Editado por: Federico Gordo - Medicina Intensiva del Hospital Universitario del Henares (Coslada-Madrid)
Última actualización: Octubre 2023
Más datosTo determine the incidence and impact of Aspergillus spp. isolation (AI) on ICU mortality in critically ill patients with severe influenza pneumonia during the first 24h of admission.
DesignSecondary analysis of an observational and prospective cohort study.
SettingICUs voluntary participating in the Spanish severe Influenza pneumonia registry, between June 2009 and June 2019.
PatientsConsecutive patients admitted to the ICU with diagnosis of severe influenza pneumonia, confirmed by real-time polymerase chain reaction.
InterventionsNone.
Main variables of interestIncidence of AI in respiratory samples. Demographic variables, comorbidities, need for mechanical ventilation and the presence of shock according at admission. Acute Physiology and Chronic Health Evaluation II (APACHE II) scale calculated on ICU admission.
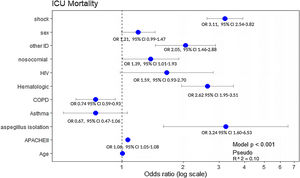
Results3702 patients were analyzed in this study. AI incidence was 1.13% (n=42). Hematological malignancies (OR 4.39, 95% CI 1.92–10.04); HIV (OR 3.83, 95% CI 1.08–13.63), and other immunosuppression situations (OR 4.87, 95% CI 1.99–11.87) were factors independently associated with the presence of Aspergillus spp. The automatic CHAID decision tree showed that hematologic disease with an incidence of 3.3% was the most closely AI related variable. Hematological disease (OR 2.62 95% CI 1.95–3.51), immunosuppression (OR 2.05 95% CI 1.46–2.88) and AI (OR 3.24, 95% CI 1.60–6.53) were variables independently associated with ICU mortality.
ConclusionsEmpirical antifungal treatment in our population may only be justified in immunocompromised patients. In moderate-high risk cases, active search for Aspergillus spp. should be implemented.
Determinar la incidencia y el impacto sobre la mortalidad del aislamiento de Aspergillus spp. (AI) en paciente críticos con neumonía por influenza en las primeras 24h de ingreso.
DiseñoAnálisis secundario de estudio de cohortes observacional y prospectivo.
ÁmbitoUnidades de cuidados intensivos (UCI) participantes de forma voluntaria en el registro español de neumonía por influenza grave, desde junio de 2009 hasta junio de 2019.
PacientesPacientes consecutivos con diagnóstico de neumonía grave por influenza, confirmado por prueba de reacción en cadena de la polimerasa.
IntervencionesNinguna.
Variables principalesIncidencia de AI. Variables demográficas, comorbilidades, necesidad de ventilación mecánica y presencia de shock al ingreso. APACHE II.
ResultadosSe analizaron 3.702 pacientes. La incidencia de AI fue del 1,13% (n=42). Las neoplasias hematológicas (OR: 4,39; IC 95%: 1,92-10,04); VIH (OR: 3,83; IC 95%: 1,08-13,63) y otras situaciones de inmunosupresión (OR: 4,87; IC 95%: 1,99-11,87) fueron variables que se asociaron de forma independiente con AI. El árbol de decisión de CHAID mostró que la variable neoplasias hematológicas era la más relacionada con la variable Aspergillus spp. con una incidencia del 3,3%. Neoplasias hematológicas (OR: 2,62; IC 95%: 1,95-3,51), inmunosupresión (OR: 2,05; IC 95%: 1,46-2,88) y AI (OR: 3,24; IC 95%: 1,60-6,53) se asociaron de forma independiente con mayor mortalidad en la UCI.
ConclusionesEl tratamiento antifúngico empírico en nuestra población estaría justificado en los pacientes con inmunosupresión. En los pacientes con riesgo moderado-grave, la búsqueda activa de Aspergillus spp. debería implementarse.
Respiratory infections caused by the influenza virus can develop severe pneumonia and acute respiratory failure requiring Intensive Care Unit (ICU) admission. One of the possible complications of influenza pneumonia is the presence of community-acquired respiratory co-infection (CARC).1 Our group reported an incidence of coinfection close to 17%, and it was observed an independent association of CARC and ICU mortality.1
In recent years, the incidence of Influenza-Associated Pulmonary Aspergillosis (IAPA) has increased, and currently the reported incidence rates range from 9% to 23%.2–5 The appearance of IAPA worsens the prognosis in mechanically ventilated patients, with a mortality rate reported between 50% and 60%.2,3,6 However, the incidence of IAPA in critically ill patients and the risks factors related to the development of this entity vary among currently available reports, possibly due to the populations considered, the different definitions used, and the diverse active microbiological search strategies carried out.
Knowing the incidence of Aspergillus spp. isolation (AI) in critically ill patients would help to better characterize the affected type of population and therefore to identify the need to initiate early empirical antifungal treatment. In this context, the aims of our study were to determine the incidence and impact on ICU mortality of AI during the first 24h of admission in a large population of critically ill patients admitted due to proven severe influenza infection.
Material and methodsThis is a secondary analysis of an observational and prospective cohort study. Data were obtained from a voluntary registry created by the Spanish Society of Critical Intensive Medicine and Coronary Units (SEMICYUC, Spanish acronym), in which 184 Spanish ICUs participated between June 2009 and June 2019. The study was approved by the Ethics Committee of the Joan XXIII University Hospital (IRB # 11809) and by each of the participating centers. Informed consent was not obtained from the patients given the observational nature of the study, and the anonymity of the data was guaranteed.
