Early warning systems (EWS) identify patients at risk with a view to improving morbidity and mortality rates using early therapeutic and transfer actions. We have recently implemented an EWS that focuses on two main aspects: the guidance of care after discharge from the ICU, and recognition of the onset of deteriorating health among adult patients in general wards through physiologically based early warning scores.
El Servicio Extendido de Cuidados Intensivos (SECI) pretende mejorar la morbimortalidad mediante la detección precoz del paciente grave fuera del ámbito de la UCI y poner en práctica acciones precoces, terapéuticas y/o de traslado. En nuestro hospital se ha implantado un SECI con unas características específicas que trabaja en dos vertientes fundamentales: el seguimiento de los pacientes dados de alta de la UCI y la detección de los pacientes graves fuera del ámbito de la UCI en base a la puntuación de un Sistema de aviso temprano.
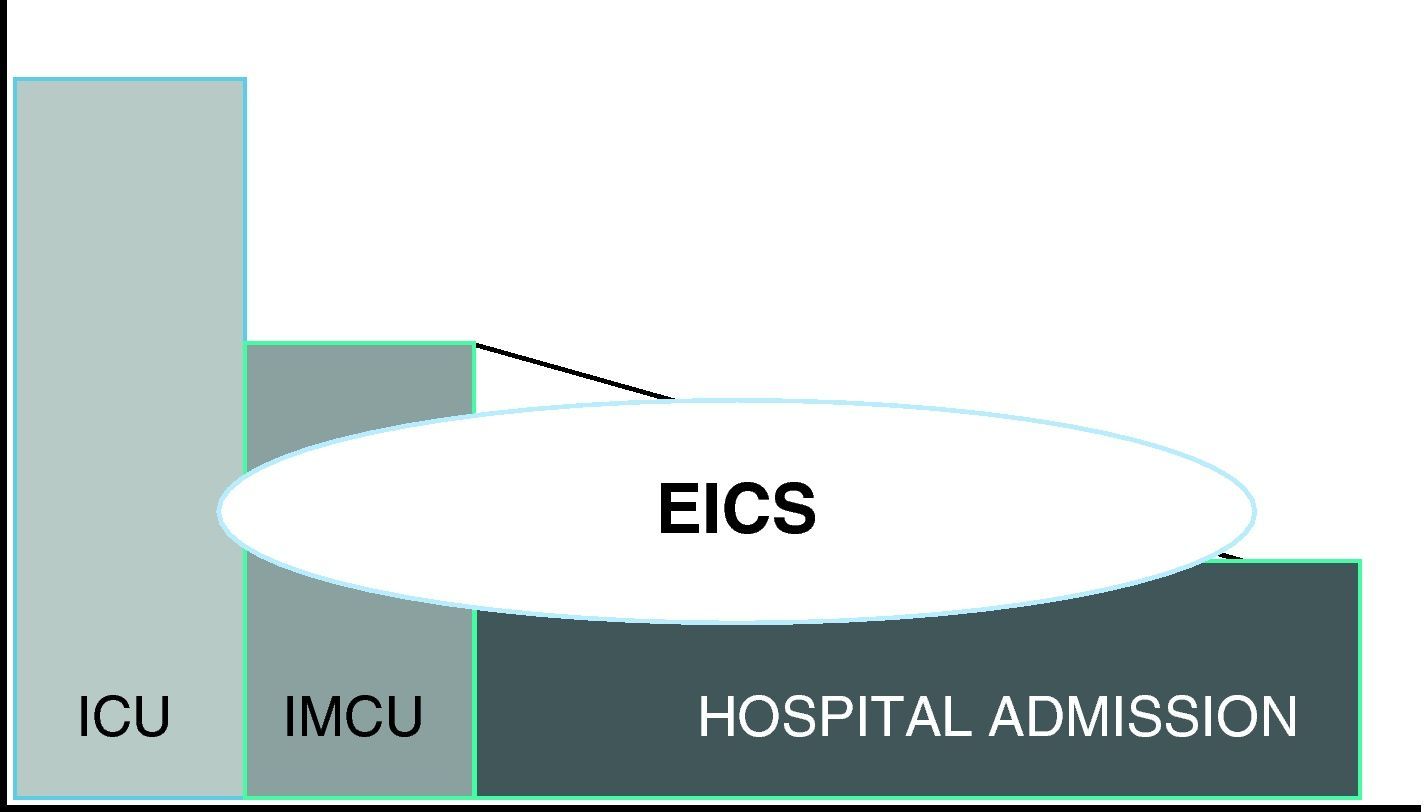
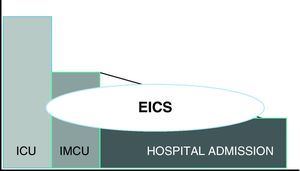
The concept of extended intensive care service (EICS) (referred to EWS: early warning systems) was introduced in response to the need for a more balanced and broad hospital approach to the management of patients “at risk”. It classifies seriously ill patients according to the need or level of care required, not according to the geographical constraints or limits of the place in which the patient is located.1,2
In this context, the literature describes different EWS for the detection and early treatment of those ward patients who experience a worsening of their clinical condition. These systems are fundamented upon teams mostly ascribed to multidiscipline ICUs, with differences in coverage and particularities dependent upon the characteristics of each hospital and on the cultural setting involved—though with a series of similar principles: the identification of patients at risk, based on an EWS score. Thus, extended teams with different nomenclatures can be found: Critical Care Outreach Teams (CCOT) or Patient at Risk Teams (PART) in the United Kingdom,3 Medical Emergency Teams (MET) in Australia,4 or Rapid Response Teams (RRT) in the United States.5
These teams were originally established in response to in-hospital cardiac arrest,4,6 with the purpose of reducing the incidence and improving the outcome in such cases, though their scope of intervention was expanded posteriorly according to the needs of the setting in which they were operative.7
However, and although these systems have been acknowledged as one of the strategies for preventing avoidable deaths, the efficacy of the Rapid Response Teams remains the subject of debate.8 The reasons for this are varied: heterogeneity of the different studies, differences in the actions taken or difficulties in assessing the results obtained, etc. In turn, the factors requiring intervention in order to change these results could be: clarification of the composition of these teams, definition and protocolization of tasks, or the conduction of cost-effectiveness studies capable of reliably demonstrating their usefulness.
