applying NIPPV, a respirator is essential. For the treatment of ACPE, the PEEP values (in the same way as in CPAP) should not be less than 5cmH2O, and the inspiratory support can vary between 6 and 15cmH2O in order to secure tidal volumes of about 8ml/kg. Fig. 2 proposes an interventional ventilation algorithm for patients with ACPE.
As we have explained in this review, on choosing a ventilation mode in ACPE it has not been possible to demonstrate the superiority of one technique over the other in terms of the need for intubation, hospital mortality, hospital stay or the incidence of myocardial infarction. The technique chosen should firstly depend on equipment availability. In the equal availability of resources, and in the absence of confirmation of differences in patient outcome between the two techniques, we preferentially should use NIPPV instead of CPAP, since shortening of the time to clinical resolution in itself should be a primary objective in the management of these patients.
ConclusionsThe use of NIV is currently regarded as a first-line management option in patients with hypercapnic ARF in the context of COPD or in ACPE.
In patients with COPD and hypercapnic ARF, the use of NIPPV has been shown to reduce the time to clinical and blood gas resolution, with fewer intubations, lesser hospital mortality and a shorter hospital stay. As such, the technique is currently included within the conventional treatment of ARF in these patients. The prophylactic use of NIPPV in weaning from invasive MV can be considered in high-risk COPD patients.
Likewise, in ACPE, NIV has been found to shorten the time to clinical resolution, with faster improvement of gas exchange, and reduces the need for endotracheal intubation. A tendency toward lesser mortality is also observed with the use of both CPAP and NIPPV. The indicated ventilation mode in ACPE fundamentally depends on the available resources, though NIPPV is to be preferred due to its greater effect in reducing respiratory effort and faster clinical improvement.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Rialp Cervera G, del Castillo Blanco A, Pérez Aizcorreta O, Parra Morais L, por el GT-IRA de la SEMICYUC. Ventilación mecánica no invasiva en la enfermedad pulmonar obstructiva crónica y en el edema agudo de pulmón cardiogénico. Med Intensiva. 2014;38:111–121.