Study populationAll consecutive patients who required admission to the ICU with a diagnosis of respiratory infection by influenza A H1N1, A seasonal, B or other virus were included during the period described. The presence of the virus was determined by performing a real-time polymerase chain reaction (rt-PCR) at each hospital, according to the recommendations of the Infectious Diseases Society of America (IDSA).7 Only patients with respiratory symptoms were analyzed, either due to pneumonia, exacerbation of asthma or acute exacerbation of chronic obstructive pulmonary disease (COPD). The clinical diagnosis of pneumonia was determined by the current American Thoracic Society (ATS)/IDSA guidelines, including the presence of 2 or more of the following signs and symptoms: fever (>38°C), cough, bronchial expectoration, and myalgia. These manifestations had to be associated with at least one organ failure (respiratory failure, hemodynamic instability, renal failure or sensorial disturbance). Patients under 15 years and those admitted to the ICU for reasons other than respiratory causes were excluded.
Study variablesDemographic data, comorbidities, laboratory and clinical results were collected. We recorded the need for mechanical ventilation and the presence of shock at ICU admission Severity of illness was determined using the Acute Physiology and Chronic Health Evaluation II (APACHE II) scale calculated on ICU admission. Renal Replacement Therapy (RRT) was recorded during ICU stay.
Definitions of Pulmonary Aspergillosis (PA) and microbiological search for isolation of Aspergillus spp. in respiratory samplesPA diagnosis was defined according to criteria modified recently proposed by Verweij et al.8: Proven PA was defined by lung biopsy showing invasive fungal elements and Aspergillus spp. growth on culture or positive Aspergillus spp. PCR in tissue. Probable PA was defined with pulmonary infiltrate and bronchoalveolar lavage (BAL) culture OR cavitating infiltrate and positive sputum/tracheal aspirate for Aspergillus spp. Possible PA was defined with pulmonary infiltrate and positive tracheal aspirate or mini-BAL culture.
The diagnostic procedures for Aspergillus spp. active search were not standardized and were requested upon the attending physicians’ consideration. No information regarding levels of galactomannan in BAL or serum were recorded in our general registry of patients with severe influenza virus pneumonia. Therefore, we do not have data in this regard.
ObjectivesThe primary objective of our study was to determine the incidence of AI in respiratory sample within the first 24h of ICU admission. The secondary objectives were to evaluate the general characteristics of critically ill patients with AI, and to determine the risk factors on ICU mortality in patients with influenza infection.
Statistical analysisQualitative variables were expressed as percentages while quantitative variables, as mean and standard deviation (SD) or median and interquartile range 25–75%. In order to determine clinical differences among groups, the Chi-square and Fisher tests were used for categorical variables, and the Student's t or Mann–Withney U test, for quantitative variables.
A multivariate analysis was performed using binary logistic regression (linear analysis) to determine the risk factors for developing AI and to determine the association of variables with ICU mortality. In addition, an automatic and non-linear analysis (Chi-square automatic interaction detection [CHAID]) was performed to determine which variables are most closely related to AI.