Setting the basesDelays in treatment or inadequate care of ward patients often result in unplanned admissions to the ICU, a longer duration of hospital stay, and cardiac arrest or death.1 McQuillian9 showed that up to 50% of all patients admitted at hospital receive suboptimal care before admission to the ICU, and that up to 41% of all admissions to intensive care are potentially avoidable. Similar data have been reported by many other studies.10,11
The detection and diagnosis of severity, in the context of a disease process of any kind, is not always easy for the attending healthcare professionals. In some cases the existence of certain clinical signs is underestimated, whereas in other cases decision-taking is delayed or there are delays in seeking advice or consultation, for different reasons—all this leading to potentially life-threatening situations for the patient, or at least to delays in making use of the necessary therapeutic resources. The number of avoidable deaths and unplanned admissions to the ICU could be lessened if ward patients with a worsening of their clinical condition are identified quickly.12,13
This is the fundamental premise for the creation and operation of EICS: their activation must respond to the identification of patients at risk, based on the Early Warning System score.14
EWS are simple algorithms based on direct observation and recommended for the identification of patients at risk admitted to hospital wards. They are tools that have been developed to register certain previously determined physiological parameters, as well as their deviations from a given “normal” range, allowing identification through a well established circuit of patients at risk, and facilitating their evaluation by healthcare professionals highly trained in assessing seriously ill patients. Following an opportune and early diagnosis, these professionals in turn can resort to the necessary therapeutic means, or decide patient transfer to an ICU.15,16
Our experienceThis genuine need, well known to all of those who care for critical patients, can be addressed through different forms, structures, coverage specifications and facilities, etc. In summary, different characteristics may be involved, depending on the available resources and on the structure and/or complexity of the care profile of each individual hospital.
We offer a description of our experience (starting in July 2009) with the introduction of an EICS. As such, it is probably a pioneering service in our country, since to our knowledge it is the first of its kind in Spain.