A CHAID tree is a graphic representation of a series of decision rules. Beginning with a root node that includes all cases, the tree branches are divided into different child nodes that contain a subgroup of cases. The criterion for branching (or partitioning) is selected after examining all possible values of all available predictive variables. In terminal nodes, a grouping of cases is obtained, in such a way that cases are as homogeneous as possible regarding the dependent variable value.9,10 CHAID decision trees are nonparametric procedures that make no assumption of underlying data. This algorithm determines how continuous and/or categorical independent variables best combine in order to predict a binary outcome based on “if-then” logic by portioning each independent variable into mutually exclusive subsets founded on homogeneity of the data. For this study, the response variable is the presence or absence of Aspergillus spp. in respiratory samples.
The statistical analysis and the CHAID tree were performed with the statistical program SPSS 20.0 for Windows (IBM). A value of p<0.05 is considered significant.
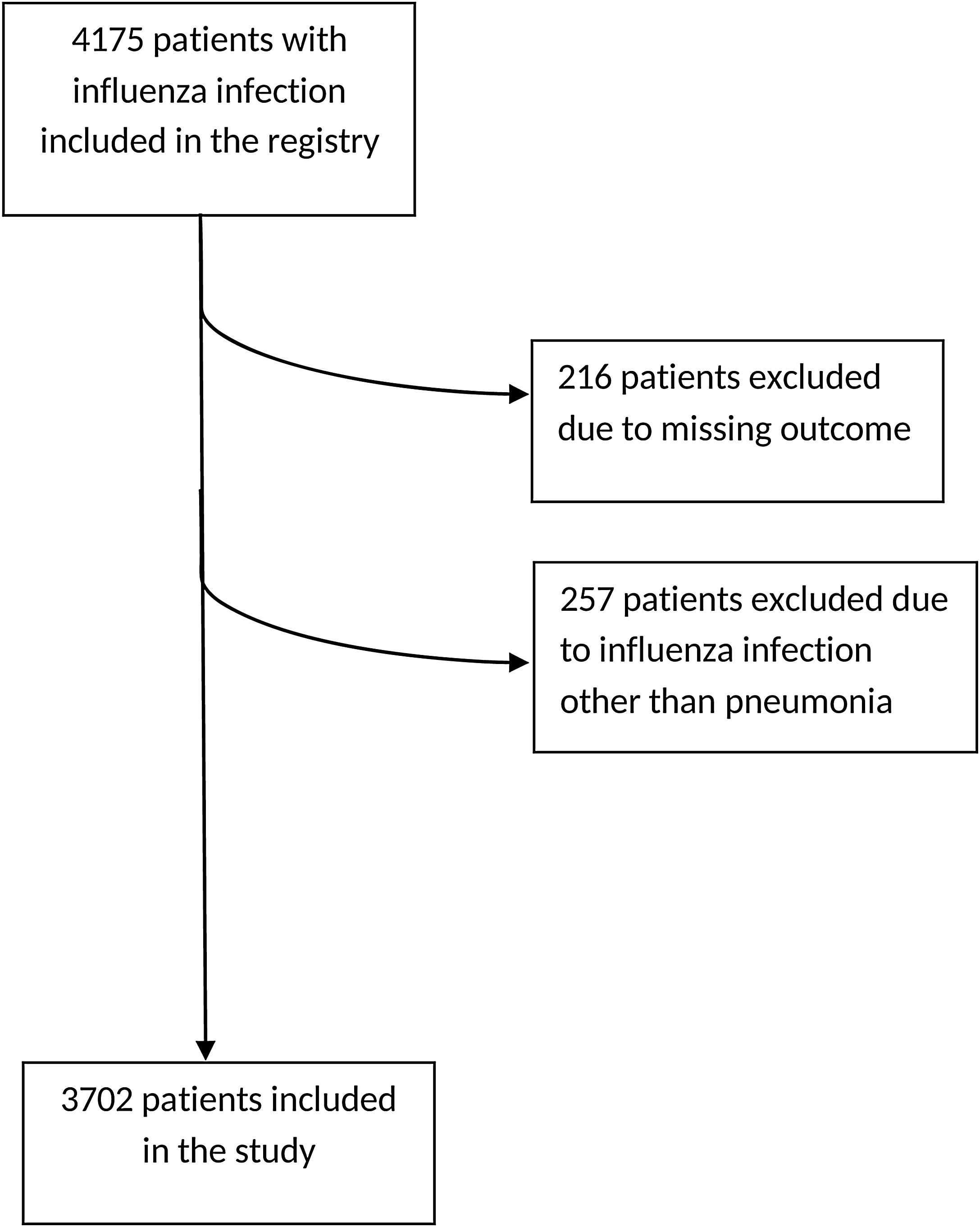
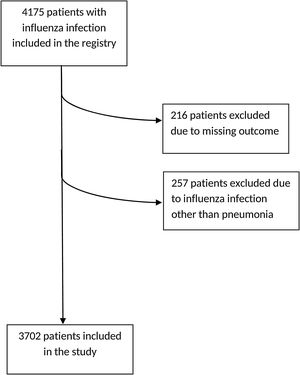
ResultsGeneral characteristicsA total of 4175 patients were included in the initial registry, from which 216 patients were excluded due to lack of clinical outcome on admission, and also 257 other patients, since they had been included in the database but presented influenza infections other than pneumonia, leaving a total of 3702 patients for the present study (Fig. 1). The general characteristics of the population are shown in Table 1.
General characteristics of the population.
| Variables | Total n=3702 | Non-AI patients n=3660 | IA patients n=42 | p value |
|---|---|---|---|---|
| Age, years – mean (SD) | 54.8 (15.9) | 54.7 (15.9) | 59.6 (13.0) | <0.05 |
| Sex, male – n (%) | 2203 (59.5) | 2170 (59.3) | 33 (78.6) | <0.05 |
| APACHE II – mean (SD) | 17.0 (7.9) | 16.9 (7.9) | 20.4 (7.6) | <0.05 |
| Mechanical ventilation – n (%) | 2936 (79.3) | 2984 (79.1) | 42 (100) | <0.05 |
| Vasoactive drugs – n (%) | 1899 (51.3) | 1868 (51) | 31 (74) | <0.05 |
| RRT – n (%) | 375 (10.1) | 368 (10.1) | 7 (16.7) | 0.19 |
| CRP, mg/dl – mean (SD) | 29 (13.7–100) | 29.0 (13.2–100) | 35.5 (19.6–186) | 0.44 |
| PCT, ng/ml – median (IQR) | 0.7 (0.2–4.3) | 0.7 (0.2–4.3) | 1.6 (0.5–4.4) | 0.25 |
| Nosocomial influenza – n (%) | 247 (6.7) | 239 (6.5) | 8 (19) | <0.05 |
| Type of influenza virus – n (%) | ||||
| A H1N1 | 2620 (70.8) | 2590 (70.8) | 30 (71.5) | 0.92 |
| A seasonal | 764 (20.6) | 756 (20.6) | 8 (19) | 0.79 |
| B | 283 (7.7) | 279 (7.6) | 4 (9.5) | 0.64 |
| Other | 35 (0.9) | 35 (1) | 0 | |
| Comorbidities – n (%) | ||||
| Asthma | 367 (9.9) | 360 (9.8) | 7 (17) | 0.18 |
| COPD | 908(24.5) | 894 (24.4) | 14(33.3) | 0.20 |
| CHF | 447 (12.1) | 442 (12.1) | 5 (12) | 1 |
| CRD | 314 (8.5) | 308 (8.4) | 6 (14) | 0.16 |
| Pregnancy | 460 (12.4) | 458 (12.5) | 2 (4.8) | 0.16 |
| Obesity | 1178 (31.8) | 1165 (31.8) | 13 (31) | 1 |
| NMD | 106 (2.9) | 106 (2.9) | 0 | 0.63 |
| Autoimmune disease | 149 (4) | 147 (4.0) | 2 (4.8) | 0.68 |
| HIV positive | 86 (2.3) | 83 (2.3) | 3 (7.1) | 0.07 |
| Hematological disease | 272 (7.3) | 263 (7.2) | 9 (21.4) | <0.05 |
| Other IS | 193 (5.2) | 186 (5.1) | 7 (16.6) | <0.05 |
| Days until ICU admission, days – median (IQR) | 1 (1–2) | 1 (1–2) | 2 (1–5) | <0.05 |
| ICU LOS, days – median (IQR) | 9 (4–19) | 9 (4–18) | 14.5 (6–22.3) | 0.17 |
| Hospital LOS, days – median (IQR) | 17 (10–29) | 17 (10–29) | 21.5 (10.8–30) | 0.42 |
| Mortality – n (%) | 796 (21.5) | 771 (21.1) | 25 (59.5) | <0.05 |
AI: Aspergillus spp. isolation; APACHE II: acute physiology and chronic health evaluation II; RRT: renal replacement techniques; CRP: C-reactive protein; PCT: procalcitonin; COPD: chronic obstructive pulmonary disease; CHF: congestive heart failure; NMD: neuromuscular disease; HIV: human immunodeficiency virus; IS: immunosuppression; ICU: Intensive Care Unit; SD: standard deviation; IQR: interquartile range.
Aspergillus spp. was isolated in only in 1.13% (42/3702) of the included patients; among them, 4.8% (2/42) met criteria of proven PA, and the rest, 95.2% (40/42), had probable/possible PA. Given the small number of patients with AI and the smaller number of patients with proven PA, and in order to reach stronger comparisons, we considered all cases as AI.
AI patients presented a higher severity of illness according to APACHE II score upon ICU admission, and a greater need for invasive mechanical ventilation (IMV), and vasoactive drugs (Table 1). Among AI patients, 19.0 (8/42) had hospital-acquired influenza infection, and a higher incidence regarding non-AI patients (6.5% [239/3660] p<0.05). Only hematological malignancies (21.4% [9/42] vs 7.1% [263/3660]; p<0.05) and other immunosuppression (16.6% [7/42] vs 5.3% [186/3660]; p<0.05) were more frequent in AI patients (Table 1). Patients with Aspergillus isolation showed more days of hospitalization previous ICU admission (median 1 day, IQR 1–2 vs median 2 days, IQR 1–5, p<0.05, respectively) (Table 1).
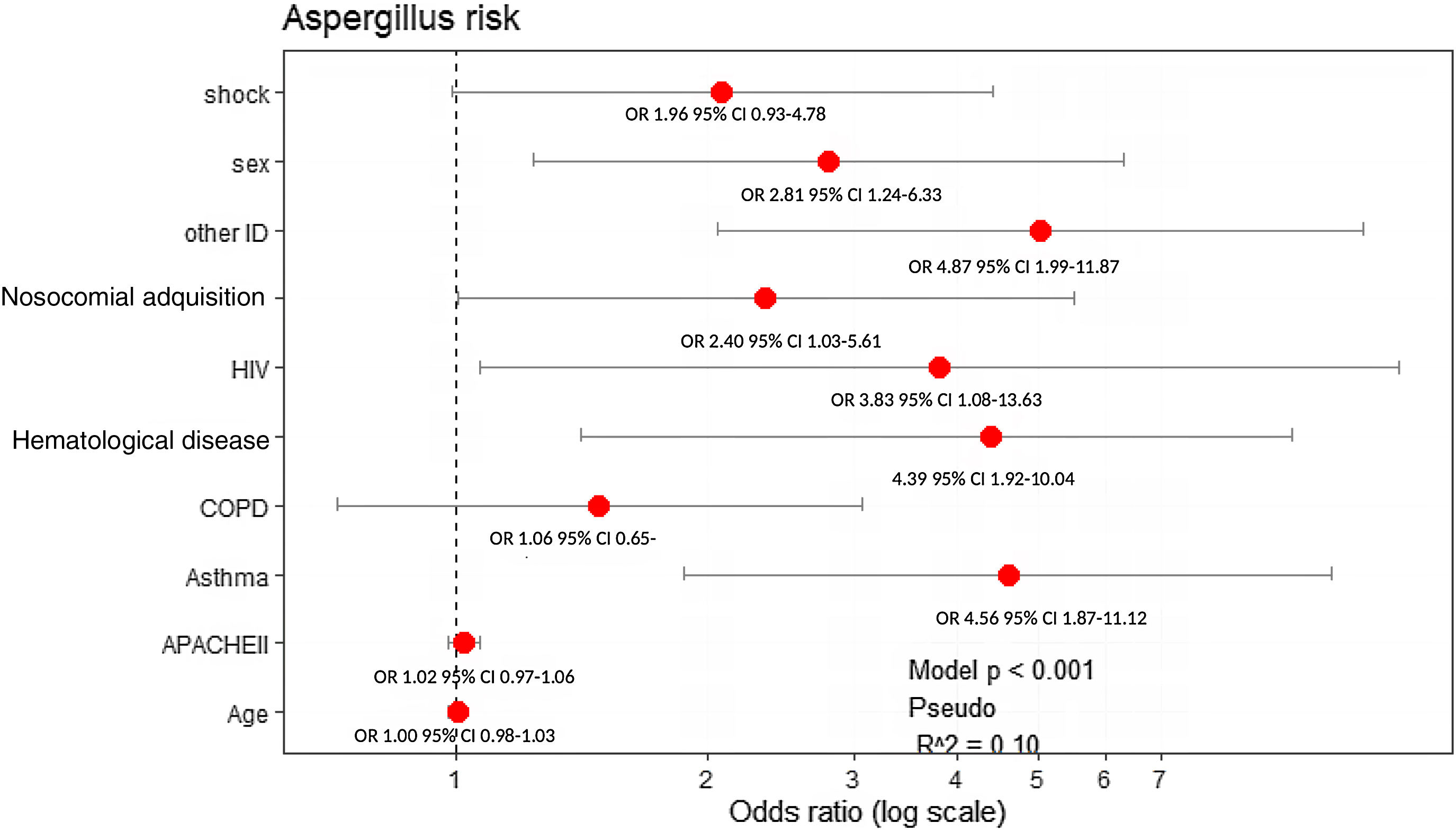
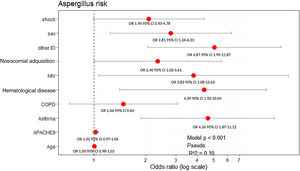
All variables that showed statistical significance in the univariate analysis (Table 1), asthma and COPD were included in the multivariate model due to their clinical importance. Male sex (OR: 2.81, 95% CI: 1.24–6.33), presence of asthma (OR: 4.56, 95% CI: 1.87–11.12), nosocomial-acquired influenza infection (OR: 2.40, 95% CI: 1.03–5.61), hematological malignancies (OR: 4.39, 95% CI: 1.92–10.04), HIV (OR: 3.83, 95% CI: 1.08–13.63), and other immunosuppression (such as chronic corticosteroid treatment or chemotherapy) (OR: 4.87, 95% CI: 1.99–11.87) were factors independently associated with AI (Fig. 2).