Marqués de Valdecilla University Hospital is a third-level center in the city of Santander, with 900 beds. The General ICU (GICU) is divided into four modules:
- •
GICU-A (12 beds): designed for polyvalent medical patients, postoperative general surgery cases, and patients from specialized surgery and liver and pancreas transplantation.
- •
GICU-B (12 beds): designed for critical neurological patients, polytraumatized cases, postoperative specialized surgery patients, and lung transplantation cases.
- •
Maternal-children's ICU (8 beds): due to the characteristics of our center (currently undergoing construction work), this module receives polyvalent medical, pediatric and gynecological patients.
- •
Intermediate care unit (IMCU) (4 beds): this is a recently created module with four beds for non-critical seriously ill patients, as corresponds to the cases typically admitted to an intermediate care unit.
Within the specific context of our hospital, and for a number of years, the intensivists have alerted to the great difference in healthcare between the complete management received by patients in the ICU and the often insufficient care received by these same patients once they are moved to the hospital ward. Likewise, patients in the hospital ward are mostly admitted “late” to the ICU, because for one reason or other a series of parameters indicative of the seriousness of the situation are not identified. Even worse, another percentage of patients in turn conform the list of occult mortality in the ICU, due to failure to ensure follow-up adapted to their characteristics.
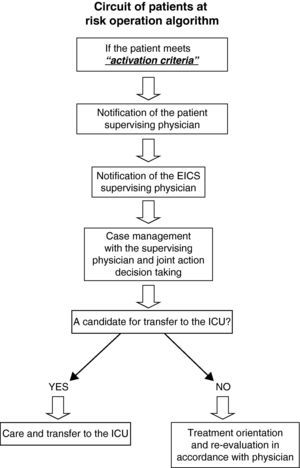
In this context, in June 2009, our hospital started what is now accepted as an EICS. Its creation involved several phases: a first phase in which the EICS project was defined; a second phase in which the activation criteria were established; a third phase for personnel training and diffusion among the implicated services; a fourth phase comprising the start of operability and periodic evaluation; and a fifth phase (the current phase) that completes the intensive care extension project with the opening of an intermediate care unit as a perfect complement to the ICU, and which as can be seen in Fig. 1 aims to care for the seriously ill patient beyond the physical limits of the ICU, thereby clearly reducing the aforementioned difference or defect in care between the ICU as such and the seriously ill ward patient scenario.17