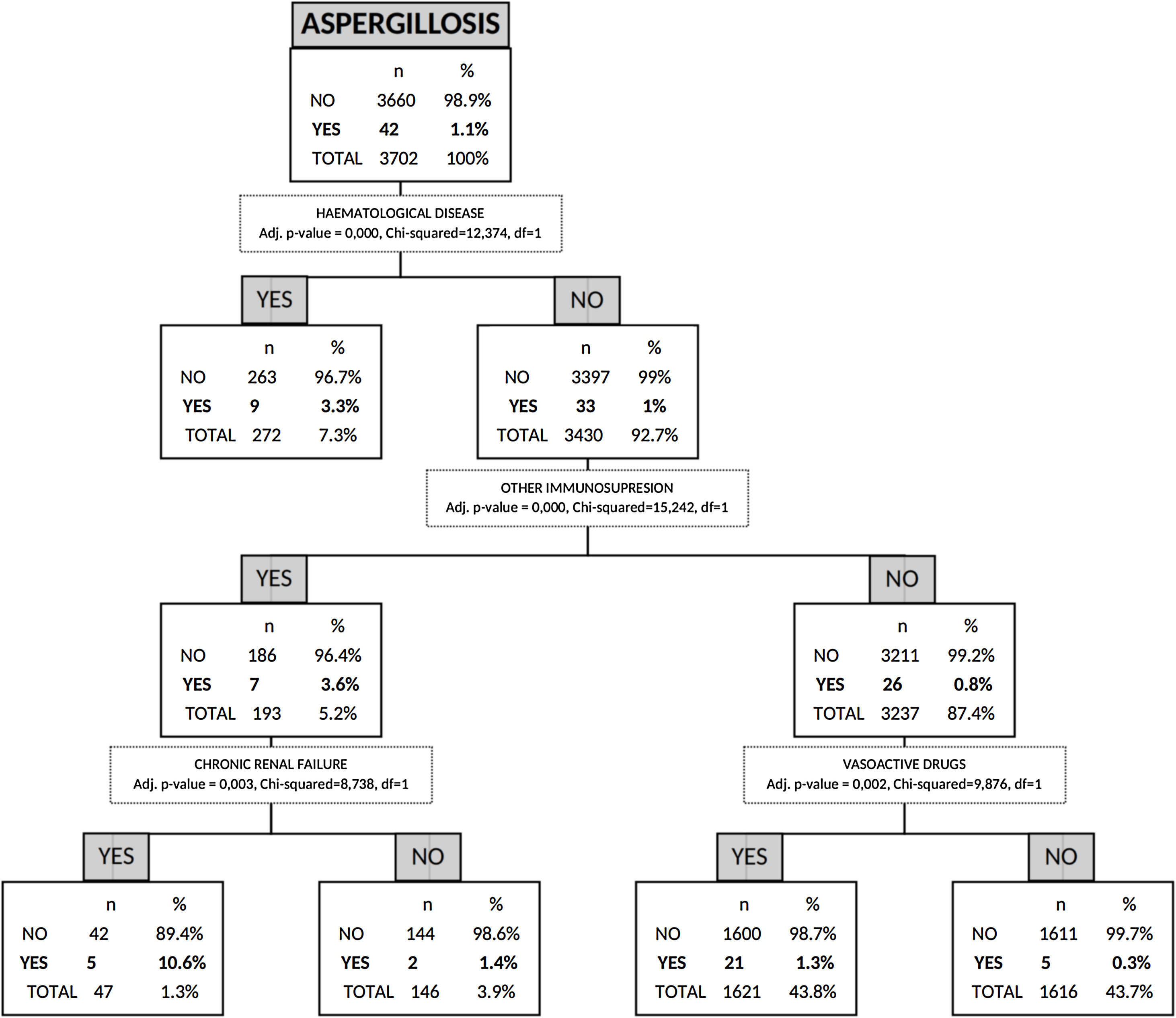
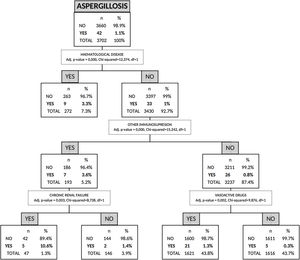
Risk factors for AI by non-parametric CHAID analysisIn the CHAID analysis, AI was the dependent variable and age, sex, APACHE II score on hospital admission, presence of shock, asthma, COPD, chronic renal failure, hematologic disease, human immunodeficiency virus (HIV), other immunosuppression and hospital-acquired influenza infection were included as independent variables. A tree with a maximum depth of 3 branches was generated, with a minimum of 50 cases in the parent node and 9 terminal nodes. The automatic CHAID decision tree showed that hematologic disease with an incidence of 3.3% was the variable most closely associated with AI followed of other immunosuppression as second branch division (Fig. 3).
Risk factors associated with mortalityThe crude ICU mortality of our cohort was 21.5% (796/3702). ICU mortality in the AI group of patients was 59.5% (25/42), almost three times than that observed in non-AI patients (21.0% [771/3660]) (p<0.05) (Table 1). Patients who died were older, had a higher level of severity (determined by APACHE II), and more often required invasive mechanical ventilation and vasoactive drugs on admission compared to survivors (Table 2).
Characteristics of the population depending on mortality.
| Survivorsn=2906 | Non-survivorsn=796 | p value | |
|---|---|---|---|
| Age, years – median (SD) | 53.8 (15.9) | 58.3 (15.6) | <0.05 |
| Sex, male – n (%) | 1691 (58.2) | 512 (64.3) | <0.05 |
| APACHE II – median (SD) | 15.7 (7.3) | 21.5 (8.5) | <0.05 |
| Mechanical ventilation – n (%) | 2173 (74.8) | 763 (95.9) | <0.05 |
| Vasoactive drugs – n (%) | 1276 (43.9) | 623 (78.3) | <0.05 |
| Nosocomial influenza – n (%) | 165 (5.7) | 82 (10.3) | <0.05 |
| AI – n (%) | 17 (0.5) | 25 (3) | <0.05 |
| COPD – n (%) | 747 (24) | 191 (22.6) | 0.4 |
| Asthma – n (%) | 327 (10.5) | 52 (6.2) | <0.05 |
| CHF – n (%) | 390 (12.5) | 141 (16.7) | <0.05 |
| CRF – n (%) | 242 (7.8%) | 113 (13.4%) | <0.05 |
| HIV – n (%) | 59 (1.9%) | 30 (3.6%) | <0.05 |
| Hematological disease – n (%) | 161 (5.2%) | 126 (14.9%) | <0.05 |
| Other IS – n (%) | 135 (4.3%) | 79 (9.3%) | <0.05 |
SD: standard deviation; APACHE: acute physiologic and chronic health evaluation; AI: Aspergillus spp. isolation; COPD: chronic obstructive pulmonary disease; CHF: chronic heart failure; CRF: chronic renal failure; HIV: human immunodeficiency virus; IS: immunosuppression.
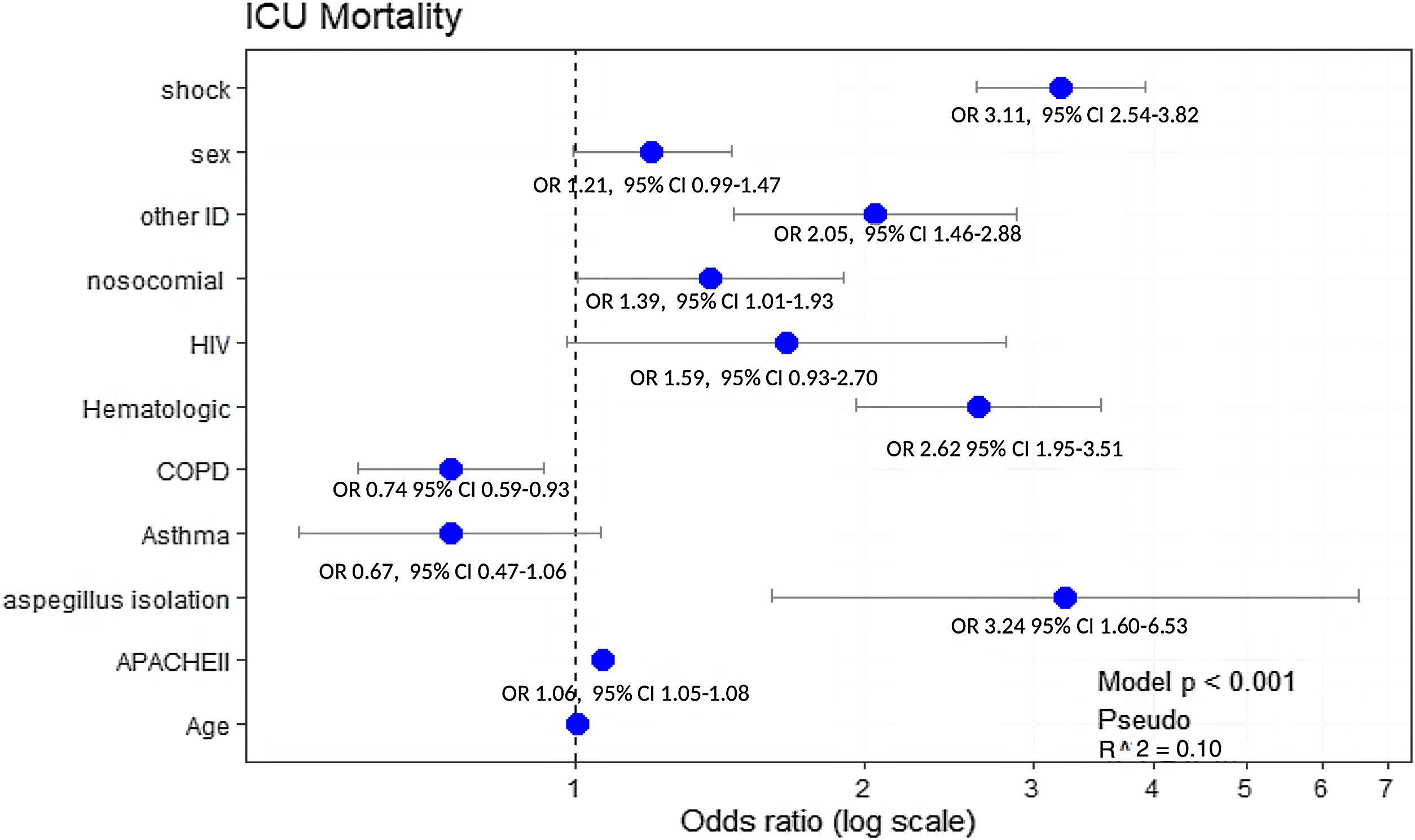
All significant variables associated with ICU-mortality (Table 2) were included in the multivariate analysis. Hematological disease (OR: 2.62, 95% CI: 1.95–3.51), immunosuppression (OR: 2.05, 95% CI: 1.46–2.88) and AI (OR: 3.24, 95% CI: 1.60–6.53) were the variables independently associated with ICU mortality. Notably, COPD showed a protective effect (OR: 0.74, 95% CI: 0.59–0.93) (Fig. 4).