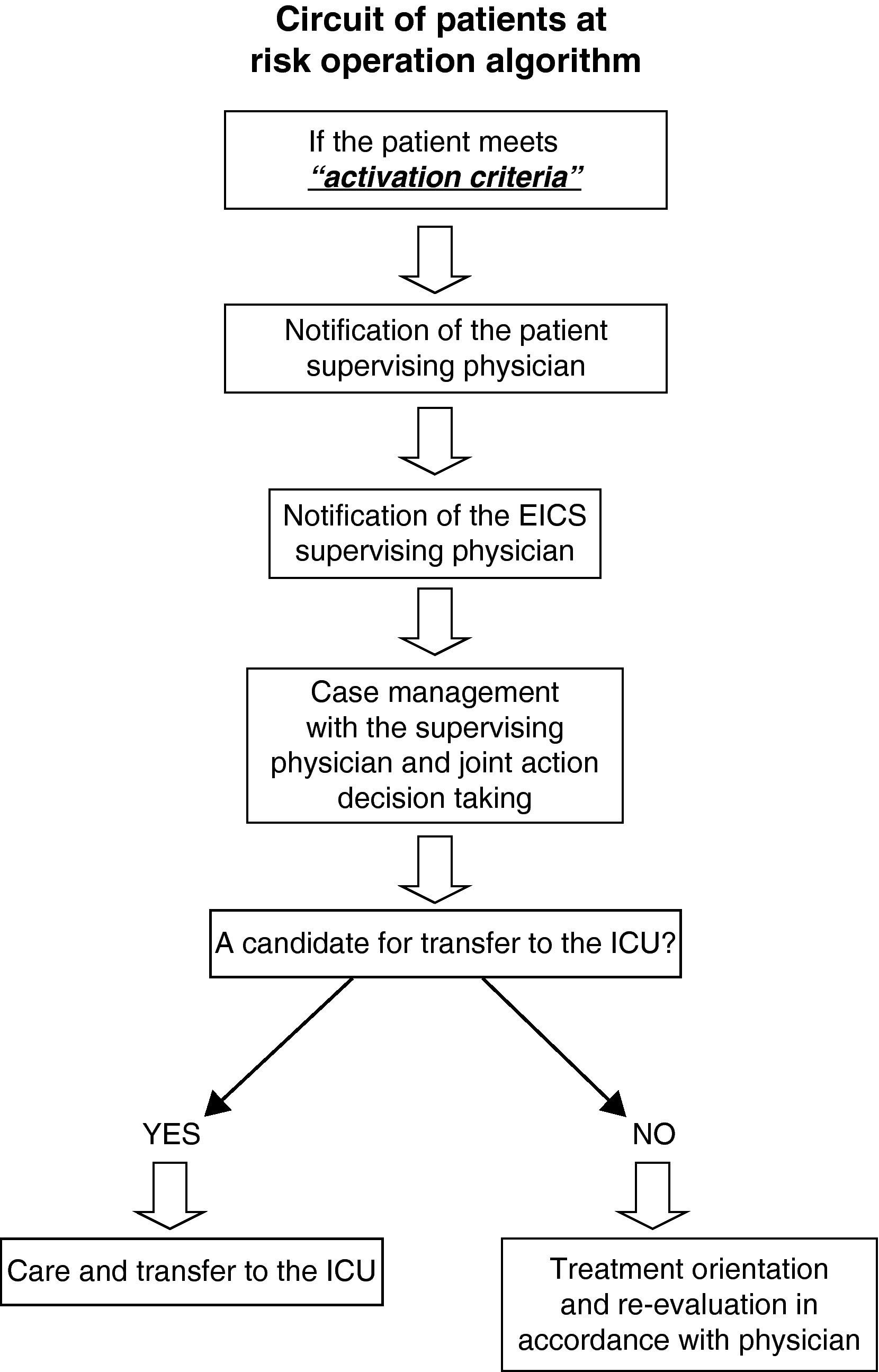
The EICS started its operations with an intensivist working in the morning (from 8:00 a.m. to 15:00 p.m.). Since the opening of the intermediate care unit (IMCU) 5 months ago, the activity of the EICS has also been extended to the afternoon shift. During the activity period of our extended service, the alerts are directed exclusively to the EICS physician, who together with the ICU physician attends, assesses and coordinates admission, if needed. The ICU physician on duty continues with his or her normal activities in the ICU. During the period of time not covered by the EICS (now 12h and holidays), alerts continue to be directed to the physician on duty as before. The operating protocol of the EICS comprises two aspects. On one hand, daily follow-up is made of each and every one of the patients who have been in the ICU, based on a checklist designed to the effect. On the other hand, the EICS takes charge of all those patients admitted to the hospital or to the Emergency Service who by meeting certain activation criteria may require intervention if so requested by the physician attending the patient. In this latter case, the EICS, always in close collaboration with the physician attending the patient, may decide the following:
- •
Treat the patient in the actual ward, with close follow-up over the subsequent days.
- •
Transfer the patient to the ICU or IMCU, according to the specific requirements of the case, and attending the patient during transfer.
- •
Collaborate with the physician in establishing correct limitation of therapeutic effort, if so warranted by the condition of the patient. This conforms the so-called circuit of the seriously ill patient (Fig. 2). This second aspect differs considerably from the classical call to the intensivist on duty, since there are significant and important differences such as:
- •
Rapid response (under 10min) – this is not always being possible with the usual activity of the intensivist on duty.
- •
Follow-up and treatment of the patient in the ward if transfer to the ICU is not required.
- •
Earlier calls to the intensivist, since admission to the ICU is not involved – the activities being limited to the assessment and optimization of measures, and patient selection avoiding unnecessary admissions and/or even facilitating limitation of therapeutic effort as established by consensus among the physician attending the patient, the family, and the intensivist.