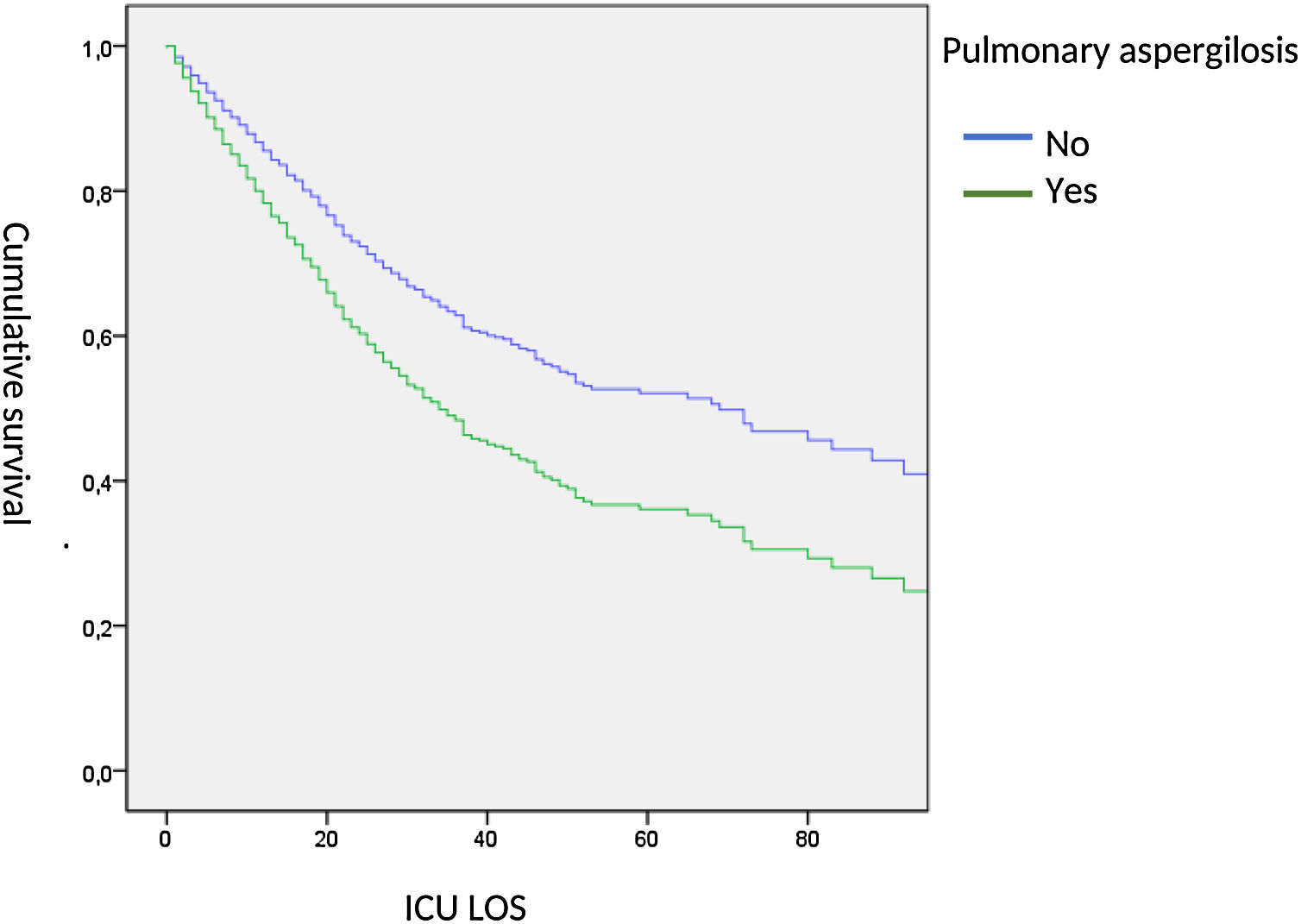
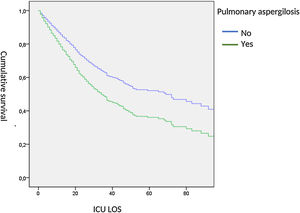
When adjusted, ICU-survival time was determined by Cox hazard proportional analysis, we observed a significant lower survival in AI patients (HR: 1.53, 95% CI: 1.03–2.38) (Fig. 5).
DiscussionThe main finding of our study is that the incidence of AI in respiratory sample of critically ill patients with severe influenza pneumonia is low in Spain, and it was closely associated with crude ICU mortality. Another interesting finding is that COPD did not appear as a AI associated factor, while hematological disease or other causes of immunosuppression were highly related with AI development.
To our knowledge, this is the first study to asses the incidence and impact of AI in patients with severe influenza pneumonia within the first 24h of ICU admission. Other studies have evaluated the Aspergillus incidence in an ICU evolution period. This difference makes it difficult to compare between studies. However, given the impact of our results, we believe it is important to highlight some considerations of clinical relevancy.