Thus, the strategy of the EICS is not mainly to prevent cardiorespiratory arrest, this being regarded as too extreme an objective, but rather to improve morbidity–mortality through the early detection of patients at risk (seriously ill patients) outside the setting of the ICU, and to implement early therapeutic and/or transfer measures to special areas (ICU/IMCU).
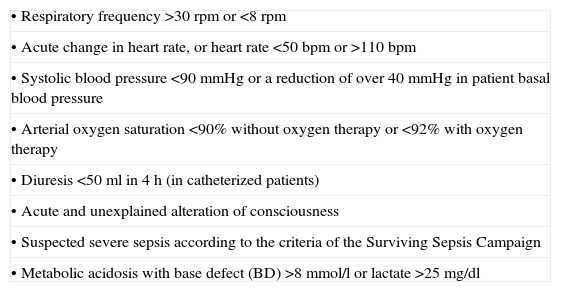
Table 1 describes the activation criteria based on the literature sources and revised by our service following a consensus-seeking process. Activation is based on the alteration of certain easily applicable physiological and/or laboratory test parameters. In the case of such alterations, the physicians caring for the patient can activate the EICS independently of where the patient is actually located.
EICS activation criteria.
| • Respiratory frequency >30rpm or <8rpm |
| • Acute change in heart rate, or heart rate <50bpm or >110bpm |
| • Systolic blood pressure <90mmHg or a reduction of over 40mmHg in patient basal blood pressure |
| • Arterial oxygen saturation <90% without oxygen therapy or <92% with oxygen therapy |
| • Diuresis <50ml in 4h (in catheterized patients) |
| • Acute and unexplained alteration of consciousness |
| • Suspected severe sepsis according to the criteria of the Surviving Sepsis Campaign |
| • Metabolic acidosis with base defect (BD) >8mmol/l or lactate >25mg/dl |
After 18 months of activity, and having attended over 1000 patients, we consider that the results (soon to be published) are clearly positive, both referred to hospital care activity considered as a whole, and as concerns the activity of the ICU proper. Specifically, a total of 1302 patients have been attended, of which 492 corresponded to “alerts to the EICS” and 810 corresponded to “follow-up after discharge from the ICU”.
We have created a service which is now known and respected throughout the hospital. Collaboration with other services (medical and surgical) is close, with clear improvement of the times related to the evaluation, follow-up, treatment and transfer of seriously ill patients. Educational activities have been carried out, with specific information sessions and the distribution of graphic cards describing the activation criteria and the circuit of the seriously ill patient, in each of the medical-surgical services in which the EICS operates.
A derived consequence of great importance for intensivists is the capacity to apply a measure of control of the serious pathology in the hospital, thus making it possible to better and more quickly select the appropriate patients and to improve general communication between the ICU and other services—this not always being easy in hospital with over 900 beds and different units, with responses that are not always uniform. We consider this to be one of the reasons explaining improvement in certain management parameters of the ICU itself.
ConclusionsThe EICS is defined as a structure of the ICU in charge of detecting seriously ill patients located outside the ICU, based on the definition of a series of activation criteria with a view to assessing, treating, and eventually transferring the patient to the most appropriate place for care (ICU-IMCU) in a faster and more effective manner.
The aim of the circuit of the seriously ill patient is therefore the detection and adequate treatment of the patient at risk, in the shortest time possible, and in the best place possible. This aim is achieved from the first moment, with the intensivist arriving at the patient bedside, applying treatment in situ, accelerating the decisions relating to transfer, and coordinating the latter in the most effective and fastest way possible.17,18
Theory suggests that extended services, though the detection and early treatment of seriously ill patients admitted to hospital wards, will contribute to improve treatment quality and the prognosis of such patients.19–21
Through the incorporation of an EICS, we will be able to show that intensivist intervention outside the physical limits of the ICU results in improved care and survival of the hospitalized seriously ill patient.
In addition, and independently of compliance with the criteria described above, consideration is made of ALL patients in which the supervising physician considers that the clinical situation is of concern and may pose a threat to the life of the patient.
Please cite this article as: Holanda Peña MS, et al. SECI (Servicio Extendido de Cuidados Intensivos): Mirando fuera de la UCI. Med Intensiva.2011;35:349–53.