In our study, AI incidence is notably lower regarding the incidence of IAPA reported by other authors in other countries.3–5 The association between pulmonary aspergillosis (PA) and influenza infection or other respiratory viruses has been described previously.11–14 This association can respond to various factors. Viral infections directly injure the epithelium of the bronchial mucosa,11,12 facilitating the invasion by fungi present in the respiratory tract. Furthermore, influenza infection alters cytokines levels at both pulmonary and systemic levels, and the function of pulmonary macrophages and T-lymphocytes, producing a certain degree of immunodeficiency that favors Aspergillus spp. infection.13,14
The association between PA and viral infection is frequent in immunosuppressed patients. In a cohort of 219 patients receiving an allogeneic transplant,15 viral respiratory tract infection was shown to be a risk factor for the development of PA (HR: 4.3, 95% CI: 2–9.4). García-Vidal et al.4 reported an 8.8% incidence of IAPA in 57 immunosuppressed patients admitted to hospital due to influenza A(H1N1)pdm09 virus infection during the 2009 pandemic. The same author16 reported an association between influenza virus infection (HR: 2.5, 95% CI: 1.1–5.8) and parainfluenza (HR: 2.6, 95% CI: 1.6–4.3) with invasive infection by filamentous fungi. However, in critically ill patients IAPA incidence and risk factors are uncertain and highly variable.17–19 The low incidence of AI in our population (1.1%) differs from that published in other series. Wauters et al.5 observed a 23% incidence of PA in a cohort of 40 patients with influenza A(H1N1)pdm09 infection. The Dutch/Belgian mycosis study group has recently published3 an incidence of 19% of IAPA in patients with influenza A(H1N1) virus infection, clearly higher than the 5% observed in the control group of patients admitted for bacterial community-acquired pneumonia. Finally, this group also found a higher incidence of PA within the influenza cohort group of immunocompromised patients versus non-immunocompromised patients (32% vs 14%). These differences with respect to our results may be related to the characteristics of the studied populations, especially regarding the frequency of immunosuppressed patients, as well as the implemented diagnostic methods and the observation period. The determination of galactomannan in BAL, used in some studies20 for aspergillosis diagnosis, is a rarely used technique (<20%) in invasive mechanically ventilated patients with community-acquired pneumonia 21. The importance of immunosuppression in patients with hematological disease as a risk to AI has been confirmed by our automatic CHAID analysis; this relation was described by Vanderbeke et al.22 in a recent review. In this regard, the GLIMP study,23 a multicentric, international, observational point-prevalence study for patients hospitalized due to community-acquired pneumonia, reported that Aspergillusfumigatus as the causative pathogen was found more frequently in immunocompromised patients (8 [1.3%] vs 10 [0.4%]; p<0.01) and that AIDS (OR: 15.10, 95% CI: 6.36–35.88) and hematological cancer (OR: 4.65, 95% CI: 1.85–11.69) were independent risk factors for A.fumigatus.
Male sex, asthma, nosocomial-acquired influenza infection, and immunosuppression were the variables associated with the risk of presenting AI in our cohort of critically ill patients. The relationship between pulmonary disease (e.g., asthma and COPD) and PA has been previously studied. It has been proposed that either the chronic inflammatory disease itself or its management, including chronic corticosteroids treatment and broad-spectrum antibiotic therapy, could be the factors which explain this association.24,25 Notably, COPD was correlated with a lower risk of AI in our study. In a similar line, Schauwvlieghe et al.3 found that COPD was not an independent risk factor for IAPA (OR: 1.10, 95% CI: 0.53–2.26, p=0.80). Several studies describe COPD as a risk factor for aspergillosis, especially in patients receiving chronic corticosteroid treatment.19,26–29 In our population, the proportion of patients with COPD was similar in the group with and without AI, and within COPD patients, the use of corticosteroids was similar in patients with and without aspergillosis (1.8% vs 1.5% respectively, p=0.8, data not shown).
The association observed between nosocomial-acquired influenza and AI could be explained by the increased risk of hospitalized patients to acquire influenza as an expression of a subclinical condition of impaired immunity or immuno-paralysis associated with the serious processes that led to hospitalization, something that facilitates the development of opportunistic infections.30 In this regard, patients with AI were admitted in the ICU with a higher median of hospital days previous admission than patients without AI, but these differences were no significant in the multivariate analysis.
Finally, AI patients presented three times greater ICU-mortality than those in whom AI was not present (59% vs 21%). This high mortality coincides with the one published by Schauwvlieghe et al.3 with a mortality of 45% in patients with aspergillosis compared to 20% in the rest of the cohort patients, and with the study published by Vanderbeke et al.22 who observed a mortality of 57% in patients with aspergillosis and influenza infection.
The main strength of our study is that it is a homogeneous, multicenter cohort with a numerous number of critically ill patients with confirmed influenza infection, which allows the results to be generalizable. Nonetheless, we must recognize several limitations. First, it was a secondary analysis of an observational study, and although patients have been included consecutively, we cannot rule out possible biases in reporting the cases; yet, the high number of patients and the rigorous statistical analysis performed minimize these risks and make the results generalizable. Another major limitation is the fact that we cannot confirm proven PA diagnosis in most of our patients because we only have microbiological isolation and imaging tests; furthermore, we do not have information about in how many of our patients a bronchoscopy had been performed and diagnosis was made by BAL. In this way, there is the possibility that “colonized” patients may have been included. However, the low incidence of AI and high associated mortality make this scenario unlikely. Moreover, these are real-world data, because this is what clinician do during the current clinical practice, which makes our results very important for patient care and clinical practice. Another limitation could be related to the low number of patients with AI included. Despite this, the observed differences in mortality have achieved significance with a post-hoc calculated statistical power of 89%. Finally, it is possible that the low incidence observed is due to the lack of an active search investigation through the determination of galactomannan in BAL. Although we cannot rule out this possibility, this technique is not available in all hospitals in Spain.
ConclusionsDue to the low incidence of AI in our population of critically ill patients with influenza infection, the extensive use of empirical antifungal treatment in all patients may not be justified. In moderate-high risk patients, the active search for Aspergillus spp. should be systematically implemented.
FundingThis research received no external funding. This study was supported by the Spanish Intensive Care Society (SEMICYUC) and Ricardo Barri Casanovas Foundation (Alejandro Rodríguez). The study sponsors have no role in the study design, data collection, data analysis, data interpretation, or writing of the report.
Author contributionsL.C., X.D., I.M.-L., M.B., L.F.R. and A.R. designed the study, design of statistical analyses, and data analysis. All authors except G.Q., J.P, L.F.R. and J.J.G. contributed to the patient's data for the general database. L.C., X.D., I,M-L., M.B., F.G.-B., C.S., E.P., E.D., S.T., J.P., G.Q., J.M-C., L.F.R., J.S.-V., M.B-S., R.Z., J.J.G.., A.R. contributed to critical examination of the paper for important intellectual content and approval of the final manuscript. All authors have read and agreed to the published version of the manuscript.
Conflicts of interestThe authors declare that they have no conflict of interest.
The authors would like to thank the GETGAG group (Grupo Espanol de Trabajo de Gripe A Grave) SEMICYUC (Spanish Society of Critical Care). Complete list of GETGAG Investigators are shown in the Supplementary material.